Purpose
To report long-term clinical outcomes of deep anterior lamellar keratoplasty (DALK) in patients with keratoconus.
Design
Retrospective noncomparative interventional study.
Methods
setting : Single center. patients : Total of 158 eyes/150 consecutive patients with keratoconus with postoperative follow-up time equal to or greater than 4 years. intervention : DALK. main outcome measure(s) : Uncorrected and corrected distance visual acuity (UDVA, CDVA), mean refractive spherical equivalent (MRSE), keratometry, endothelial cell density (ECD).
Results
Mean postoperative follow-up was 76.9 ± 23.2 (range 48–120) months. Preoperative UDVA was 20/400 (1.5 ± 0.4 logMAR), CDVA 20/50 (0.7 ± 0.2 logMAR), MRSE −11.1 ± 5.6 diopters (D), mean keratometry 60.7 ± 6.1 D, topographic astigmatism 4.7 ± 2.6 D. At last postoperative follow-up visit, UDVA improved to 20/50 (0.5 ± 0.3 logMAR), CDVA to 20/25 (0.09 ± 0.1 logMAR), MRSE to −2.6 ± 3.5 D, mean keratometry to 44.4 ± 2.2 D, and topographic astigmatism to 2.9 ± 1.3 D. Postoperative ECD did not vary from preoperative values being 2070.5 ± 367.5 cell/mm 2 and 2198 ± 373 cell/mm 2 , respectively, with 70% of eyes (111/158) showing ECD ≥2000 cells/mm 2 . Eighteen eyes (11.3 %) developed stromal or epithelial rejection, 3 (1.8%) required regrafting. Eyes with successful big bubble showed greater CDVA than those that required manual dissection at follow-up equal to or less than 5 years but comparable results in the longer term.
Conclusions
DALK provides stable long-term visual and refractive outcomes. Risk of graft rejection, postoperative complication, and late ECD decay is reduced when compared to standard penetrating keratoplasty.
Despite the introduction of novel conservative treatments for keratoconus (KC), namely corneal cross-linking and intrastromal ring segments, corneal transplantation remains the only therapeutic choice for patients with advanced keratoconus, intolerant to or inadequately corrected with contact lenses.
Deep anterior lamellar keratoplasty (DALK) is an increasingly popular alternative to penetrating keratoplasty (PK) for patients with corneal diseases that spare the Descemet membrane (DM) and endothelium, such as KC. Data from the Eye Bank Association of America showed that KC is currently the leading indication for anterior lamellar keratoplasty in the United States (43.4% of the cases). Similarly, reports from the United Kingdom, Singapore, and Australia eye banks suggest that the number of DALK procedures has increased gradually during the last 10 years. Different DALK techniques have been described, based essentially on either manual dissection/delamination of the corneal stroma or separation of the DM from the stroma by means of intrastromal injection of fluid, viscoelastic, or air. The “big-bubble” technique applies a forceful injection of air into the deep stroma to obtain cleavage separation of the descemetic/predescemetic plane (Dua layer) from the overlying stroma, with formation of a large air bubble between these 2 layers. DALK provides several advantages over standard PK. Primarily it allows preservation of the host endothelium, abolishing the risk of endothelial immunologic rejection and potentially reducing the issue of late endothelial cell decay. The surgical procedure is carried out “closed-sky,” with decreased postoperative intraocular inflammation and risk of intraocular infection. Consequently, topical steroids after DALK can be discontinued earlier than after PK, reducing cataract development, glaucoma, secondary infections, and delayed wound healing. DALK is also thought to produce a tectonically stronger cornea, less prone to spontaneous or post-traumatic wound dehiscence. Previous studies showed that DALK graft survival is longer than PK at short- and medium-term follow-up, with a more favorable cost-benefit ratio in comparison to PK. On the other hand, DALK, presenting a steep learning curve, requires longer operating time than PK, limiting the overall diffusion of this procedure. As DALK has only recently been introduced in many tertiary centers, reports of long-term postoperative outcomes of this procedure are scarce, often including different DALK techniques and patients with various corneal diseases grouped together. Comparison to PK outcomes and evaluation of the potential advantages provided by DALK in the long run are, therefore, at present mostly speculative.
This study focused on the long-term (≥4 years postoperative) outcomes of DALK in a cohort of patients affected by KC.
Methods
This retrospective noncomparative interventional study adhered to the tenets of the Declaration of Helsinki and was approved by the Institutional Review Board, Reggio Emilia Hospital, Emilia-Romagna, Italy. An informed consent for the surgery was obtained from all participants.
One hundred and fifty consecutive patients (158 eyes) with KC, who underwent DALK between March 2003 and March 2008, were included in this retrospective single-center study. Diagnosis of KC was made on the basis of clinical slit-lamp findings (stromal thinning, ectasia, Fleisher ring, Vogt striae, epithelial and subepithelial scarring), keratometry, and corneal topography. All patients were contact lens intolerant and/or had corrected distance visual acuity ≤20/40 prior to DALK. Inclusion criteria were preoperative clinical and topographic diagnosis of KC and postoperative follow-up time equal to or more than 4 years. Exclusion criteria were any coexisting ocular disease that could affect visual acuity and other previous intraocular surgery.
All surgeries were performed by a single surgeon (L.F.). The “big-bubble” technique described by Anwar and Teichmann was used for lamellar dissection. Surgical procedures were performed under general anesthesia using a standard technique previously described. The postoperative medication regimen included tobramycin 0.3% and dexamethasone phosphate 0.1% 4 times a day for a month, tapered over a period of 6 months, and artificial tears instilled 6 times a day for 3 months. Continuous suture adjustment or selective suture removal was performed at the slit lamp or in the operating room in eyes with more than 6 diopters of postoperative topographic astigmatism. Sutures were otherwise retained for at least 18 months postoperatively. All sutures were removed within 36 months of surgery. Preoperative and postoperative eye examination included uncorrected (UDVA; logMAR) and corrected (CDVA; logMAR) distance visual acuity, manifest refraction (diopters [D]), slit-lamp examination, applanation tonometry, fundus examination, corneal topography (Pentacam HR; Oculus, Wetzlar, Germany), ultrasonic central corneal pachymetry (Altair; Optikon 2000, Rome, Italy), and specular microscopy (Topcon SP2000p; Topcon Corp, Tokyo, Japan). Postoperative complications (cataract, glaucoma, etc) and additional surgical interventions were recorded. Data from patients in whom a successful big bubble formation was accomplished were grouped (Group 1) and compared to patients in whom layer-by-layer manual deep stromal dissection was carried out (Group 2).
Data are expressed as mean ± standard deviation. Statistical analysis was carried out by STATA software, version 11.0 (STATA/IC 11, College Station, Texas, USA). The Student t test for paired or unpaired data was applied to assess difference between groups. A value of P < .05 was considered statistically significant.
Results
One hundred and fifty patients (158 eyes) were included in this retrospective analysis, 98 were male (65.3%) and 52 female (34.7%). The mean age at the time of surgery was 33.6 ± 8.8 years (range 19–60). Twenty-two of 150 patients had atopic dermatitis. No other comorbidities were present. In all patients preoperative UDVA was lower than 20/100 (0.5 logMAR) and CDVA was equal to or lower than 20/40 (0.3 logMAR). Preoperative mean refractive spherical equivalent (MRSE) was −11.1 ± 5.6 D. Preoperative mean keratometry was 60.7 ± 6.1 D, with 48% of eyes (76/158) of patients showing a mean 3 mm central keratometry greater than 60.5 D. Preoperative mean topographic astigmatism was 4.7 ± 2.6 D. Average thinnest point measured on the pachymetric map was 345.8 ± 74.4 μm. Specular microscopy and endothelial cell counts were obtained before surgery in only 73 patients (48.6%) because of advanced keratoconus. The mean endothelial cell density (ECD) was 2198 ± 373 cells/mm 2 preoperatively.
Mean postoperative follow-up was 76.9 ± 23.2 (range 48–120) months. Cleavage separation of the stroma from DM with air injection was achieved in 109 eyes (68.9%). In the remaining cases a layer-by-layer manual deep stromal dissection was performed. Intraoperative microperforations occurred in 16 eyes (10.1%). Postoperative formation of a double anterior chamber was observed in 4 eyes (2.5%), which resolved spontaneously in 2 eyes and required intracameral air injection in the others. In 36 eyes (22.7%), a topographic astigmatism of 6 D or more was observed 4–6 weeks after surgery, which prompted suture adjustment or selective suture removal. All sutures were removed after an average time of 18.1 ± 5.1 months (range 6–30 months). Eighteen eyes (11.3%) developed stromal or epithelial immunologic rejections that occurred before 2 years from surgery; 4 patients had atopic dermatitis. Medical treatment was sufficient to control rejection in all cases, except in 3 eyes (1.8%) who underwent regrafting owing to the development of irreversible graft opacities and interface neovascularization in 1 case. Repeat graft procedures were carried out by removing and replacing the affected donor stromal disc, while preserving the healthy host Descemet membrane. Eighteen eyes (11.3%) required astigmatic keratotomy or laser refractive surgery for the correction of postkeratoplasty astigmatism greater than 6 D.
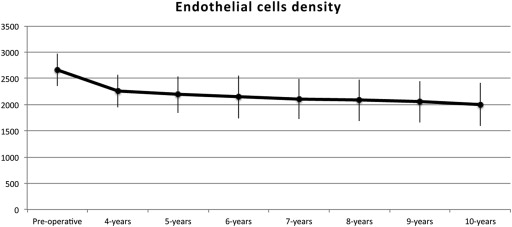
Clinical outcomes are reported in Table 1 and Table 2 . At last examination mean UDVA was 0.5 ± 0.3 logMAR (20/50), while mean CDVA was 0.09 ± 0.1 logMAR (20/25). UDVA went from <20/100 in all eyes preoperatively to ≥20/100 in 108 eyes (68%) postoperatively. CDVA went from <20/50 in all eyes preoperatively to ≥20/50 in 145 eyes (91%) postoperatively, with 62 eyes (39%) reaching ≥20/20 vision. Twenty-six percent of patients (42/158) were able to reapply contact lenses. Postoperative MRSE improved to −2.6 ± 3.5 D, and postoperative average topographic astigmatism to 2.9 ±1.3 D. Final average keratometry was 44.4 ± 2.2 D. A clear graft-host interface was observed from the early postoperative days in 155 eyes (98%). Final mean central pachymetry was 526.2 ± 44.6 μm and ECD was 2070.5 ± 367.5 cells/mm 2 . Seventy percent of patients (111/158) showed an ECD ≥2000 cells/mm 2 at last visit. Figure 1 shows the variation of ECD in 20 eyes over a 10-year follow-up time. None of the patients developed cataract or ocular hypertension during the duration of this study.
| UDVA (logMAR) | CDVA (logMAR) | MRSE (Diopters) | Topographic Astigmatism (Diopters) | Average Keratometry (Diopters) | CCT (μm) | ECD (Cells/mm 2 ) | |
|---|---|---|---|---|---|---|---|
| Preoperative values | 1.30 ± 1.30 | 0.7 ± 1.0 | −10.27 ± 7.14 | 4.7 ± 2.64 | 60.72 ± 6.1 | 347.3 ± 76.0 | 2198 ± 373 |
| Last follow-up | 0.40 ± 0.60 | 0.10 ± 0.70 | −2.5 ± 3.5 | 2.84 ± 1.52 | 44.5 ± 2.3 | 525.7 ± 45.5 | 2070.5 ± 367.5 |
| P | <.01 | <.01 | <.01 | <.01 | <.01 | <.01 | .28 |
| Follow-up | Number of Eyes (%) | UDVA (logMAR) | CDVA (logMAR) | MRSE (Diopters) | CYL (Diopters) | Keratometry (Diopters) | CCT (μm) | ECD (Cells/mm 2 ) |
|---|---|---|---|---|---|---|---|---|
| Baseline | 158 (100%) | 1.30 ± 1.30 | 0.7 ± 1.0 | −10.27 ± 7.14 | 4.7 ± 2.64 | 60.72 ± 6.1 | 347.3 ± 76.02 | 2198 ± 373 |
| 4 years | 103 (65.1%) | 0.40 ± 0.54 | 0.18 ± 0.8 | −3.78 ± 3.55 | 2.7 ± 1.36 | 45.22 ± 1.98 | 523.2 ± 49.1 | 2340 ± 371 |
| 5 years | 66 (41.7%) | 0.40 ± 0.54 | 0.1 ± 0.76 | −2.8 ± 3.49 | 2.5 ± 1.20 | 45.3 ± 2.30 | 522.1 ± 42.4 | 2372 ± 378 |
| 6 years | 28 (17.7%) | 0.54 ± 0.90 | 0.1 ± 0.80 | −2.33 ± 3.26 | 2.9 ± 1.5 | 44.4 ± 1.77 | 531.5 ± 31.9 | 2254 ± 312 |
| 7 years | 14 (8.8%) | 0.40 ± 0.60 | 0.1 ± 0.70 | −2.12 ± 4.96 | 2.7 ± 1.59 | 41.7 ± 2.71 | 497 ± 55.9 | 2228 ± 86 |
| 8 years | 28 (17.7%) | 0.48 ± 0.60 | 0.10 ± 0.70 | −2.68 ± 4.01 | 2.9 ± 1.19 | 43.8 ± 2.30 | 529.7 ± 45.4 | 2203 ± 98 |
| 9 years | 19 (12.0%) | 0.30 ± 0.90 | 0.20 ± 0.80 | −1.06 ± 2.82 | 3.5 ± 1.54 | 44.9 ± 1.92 | 525.8 ± 32.1 | 2107 ± 96 |
| 10 years | 20 (12.6%) | 0.40 ± 0.60 | 0.10 ± 1.18 | −1.43 ± 1.17 | 1.4 ± 3.44 | 45.6 ± 3.28 | 560 ± 51.5 | 2150 ± 164 |
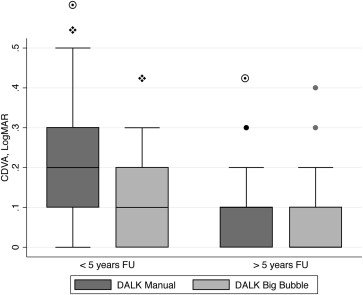
As shown in Table 3 , no difference in final clinical outcomes was seen in eyes with big-bubble cleavage separation and Dua layer exposure (Group 1) compared to eyes that required manual stromal dissection (Group 2). A significant difference in CDVA between groups was seen comparing patients with follow-up equal to or less than 5 years ( P < .001) ( Figure 2 ). This difference was not significant compared to patients with longer follow-up ( P = .191). A time-dependent improvement in CDVA was noticed in Group 2, comparing patients with follow-up equal to or less than 5 years to those with longer postoperative time ( P = .003). The same analysis repeated for patients in Group 1 was not significant ( P = .907).
| Outcomes | Big Bubble | Manual | P Value |
|---|---|---|---|
| UDVA (logMAR) | 0.40 ± 0.70 | 0.40 ± 0.54 | >.999 |
| CDVA (logMAR) | 0.10 ± 0.70 | 0.10 ± 0.76 | >.999 |
| MRSE (diopters) | −2.26 ± 3.36 | −2.29 ± 3.9 | .9 |
| Mean keratometry (diopters) | 44.29 ± 2.23 | 44.83 ± 2.43 | .09 |
| Topographic astigmatism (diopters) | 2.86 ± 1.50 | 2.82 ±1.58 | .8 |
| CCT (μm) | 523.5 ± 44.28 | 528.6 ± 47.71 | .5 |
| ECD (cell/mm 2 ) | 2116.4 ± 308.45 | 2009.4 ± 432.14 | .07 |

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


