Purpose
Patchy atrophy is a type of chorioretinal atrophy located outside of the fovea in eyes with myopic retinopathy. Bruch membrane defects have previously been described to occur in highly myopic eyes in foveal chorioretinal atrophy associated with choroidal neovascularization (CNV). We examined whether Bruch membrane defects can be found also in patchy atrophy.
Design
Retrospective observational case series.
Methods
The study included all patients who were consecutively examined for high axial myopia (axial length ≥26.5 mm) and patchy atrophy in the study period from September to November 2015. The patients underwent a comprehensive ophthalmologic examination including swept-source optical coherence tomography (OCT) of the macula. Main outcome measures were macular Bruch membrane defects.
Results
Out of 22 eyes (17 patients) with patchy atrophy, 21 eyes (96%) showed macular Bruch membrane defects, which were characterized by a lack of Bruch membrane, retinal pigment epithelium (RPE), photoreceptors, and choriocapillaris. At the edges of the macular Bruch membrane defects, the ends of the Bruch membrane were folded and the RPE was upturned. The inner retina overlying the area of the Bruch membrane defect was markedly thinned.
Conclusions
Macular Bruch membrane defects belong to the hallmarks of a type of myopic chorioretinal atrophy not associated with CNV (ie, patchy atrophy). Considering that Bruch membrane defects were also observed in myopic CNV-related foveal atrophy, macular Bruch membrane defect might be a common finding in fundus lesions related to pathologic myopia.
Pathologic myopia is a major cause of irreversible vision loss and blindness worldwide, and in particular in East Asia. Myopic maculopathy as a hallmark of pathologic myopia develops owing to an excessive axial elongation and can be associated with the formation of posterior staphyloma. In a recently published international photographic classification and grading system for myopic maculopathy, 5 categories of myopic maculopathy have been defined, ranging from category 0, with no macular lesions; to category 1, with tessellated fundus; category 2, characterized by diffuse chorioretinal atrophy; category 3, with patchy chorioretinal atrophy; and category 4, showing macular atrophy.
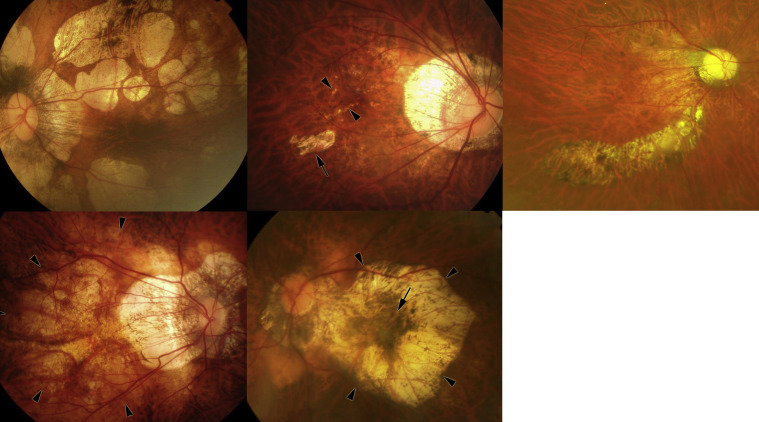
Patchy chorioretinal atrophy has been defined as a grayish-white, well-defined, sharply demarcated atrophy ( Figure 1 ). Patchy atrophy has been subclassified into 3 types : patchy atrophy that develops from lacquer cracks, patchy atrophy developing within the area of an advanced diffuse chorioretinal atrophy, and patchy atrophy that can be seen along the border of a posterior staphyloma. Patchy chorioretinal atrophy can be distinguished from ill-defined yellowish “diffuse atrophy” ( Figure 1 ), which is characterized by choroidal thinning in optical coherence tomographic (OCT) images. Angiographic findings showing a choroidal filling defect suggested that patchy atrophy was associated with a complete closure or nonperfusion or lack of the choriocapillaris. Swept-source OCT additionally revealed that patchy atrophy showed a loss of most layers of choroid, of the retinal pigment epithelium (RPE), and of the outer retina layers. This finding differed from observation made in diffuse atrophy in which the RPE and outer retina and some parts of the choroid were usually preserved.
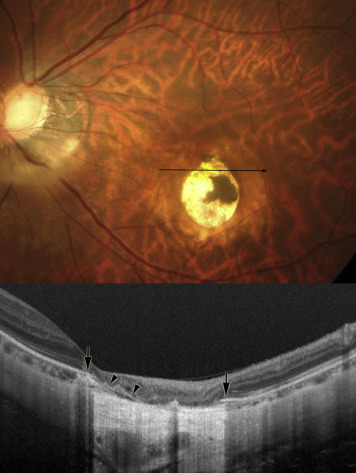
It has generally been assumed that the Bruch membrane in highly myopic eyes remained intact in the area of myopic macular chorioretinal atrophy despite the loss of RPE, choriocapillaris, and photoreceptors. In a recent histologic report, macular Bruch membrane defects also were detected in highly myopic globes. Using swept-source OCT, Ohno-Matsui and associates found that Bruch membrane was discontinuous in the region with macular atrophy associated with chorioretinal neovascularization (CNV), so that macular Bruch membrane defects were a feature of myopic CNV-related macular atrophy that could be detected by clinical imaging.
Among various lesions of myopic maculopathy, the fundus appearance of patchy atrophy is similar to macular atrophy developing around the myopic CNV (myopic CNV–related macular atrophy; Figure 1 ). CNV-related macular atrophy is the late complication of myopic CNV and determines the long-term visual outcome. The main difference between CNV-related macular atrophy and patchy atrophy is that the former develops in and just around the fovea and enlarges concentrically around the fovea, whereas patchy atrophy develops outside the foveal region and even when it enlarges it does not commonly involve the foveal center.
Because the fundus appearance of patchy atrophy is similar to CNV-related macular atrophy although the location relative to the fovea and the presence of CNV is different between these two lesions, we postulated that Bruch membrane defects may be a common hallmark of both these atrophic lesions in pathologic myopia. We therefore conducted the present study to examine frequency and associations of macular Bruch membrane defects in regions with patchy atrophy.
Methods
The retrospective, observational case series included 22 eyes of 17 patients with pathologic myopia including patchy chorioretinal atrophy. The study was in agreement with the tenets of the Declaration of Helsinki and was approved by the Ethics Committee of Tokyo Medical and Dental University. The patients had consecutively been examined for high myopia (defined as an axial length ≥26.5 mm) and patchy atrophy in the High Myopia Clinic of the Tokyo Medical and Dental University in the study period between September 18 and November 20, 2015. Out of 3 subtypes of patchy atrophy, the type of patchy atrophy along staphyloma borders was not found in any patient during the study period, so that the 2 remaining subtypes—lacquer crack–associated patchy atrophy and diffuse chorioretinal atrophy–associated patchy atrophy—were examined in this study. The mean age of the study participants was 62.2 ± 9.6 years (range, 46–80 years), and mean axial length was 30.3 ± 2.0 mm (range, 26.5–33.7 mm). All patients underwent a comprehensive ophthalmologic examination including refractometry, measurement of axial length (IOL Master; Carl Zeiss Meditec Co, Jena, Germany), color fundus photography, and assessment of fundus autofluorescence (TRC-50DX; Topcon, Tokyo, Japan). The size of patchy atrophy was determined on the images of the fundus autofluorescence. We measured the area of patchy atrophy by using the software of the imaging system (PDT/MPS software; Topcon, Tokyo, Japan). We took into account axial length for correction of the fundus image magnification by the optic media of the eye.
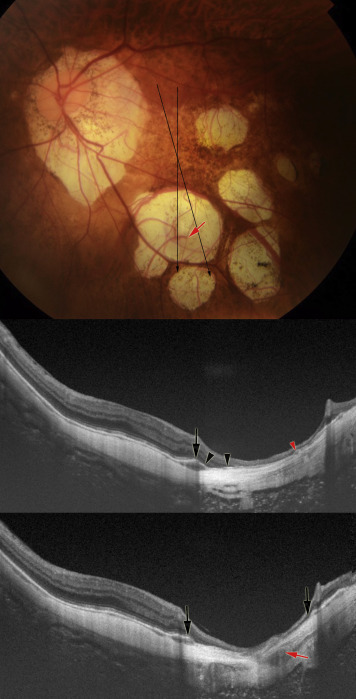
The status of the Bruch membrane was examined using swept-source optical coherence tomography (OCT) (DRI-OCT; Topcon). As scanning protocols, 9 mm or 6 mm radial scan sections centered on the fovea were performed. Each section was composed of 96 single images that were registered and averaged by the software to create a single image. Each image consisted of 1024 A-scans. The Bruch membrane in the area of patchy atrophy was investigated by a single examiner (KOM).
For the statistical analysis, we applied the Mann-Whitney U test to compare the eyes with different types of patchy atrophy with respect to age, axial length, number of patchy atrophy, and size of patchy atrophy. A P value less than .05 was considered significant.
Results
The average number of patchy atrophic regions was 3.7 ± 2.1 per eye (range, 1–8). The average total area of patchy atrophy was 13.5 ± 10.5 mm 2 per eye (range, 2.2–34.0 mm 2 ). The mean area of each one of the lesions of patchy atrophy per eye was 5.3 ± 5.8 mm 2 (range, 0.6–12.3 mm 2 ).
Patchy atrophy associated with lacquer cracks was found in 8 eyes (7 patients) and patchy atrophy associated with advanced diffuse chorioretinal atrophy was detected in 14 eyes (12 patients). Two patients had lacquer crack–associated patchy atrophy in 1 eye and advanced diffuse chorioretinal atrophy–associated patchy atrophy in the other eye. The eyes with lacquer crack–associated patchy atrophy and the eyes with advanced diffuse chorioretinal atrophy–associated patchy atrophy did not differ significantly in age (59.3 ± 8.6 years vs 63.9 ± 10.0 years, P = .40), axial length (29.4 ± 1.5 mm vs 30.8 ± 2.1 mm, P = .17), number of patchy atrophy regions (4.3 ± 2.1 vs 3.4 ± 2.1, P = .40), total area of all lesions of patchy atrophy per eye (10.0 ± 9.3 mm 2 vs 15.5 ± 10.8 mm 2 , P = .48), and the average size of each one of the lesions of patchy atrophy per eye (3.5 ± 4.3 mm 2 vs 6.4 ± 6.5 mm 2 , P = .24).
In 1 of the 22 eyes with patchy atrophy (4.5%), the Bruch membrane was intact and continuous over the entire area of patchy atrophy as examined in the 12 radial OCT scans. This eye had 7 small lesions of lacquer crack–associated patchy atrophy with a total area of 5.3 mm 2 , with a mean size of each lesion of 0.8 mm 2 . In the remaining 21 eyes (95.5%) in which the size of the patchy atrophic lesions was larger, the Bruch membrane showed discontinuities in the area of patchy atrophy ( Figures 2–4 ).


Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


