Purpose
To describe the results of a novel treatment approach to the acute ophthalmic management of Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN).
Design
Retrospective interventional case series.
Methods
setting: Institutional. study population: Sixteen eyes of 8 patients with acute, biopsy-proven SJS or TEN and significant ophthalmic involvement. interventional procedure(s): Application of amniotic membrane to the ocular surface, either in the operating room or at the bedside, and short-term use of intensive topical corticosteroid medication. main outcome measures: Visual acuity, slit-lamp appearance of the ocular surface, and patients’ subjective impression of ocular comfort.
Results
Two patients expired during the hospitalization. Mean follow-up time for the surviving patients was 7.7 months. Four surviving patients in whom the entire ocular surface (ie, the cornea, bulbar and palpebral conjunctiva, and eyelid margins) was treated with amniotic membrane retained visual acuities of 20/40 or better and an intact ocular surface. In contrast, the initial 2 patients in the study who were treated with only a Prokera device or unsutured amniotic membrane sheets, leaving the palpebral conjunctiva and eyelid margins uncovered, developed more significant ocular surface abnormalities, and 1 developed a corneal perforation.
Conclusions
Amniotic membrane coverage of the ocular surface in its entirety coupled with the use of intensive short-term topical corticosteroids during the acute phase of SJS and TEN is associated with the preservation of good visual acuity and an intact ocular surface. Partial amniotic membrane coverage of the ocular surface may not serve to minimize the cicatrizing ocular sequelae of SJS and TEN as effectively as complete coverage.
Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) encompass a spectrum of rare, acute systemic vesiculobullous disorders that result in extensive sloughing of the skin and mucous membrane surfaces. While the pathogenesis is unknown, they are thought to represent an aberrant immunologic response to an inciting agent, such as a medication (typically an antibiotic or an anticonvulsant) or, less commonly, an infectious agent. The skin detachment occurs at the epidermis-dermis junction and, while definitions vary, is generally categorized as involving less than 10% total body surface area in SJS, between 10% and 30% in SJS-TEN overlap syndrome, and greater than 30% in TEN. TEN and, to a lesser extent, SJS are associated with a high risk of mortality and these patients are often treated acutely in burn centers or other types of intensive care units.
The conjunctiva and oropharynx represent the most frequently involved mucosal surfaces. Ocular involvement has been reported in 69% and 81% of SJS patients and 50% and 67% of patients with TEN. The acute ophthalmic manifestations are typically that of a membranous or pseudomembranous conjunctivitis with varying degrees of conjunctival and corneal epithelial sloughing. The chronic consequences are that of a cicatrizing conjunctival process with symblepharon formation, severe dry eye, trichiasis, eyelid margin keratinization, and limbal stem cell deficiency, all of which combine to result in corneal ulceration and scarring and vision loss. In addition, these patients often suffer from debilitating chronic photophobia and eye pain.
Ophthalmic interventions during the acute stage have traditionally been supportive in nature, with aggressive lubrication, prophylactic topical antibiotics, and lysis of adhesions forming the cornerstones of treatment. However, it is well recognized that these ophthalmic interventions in general do little to improve the poor ophthalmic prognosis associated with this condition.
The application of amniotic membrane, the innermost layer of the placenta, has been used with success in the treatment of various ocular surface conditions and has been used in the attempted rehabilitation of the ocular surface in the chronic stages of SJS and TEN. Recently, a number of single- and 2-patient case reports have demonstrated improved outcomes in patients treated in the acute stages of SJS and TEN with amniotic membrane application. In addition, 2 recent articles describe a beneficial effect associated with the acute-stage use of systemic pulse corticosteroid therapy in combination with topical corticosteroid application.
The present study reports the results of amniotic membrane application to the ocular surface coupled with the use of intensive short-term topical corticosteroid medications in a consecutive series of 8 patients with acute SJS or TEN and significant ophthalmic involvement. In the majority of patients, the entire ocular surface, that is, 1) the cornea, 2) the bulbar and palpebral conjunctiva, and 3) the eyelid margins, was covered with amniotic membrane. Some patients underwent amniotic membrane application in the operating room and others, medically too unstable to withstand a trip to the operating room, at the bedside. These surgical settings required somewhat different approaches to achieving ocular surface coverage, but the goal of total coverage was the same. In contrast, the initial 2 patients in the study were treated with only the Prokera device (Bio-Tissue, Miami, Florida, USA), a sutureless method of amniotic membrane application, or unsutured sheets of amniotic membrane. These methods of application left the palpebral conjunctiva and eyelid margins uncovered, yielding only partial coverage of the ocular surface.
Methods
We retrospectively reviewed the medical records of a consecutive series of 8 patients (16 eyes) admitted to the New York–Presbyterian Hospital between April 25, 2007 and April 4, 2009 with a diagnosis of acute SJS or TEN. “Acute” was defined for the purposes of the study as onset of the skin rash within 2 weeks of presentation to the New York–Presbyterian Hospital. The criteria for inclusion in the study were: 1) histologic confirmation of the diagnosis on the basis of a skin biopsy obtained by the Weill Cornell Dermatology Service, and 2) significant ophthalmic involvement, consisting of, at a minimum, conjunctival injection with conjunctival membrane or pseudomembrane formation.
All patients underwent ophthalmic examination upon admission, and, if significant ophthalmic involvement as defined above was identified, amniotic membrane was applied to the ocular surface as soon as possible, and often within 24 hours of admission. Both eyes were treated in all patients. All procedures were performed by 1 of the members of the Weill Cornell Cornea Service (K.C.S., E.C.L., J.S.S., J.Y.).
In the majority of patients, amniotic membrane was applied in such a fashion that the ocular surface was covered in its entirety. Amniotic membrane was applied either in the operating room utilizing the technique described under “Surgical procedure: Amniotic membrane application in the operating room,” which was the preferred choice, or at the bedside (see below: “Surgical procedure: Amniotic membrane application at the bedside”) if the primary team deemed the patient medically too unstable to undergo transport to the operating room.
The first 2 patients in the series were treated with amniotic membrane in such a fashion that the ocular surface was only partially covered. The techniques utilized are outlined under “Surgical procedure: Amniotic membrane application in 2 early patients.”
The ophthalmic medical regimen in all patients consisted of frequent application of corticosteroid ophthalmic ointment (fluorometholone ointment 0.1%, applied every 1 to 2 hours). The corticosteroid medication was tapered as soon as there was evidence of healing of the skin and mucosal lesions, typically within 1 to 2 weeks of admission to the hospital. The typical infection prophylaxis regimen consisted of bacitracin ointment and a fluoroquinolone antibiotic (Vigamox; Alcon, Fort Worth, Texas, USA), but was modified depending on evidence of systemic colonization or infection with specific organisms. All patients also received topical cyclosporine (Restasis; Allergan, Irvine, California, USA) 4 times a day.
Surgical Procedure
Amniotic membrane application in the operating room
The procedure was performed under either general endotracheal anesthesia or local or topical anesthesia with intravenous sedation. Any eyelashes, if present, were trimmed. A topical application of 1:1000 epinephrine was used to aid hemostasis, if necessary.
The procedure consisted of 2 distinct steps: 1) application of amniotic membrane to the corneal and bulbar conjunctival surface, and 2) application of amniotic membrane to the eyelid margins and palpebral conjunctiva.
For the first portion of the procedure, the eyelids were retracted with a lid speculum. A 3.5 × 3.5-cm (or 2.5 × 2.0-cm) sheet of cryopreserved amniotic membrane (Bio-Tissue) was placed on the ocular surface with the basement membrane side facing away from the corneal surface. The orientation of the amniotic membrane was confirmed by touching a surgical sponge (i-Spear; Alcon) to the amniotic membrane—the “stickier” side being the stromal side and the less sticky side the basement membrane side. Under the operating room microscope, a 10-0 nylon suture (Ethicon, Somerville, New Jersey, USA), placed in a running purse-string fashion 1 to 2 mm behind the limbus, was used to secure the amniotic membrane to the corneal surface ( Figure 1 , Top left). The suture bites were used to include conjunctiva and episclera and the knot was buried. A variable number of additional 10-0 nylon sutures were placed more posteriorly to the limbus to further secure the amniotic membrane to the bulbar conjunctival surface. A large-diameter bandage contact lens (Kontur Kontact Lens Co, Hercules, California, USA), either a 22-mm or 24-mm size depending on the size of the eye, was then applied to the eye ( Figure 1 , Top right).
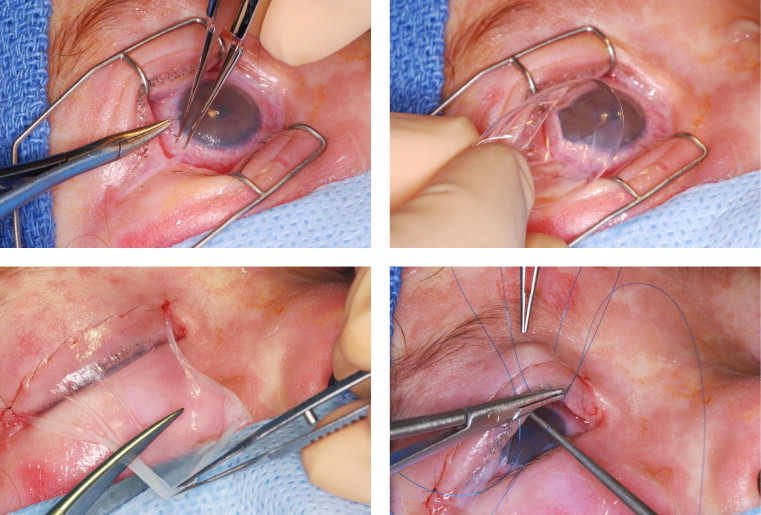
For the second portion of the procedure, the lid speculum was removed. A second 3.5 × 3.5-cm sheet of cryopreserved amniotic membrane was placed on the eyelid, again with the basement membrane side facing away from the corneal surface. One end of the sheet of amniotic membrane was sutured to the eyelid skin, close to the eyelid margin, using a running 8-0 nylon suture ( Figure 1 , bottom left). A muscle hook was then used to push the amniotic membrane into the fornix. Two double-armed 5-0 or 6-0 prolene sutures were then passed through the amniotic membrane, passed through the eyelid, and then secured over the skin with a bolster ( Figure 1 , Bottom right). The same procedure was performed for both upper and lower eyelids.
Amniotic membrane application at the bedside
At the bedside, the procedure was performed with local and topical anesthesia. Intravenous sedation with, for example, ketamine was used in young children.
As in the operating room, the procedure consisted of 2 distinct steps. For the first portion of the procedure, a Prokera device was used to achieve amniotic membrane coverage of the cornea and most of the bulbar conjunctiva. The Prokera device, a symblepharon ring with a sheet of amniotic membrane clipped to it, was applied to the ocular surface without the use of sutures in a manner akin to the placement of a bandage contact lens. Prokera does not reach into the fornices and does not cover the palpebral conjunctiva or eyelid margins.
The second portion of the procedure, the coverage of the palpebral conjunctiva and eyelid margins with amniotic membrane, was performed exactly as it was in the operating room, with the utilization of loupes as necessary to provide appropriate magnification ( Figure 2 ).
Amniotic membrane application in 2 early patients
Alternate bedside methods were used in the first 2 patients in the series; these yielded only partial amniotic membrane coverage of the ocular surface. In Patient 1 ( Table ), a Prokera device alone was initially applied, allowing for coverage of the cornea and most of the bulbar conjunctiva, but leaving the palpebral conjunctiva and eyelid margins uncovered. The Prokera device was removed a few days later and a sheet of amniotic membrane applied to the ocular surface in the following manner. A lid speculum was used to retract the eyelids and a large (3.5 × 3.5-cm) sheet of amniotic membrane placed on the corneal surface. The amniotic membrane was then swept into the fornices using a muscle hook and held in place with a 24-mm Kontur bandage contact lens. No sutures were used. This served to improve coverage of the forniceal conjunctiva; however, the palpebral conjunctiva and eyelid margins continued to remain uncovered. In Patient 2 ( Table ), solely a Prokera device was applied to each eye and the remainder of the ocular surface left uncovered. Amniotic membrane was not sutured to the palpebral conjunctiva and eyelid margins in either patient.
| Patient | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 |
|---|---|---|---|---|---|---|---|---|
| Age (years)/gender (M/F) | 5/M | 18/F | 82/M | 43/M | 10/F | 68/F | 2/F | 40/F |
| Diagnosis | TEN | TEN | TEN | TEN | TEN | TEN | SJS | TEN |
| Presumed inciting agent | Ibuprofen | Amoxicillin | Relafen (nabumetone) | Rosewood oil | Fluconazole | ? | Amoxicillin | Lamictal (lamotrigine) |
| Ophthalmic findings upon presentation | ||||||||
| Pseudomembranous conjunctivitis b | + | + | + | + | + | + | + | + |
| Conjunctival epithelial defect | + | + | + | − | + | + | + | + |
| Corneal epithelial defect | + | + | − | − | − | + | − | − |
| Amniotic membrane application | ||||||||
| Days placed after onset c | 8 d | 9 | 4 | 7 | 5 | 12 | 5 | 6 |
| Total ocular surface coverage e | + (bedside) | + (bedside) | + (OR) | + (bedside) | + (OR) | + (OR) | ||
| Partial ocular surface coverage f | + (bedside) | |||||||
| Partial ocular surface coverage g | + (bedside) | |||||||
| Ophthalmic outcome | ||||||||
| Follow-up time | 25 months | 12 months | Deceased | 1 month h | 1 month h | Deceased | 5 months | 2 months |
| BCVA OD | 20/20 | 20/50 | 20/20 | 20/25 i | 20/25 j | 20/25 | ||
| OS | 20/20 | 20/50 | 20/20 | 20/25 i | 20/25 j | 20/40 | ||
| Comfort | Good | Fair | Good | Good | Good | Good | ||
| Lid margin complications k | Moderate | Severe | − | Mild | Mild | Mild | ||
| Symblepharon | Minimal | Advanced | − | − | − | − | ||
| Dry eyes l | Moderate | Moderate | − | − | Minimal | Minimal | ||
| Corneal complications | Mild corneal pannus | Corneal perforation | − | − | − | Trace nummular haze |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


