Purpose
To evaluate the effectiveness of bepotastine besilate ophthalmic solutions 1.0% and 1.5% compared with placebo at reducing ocular itching and conjunctival hyperemia in the conjunctival allergen challenge (CAC) model of allergic conjunctivitis.
Design
Prospective, double-masked, randomized, placebo-controlled, phase 3 CAC clinical trial.
Methods
This multicenter trial enrolled 130 subjects with a clinical history of allergic conjunctivitis who were randomized to bepotastine besilate ophthalmic solution 1.0%, 1.5%, or 0.0% (placebo). One drop of test agent was instilled bilaterally before a CAC test evaluating responses at 15 minutes, 8 hours, or 16 hours after test agent instillation. Primary efficacy outcomes were unit improvements relative to placebo in mean scores for ocular itching and conjunctival hyperemia, each graded on 0- to 4-unit scales.
Results
Reductions of 1.2 units or more in mean ocular itching scores at all time points for both bepotastine besilate ophthalmic solutions 1.0% and 1.5% were observed at onset of action and 8-hour duration-of-action CAC tests ( P < .0001). Statistically significant reductions in conjunctival hyperemia ( P ≤ .0125) were observed for bepotastine besilate ophthalmic formulations only at the onset of action CAC test.
Conclusions
Bepotastine besilate ophthalmic solutions 1.0% and 1.5% both substantially decreased CAC-induced ocular itching for at least 8 hours after dosing. Reductions in conjunctival hyperemia after a CAC, although statistically significant for bepotastine besilate ophthalmic solutions 1.0% and 1.5% compared with placebo when assessed at 15 minutes after dosing, were modest.
Allergic conjunctivitis affects approximately 20% to 30% of the global population, with an increasing incidence. The hallmark symptom and sign of allergic conjunctivitis are ocular itching and conjunctival redness (hyperemia), respectively. The constellation of allergic conjunctivitis symptoms and signs also may include ocular chemosis, mucous discharge, eyelid swelling, and rhinitis and originates from a complex pathophysiology involving molecular, genetic, and environmental factors. In the allergic individual, these potential instigators of the ocular allergic response result in an immunoglobulin E-induced mast cell degranulation and the subsequent release of numerous mediators. Specifically for allergic conjunctivitis, histamine released from ocular mast cells binds to H 1 receptors on conjunctival nerves and results in ocular itching. Despite the value of earlier ophthalmic medications for treating allergic conjunctivitis, patients and physicians request novel options for obtaining symptomatic relief.
Bepotastine besilate {(+)-(S)-4-{4-[(4-chlorophenyl)(2-pyridyl) methoxy] piperidino} butyric acid monobenzenesulfonate} is a selective H 1 receptor antagonist that stabilizes mast cell function and inhibits eosinophilic migration and activation. The oral tablet formulation of bepotastine besilate (Talion; Mitsubishi Tanabe Pharmaceutical Company, Osaka, Japan) was approved by the Japanese Ministry of Health, Labour, and Welfare in 2000 to treat allergic rhinitis and in 2002 to treat urticaria and pruritus resulting from dermatosis (eczema or dermatitis, prurigo, cutaneous pruritus). Bepotastine besilate ophthalmic solutions 1.0% and 1.5% decreased ocular itching compared with placebo for at least 8 hours after dosing in a single-site, randomized clinical trial in the United States using the conjunctival allergen challenge (CAC) model of allergic conjunctivitis.
This multisite clinical trial tested the hypothesis that bepotastine besilate ophthalmic solution 1.0% or 1.5% may reduce the primary efficacy variables of CAC-induced ocular itching and conjunctival hyperemia with sustained clinical tolerability compared with placebo. In addition, the optimal dosing frequency and concentration of bepotastine besilate ophthalmic solution were evaluated.
Methods
Study Design
This was a double-masked, randomized, placebo-controlled, phase 3 CAC clinical trial conducted in 5 ophthalmic outpatient centers in Ohio, Tennessee, and California. Patient enrollment in this study occurred from November 2007 through March 2008. The 3 test agents were bepotastine besilate ophthalmic solution 1.0%, 1.5%, and 0.0% (placebo). All test agents were bottled and labeled identically except for a subject identification number. The trial was planned to evaluate approximately 130 subjects with bilateral instillation of the ophthalmic test agent and subsequent assessments of ocular itching and conjunctival redness after a CAC administered 15 minutes, 8 hours, or 16 hours after dosing at study visits spaced 2 weeks apart. The sample size was estimated to have more than 80% power to detect a true mean grade difference of 1.0 unit between 2 treatment groups, using a 2-sample t test and assuming a standard deviation of 1.0 unit for primary outcome variables graded on 0- to 4-unit scales.
Inclusion and Exclusion Criteria
Subjects to be included in the clinical trial had to be 10 years of age or older, to provide written informed consent before any study procedure took place, to have positive allergen skin test results within the previous 24 months, and to agree to avoid disallowed medications and contact lens use during the clinical trial period. A best-corrected visual acuity of 0.7 logarithm of the minimal angle of resolution units or better and intraocular pressure (IOP) of 5 to 22 mm Hg in each eye also was required. In addition, females capable of becoming pregnant had to have negative urine pregnancy test results at screening and agree to use a medically acceptable form of birth control during the clinical trial for at least 1 week before the first study visit and for 1 month after the last dose of test agent.
Subjects to be excluded from the clinical trial were those with any sign or symptom of clinically active allergic conjunctivitis in an eye at the start of any visit, an active ocular infection, a known history of retinal detachment or progressive retinal disease, or any ocular condition or other significant illness that in the opinion of the investigator could affect the subject’s safety or clinical trial parameters. A subject also could not be enrolled who had used an investigational drug or device or who was enrolled in another investigational drug or device study concurrently or within 30 days of this clinical trial. During screening visits (study visit 1 and visit 2), subjects were excluded from enrolling in the clinical trial if they did not manifest an ocular itching score of 2 or more (0 = none to 4 = incapacitating) and conjunctival redness score 2 or more (0 = none to 4 = extremely severe) in each eye.
Clinical Procedures
The observation procedures of the clinical trial primarily were designed to establish the degree of reduction in CAC-induced ocular itching and conjunctival redness and to quantify ocular comfort of bepotastine besilate ophthalmic solutions 1.0% and 1.5% compared with placebo. Investigators with previous CAC methodology experience conducted all clinical trial-related procedures and ophthalmic examinations. All ocular evaluations and interventions occurred bilaterally. Ocular itching and conjunctival redness after a CAC were assessed at 15 minutes after test agent instillation for onset of action and at 8 hours or 16 hours after dose administration for a potential prolonged effect. Ocular itching was graded by subjects at 3, 5, and 7 minutes after CAC, and conjunctival redness was graded by the investigator at 7, 15, and 20 minutes after CAC.
At visit 1 (day −21), eligible subjects were screened and graded on ocular itching and conjunctival redness using the CAC methodology. On completion of the visit 1 CAC, subjects underwent ocular examinations and IOP measurements. Subjects who continued to qualify were scheduled for visit 2. At visit 2 (day −14), each subject underwent a confirmatory screening CAC to assess clinical response reproducibility using the same allergen and concentration that elicited the positive clinical response at visit 1. Subjects with confirmed positive reactions for ocular itching and conjunctival redness in both eyes for at least 2 of 3 time points and meeting the inclusion and exclusion criteria qualified for enrollment in the study at visit 3 (day 0). At visit 3, qualifying subjects were assigned sequentially to 1 of the 3 treatment groups in a 1:1:1 ratio according to a computer-generated randomization list prepared by an independent biostatistician. The subjects were administered 1 drop of the test agent in each eye and evaluated their ocular comfort immediately on ophthalmic instillation and 5 minutes after instillation. Ocular comfort was quantified by enrolled subjects using a 4-point (0 to 3 units) ocular comfort grading scale (0 = comfortable to 3 = extremely uncomfortable or intolerable), with half-unit increments allowed. Subjects left the office and returned for a CAC test 16 hours after test agent instillation. At visit 4 (day 14 ± 3), subjects again received 1 drop of assigned test agent in each eye, subjects completed ocular comfort evaluations as at visit 3, and a CAC test was performed 8 hours after the test agent instillation. At visit 5 (day 28), enrolled female subjects capable of becoming pregnant received a second urine pregnancy test. All subjects then received a third and final drop of assigned test agent in each eye, and ocular comfort was assessed as at previous visits. Fifteen minutes after test agent instillation at visit 5, a CAC test was performed and subjects were exited from the clinical trial.
The safety assessments for bepotastine besilate ophthalmic solutions 1.0% and 1.5% included recording the incidence and frequency of adverse events. The incidence of specific adverse events was tabulated according to the Medical Dictionary for Regulatory Activities (MedDRA version 9.0) System Organ Class categorization. Other safety measurements in this clinical trial included visual acuity, slit-lamp biomicroscopy, IOP, and dilated funduscopy.
Statistical Analyses
The analyses of improvement in CAC-induced signs and symptoms of allergic conjunctivitis were conducted on the intent-to-treat population, using the last observation carried forward method for imputation of missing data. All statistical analyses and generation of randomization code were carried out using SAS statistical software (version 9.1.3; SAS Institute, Cary, North Carolina, USA). Statistical significance for differences in demographics among treatment groups, in mean ocular itching and conjunctival redness grades (placebo minus active), and in ocular comfort scores (active minus placebo) were determined using a parametric 2-sided statistical t test or, as appropriate, the Fisher exact test. To correct conservatively for bepotastine besilate ophthalmic solutions 1.0% and 1.5% comparisons with placebo and for examining 2 primary end points, treatment differences for ocular itching and conjunctival redness were considered statistically significant at visit 5 if either end point after application of corrections for multiplicity was significant at P ≤ .0125 for most time points. Treatment differences at visit 3 or visit 4 were considered statistically significant if P ≤ .00625 for most of the time points at either of these visits. The more stringent P value defined for visit 3 and visit 4 corrects for examining 2 primary efficacy end points, 2 bepotastine besilate ophthalmic formulations, and requiring that efficacy be demonstrated during only one of the duration-of-action CAC tests. A nonparametric Wilcoxon rank-sum test also was performed on data for ocular itching and conjunctival redness compared with placebo to assess the indifference of the primary outcome results to choice of statistical methodology, as was analysis of covariance across all time points at a clinical study visit.
Results
The intent-to-treat population for this study comprised 130 subjects enrolled at 5 study sites. The disposition of enrolled subjects is detailed in the Supplemental Figure . They were assigned to 1 of 3 treatment groups: bepotastine besilate ophthalmic solution 1.0% (44 subjects), 1.5% (43 subjects), and 0.0% (placebo; 43 subjects). The demographic characteristics of the 130 enrolled subjects are detailed in Supplemental Table 1 . Demographics in all categories (age, gender, ethnicity, race, and iris color) and the number of subjects in each treatment arm were well balanced among treatment groups ( P ≥ .125 in all cases). A total of 117 subjects completed the clinical trial, and no subject discontinued the trial because of an adverse event.
Efficacy Analysis
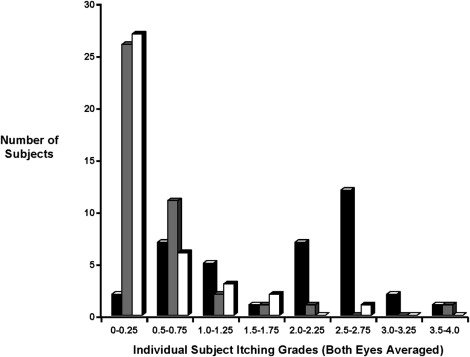
Within 3 minutes after a CAC and at every other time point after a CAC performed 15 minutes or 8 hours after test agent instillation, both bepotastine besilate ophthalmic solutions 1.0% and 1.5% demonstrated at least a 1.2-unit reduction for ocular itching ( P < .0001) when compared with placebo ( Table 1 ). Bepotastine besilate ophthalmic solution 1.5% demonstrated a slightly higher degree of reduced ocular itching than seen for the 1.0% formulation. Ocular itching results for bepotastine besilate 1.0% and 1.5% were substantially less at the 16-hour duration-of-action CAC test than at the other CAC tests ( Table 1 ). The diminished improvement in ocular itching at the 16 hour CAC test is not cosidered clinically meaningful. A representative plot of individual subject ocular itching scores for the 3-minute time point at visit 5 (onset of action CAC test) suggests that placebo data may be normally distributed, but the data for bepotastine besilate ophthalmic solution 1.0% or 1.5% is decidedly skewed to lower values ( Figure ). Despite the nonnormality of the bepotastine besilate ophthalmic solution data distributions, statistical significance was found for both parametric statistical tests ( t test) and nonparametric tests (Wilcoxon rank-sum test, analysis of covariance) at all CAC tests.
| Post-CAC Assessment Time (min) | Difference in Mean Ocular Itching Scores (Placebo a –Active) b | ||
|---|---|---|---|
| Bepotastine Besilate 1.0% (n = 44) | Bepotastine Besilate 1.5% (n = 43) | ||
| Onset-of-action CAC test | 3 | 1.4 | 1.5 |
| 5 | 1.5 | 1.6 | |
| 7 | 1.3 | 1.4 | |
| 8-hour duration-of-action CAC test | 3 | 1.2 | 1.3 |
| 5 | 1.3 | 1.3 | |
| 7 | 1.2 | 1.2 | |
| 16-hour duration-of-action CAC test | 3 | 0.6 | 0.6 |
| 5 | 0.7 | 0.7 | |
| 7 | 0.8 | 0.8 | |
a n = 43 subjects for placebo.
b The statistical significance for all differences in mean scores shown were P < .0001 by t test and by the Wilcoxon rank-sum test. Analysis of covariance statistics across all time points at a study visit also were P < .0001.
An improvement in conjunctival redness of 0.4 to 0.6 units was observed at most time points at the onset of action CAC test for both bepotastine besilate formulations ( P ≤ .0125). There was less conjunctival redness improvement seen at the 8- and 16-hour duration-of-action CAC tests ( Table 2 ).
| Post-CAC Assessment Time (min) | Difference in Mean Conjunctival Redness Scores (Placebo a –Active) b | ||
|---|---|---|---|
| Bepotastine Besilate 1.0% (n = 44) | Bepotastine Besilate 1.5% (n = 43) | ||
| Onset-of-action CAC test | 7 | 0.6 (0.0001) b | 0.4 (0.003) |
| 15 | 0.5 (0.002) | 0.4 (0.01) | |
| 20 | 0.2 (0.15) | 0.2 (0.23) | |
| 8-hour duration-of-action CAC test | 7 | 0.5 (0.0006) | 0.2 (0.11) |
| 15 | 0.3 (0.03) | 0.1 (0.36) | |
| 20 | 0.3 (0.10) | 0.1 (0.59) | |
| 16-hour duration-of-action CAC test | 7 | 0.4 (0.005) | 0.1 (0.55) |
| 15 | 0.4 (0.02) | 0.1 (0.39) | |
| 20 | 0.4 (0.04) | 0.1 (0.67) | |
a n = 43 subjects for placebo.
b Statistical significance by t test at each observation is shown in parentheses to 1 or 2 significant figures after the difference value.
Safety Analysis
A total of 40 adverse events in 31 subjects occurred during the clinical trial. There were 9 ocular and 31 nonocular adverse events ( Supplemental Table 2 ). The most common adverse events were nasopharyngitis (8.5% of all subjects), eye irritation (3.8%), and mild taste on instillation (3.1%). The nonocular adverse events were slightly higher with bepotastine besilate ophthalmic solution 1.0% or 1.5% than with placebo, with no apparent dose relationship for exposure to bepotastine besilate ( Supplemental Table 2 ). There were no reports of drowsiness or dry mouth. The adverse events generally were mild and transient, and no subject discontinued from the clinical trial because of an adverse event. Dry eye was reported for a single subject in each of the placebo and bepotastine besilate ophthalmic solution 1.0% treatment groups. All changes in visual acuity, IOP, and ocular comfort for all treatment groups were small ( Table 3 ) or there were no significant findings in ophthalmologic examinations. The mean ocular comfort score differences compared with placebo all approached 0 units, indicating both bepotastine besilate formulations were comfortable.
| Placebo | Bepotastine Besilate 1.0% | Bepotastine Besilate 1.5% | |
|---|---|---|---|
| Visual acuity a | |||
| Onset-of-action CAC test | 0.06 | 0.03 (0.25) b | 0.02 (0.07) |
| 8-hour duration-of-action CAC test | 0.08 | 0.04 (0.29) | 0.02 (0.12) |
| 16-hour duration-of-action CAC test | 0.08 | 0.04 (0.37) | 0.02 (0.25) |
| Intraocular pressure c | |||
| Initial screening visit | 15.5 | 15.9 (0.51) | 15.7 (0.65) |
| Onset-of-action CAC test | 15.2 | 15.7 (0.45) | 15.3 (0.91) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


