Purpose
To assess the safety and efficacy of dendritic cell vaccination in metastatic uveal melanoma.
Design
Interventional case series.
Methods
We analyzed 14 patients with metastatic uveal melanoma treated with dendritic cell vaccination. Patients with metastatic uveal melanoma received at least 3 vaccinations with autologous dendritic cells, professional antigen-presenting cells loaded with melanoma antigens gp100 and tyrosinase. The main outcome measures were safety, immunologic response, and overall survival.
Results
Tumor-specific immune responses were induced with dendritic cell vaccination in 4 (29%) of 14 patients. Dendritic cell-vaccinated patients showed a median overall survival with metastatic disease of 19.2 months, relatively long compared with that reported in the literature. No severe treatment-related toxicities (common toxicity criteria grade 3 or 4) were observed.
Conclusions
Dendritic cell vaccination is feasible and safe in metastatic uveal melanoma. Dendritic cell-based immunotherapy is potent to enhance the host’s antitumor immunity against uveal melanoma in approximately one third of patients. Compared with other prospective studies with similar inclusion criteria, dendritic cell vaccination may be associated with longer than average overall survival in patients with metastatic uveal melanoma.
Uveal melanoma is the most common primary intraocular malignancy in adults with an annual incidence of 4 to 10 per 1 million in the white population, although representing only 3% of all melanoma cases. Uveal melanoma arises from melanocytes residing in the uveal tract of the eye that have migrated out of the neural crest. Approximately 90% of uveal melanoma arise in the choroid, 6% in the ciliary body, and 4% in the iris. In up to 50% of the patients with primary uveal melanoma, metastatic disease ultimately develops, which occurs by hematogenous dissemination; the median time from initial diagnosis of uveal melanoma until detection of metastatic disease ranges from 2 to 5 years. Currently, there is no effective systemic treatment for metastasis to improve overall survival, resulting inevitably in tumor-related death when metastasis occurs, with the minor exceptions of a small proportion of patients who have successful curative surgery of metastasis or patients with spontaneous regression of metastatic disease.
Prognostic factors to identify patients with primary uveal melanoma at risk for metastatic disease include clinical (tumor location, tumor size, age), histologic (cell type, vascular pattern, mitotic count, extraocular extension), and genetic (chromosomal aberrations, expression profiling, gene mutations) parameters, partially included in the American Joint Committee on Cancer classification of uveal melanoma. Over the past few decades, treatment of the primary tumor has changed drastically because several forms of radiotherapy have replaced enucleation as the preferred treatment of the primary tumor, depending on size and location of the tumor and patient preference. However, despite the improvements in diagnosis and the development of eye-conserving treatments, none of these treatment methods prevents the development of metastases. The relative 5-year survival rates have not increased over the past decades, fluctuating at approximately 70% to 80%.
Only up to 2% of patients have detectable metastasis when their primary uveal melanoma is diagnosed ; most patients have a long disease-free interval before metastasis becomes clinically evident. In uveal melanoma, liver metastases are seen most frequently (90% to 95%), and it is often the sole site of metastatic disease. Other common sites of metastases, mostly in the presence of liver metastases, are lungs (25%), bone (15%), skin (10%), and lymph nodes (10%); in contrast to cutaneous melanoma, uveal melanoma infrequently metastasizes to the brain. After metastasis develops, overall survival mainly is independent of previously mentioned prognostic factors if one is identifying patients with primary uveal melanoma at risk for metastatic disease. Presence of symptomatic disease, metastatic extensiveness, and metastatic-free interval may correlate with survival time. Nevertheless, median survival is short, typically less than 9 months, with a poor 1-year survival rate (10% to 40%). The small group of patients in whom metastases are confined to extrahepatic locations have a significantly longer median survival, approximately 19 to 28 months.
Several locoregional treatment options can be considered in selected patients with metastasis confined to the liver, including surgery, isolated hepatic perfusion, or radiofrequency ablation. Although prolonged survival has been reported after surgical resection of liver metastasis, this may be the result in part of selection bias. To date, treatment options for metastatic uveal melanoma are limited, and compelling evidence that any systemic therapy, including chemotherapy, improves overall survival is lacking. Disease stabilization is described in several patients receiving ipilimumab, which recently has shown survival benefit in metastatic cutaneous melanoma patients. However, data are based on a limited number of patients. Therefore, effective therapies resulting in meaningful clinical benefit are required urgently, and immunotherapy may be a promising treatment method.
Immune-based therapies aim to induce antitumor immunity. Despite uveal melanoma developing in the immune-privileged environment of the eye, immune cells have been found within uveal melanoma, including dendritic cells and T cells. Dendritic cells are antigen-presenting cells with the unique capacity to activate naïve antigen-specific T cells, and hence are suitable for inducing immunologic antitumor responses ( Figure 1 ). Dendritic cell-based immunotherapy has shown promising results in cutaneous melanoma patients. Although uveal and cutaneous melanoma are different biologically, cutaneous melanoma and uveal melanoma share many antigenic features, including tumor antigens, providing a rationale for the application of dendritic cell-based therapies in uveal melanoma. The tumor antigens used in our dendritic cell vaccination studies for metastatic melanoma patients, gp100 and tyrosinase, are both expressed in most human uveal melanoma tumor cells, and thus constitute an appropriate target for immunotherapy in uveal melanoma.
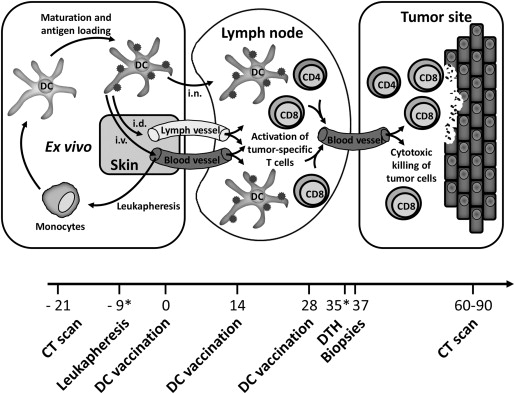
Our research group has performed several prospective dendritic cell vaccination studies in patients with melanoma, of which most consisted of patients with cutaneous melanoma. We here present data on the subset of metastatic uveal melanoma patients who were enrolled in these studies.
Methods
The studies were approved by the Dutch Centrale Commissie Mensgebonden Onderzoek (Central Committee on Research Involving Human Subjects), and written informed consent to participate in research was obtained from all patients. The trials were registered at ClinicalTrials.gov (identifiers NCT00940004 , NCT01690377 , NCT01530698 , and NCT00243529 ).
Patient Characteristics
We analyzed a cohort of 14 patients with metastatic uveal melanoma who were enrolled in our prospective dendritic cell vaccination studies between October 2002 and May 2011. Patients were required to have at least 1 measurable target lesion. Additional inclusion criteria were melanoma expressing the melanoma-associated antigens gp100 (compulsory) and tyrosinase (noncompulsory), HLA-A*02:01 phenotype (protocols I, III, IV, V, and VI), known HLA-DRB*01:04 status (protocol IV), and World Health Organization performance status 0 or 1. Patients with serious concomitant disease or a history of second malignancy were excluded.
Treatment Schedule
All patients were vaccinated with autologous dendritic cells loaded with tumor-associated antigens of gp100 and tyrosinase according to a schedule of 3 biweekly vaccinations. One to 2 weeks after the last vaccination, a skin test was performed; see the treatment schedule in Figure 1 . In absence of disease progression, patients received a maximum of 2 maintenance cycles at 6-month intervals. Variations in protocols included the type of dendritic cells, route of administration, method of antigen loading, and pretreatment with anti-CD25 antibody, described in the Supplemental Table (available at AJO.com ). Stable disease was defined according to Response Evaluation Criteria in Solid Tumors with a minimal duration of 4 months. Adverse events were graded according to the National Cancer Institute Common Terminology Criteria for Adverse Events version 3.0.
Dendritic Cell Vaccine
Monocytes, enriched from leukapheresis products, were cultured in the presence of interleukin-4 (500 U/mL) granulocyte-macrophage colony-stimulating factor (800 U/mL; both Cellgenix, Freiburg, Germany) and control antigen keyhole limpet hemocyanin (10 μg/mL; Calbiochem, Darmstadt, Germany). Dendritic cells were matured with autologous monocyte-conditioned medium (30%, vol/vol) supplemented with prostaglandin E2 (10 μg/mL; Pharmacia & Upjohn, Puurs, Belgium) and 10 ng/mL tumor necrosis factor-α (Cellgenix) for 48 hours as described previously. All administered dendritic cell vaccines met the release criteria previously described. In the Supplemental Methods (available at AJO.com ), a detailed description on dendritic cell culture is provided.
Analyses of Immunologic Responses
To assess the immune response against control and tumor peptides generated in vaccinated patients, peripheral blood was drawn and delayed-type hypersensitivity challenges were performed. In the Supplemental Methods (available at AJO.com ), a detailed description of immunomonitoring tests is provided.
Fluorescent In Situ Hybridization Analysis
Fresh tumor material from enucleated eyes containing uveal melanoma were cultured routinely for karyotyping and were used directly for fluorescent in situ hybridization (FISH) analysis of chromosome 3 as previously described. Dual-color FISH was performed with the following probes: Pα3.5 (centromere 3), RP11-64F6 (3q25), and RP11-1059N10 (5q12). Chromosome 5 is rarely involved in genetic changes in uveal melanoma and was used as a control for aneuploidy, truncation, and cutting artifacts. The concentration for centromeric probe was 5 ng per slide, whereas for the bacterial artificial chromosome probes, 50 to 75 ng per slide was used. After hybridization and washing, the slides were counterstained with 4′, 6-diamidino-2-phenylindole and mounted in antifade solution (Dabco-Vectashield 1:1; Vector Laboratories, Burlingame, California, USA). Signals were counted in 300 interphase nuclei. Scoring for deletion (>20% of the nuclei with 1 signal) or amplification (>10% of the nuclei with 3 signals or more) was adapted from the available literature. Using FISH analysis, we subdivided the variation in chromosome 3 into the following categories: monosomy 3 (loss of 1 copy), disomy 3 (normal copy numbers [2 copies]), and hyperdiploidy (gain of 1 copy).
Statistical Analysis
Overall survival was calculated from the date of leukapheresis to death. Patients who did not die during the follow-up period were censored at the time of last follow-up. The Kaplan-Meier method was used to obtain estimates of median survival times and to generate survival curves. IBM SPSS Statistics (SPSS version 20.0) software (SPSS, Inc., Chicago, Illinois, USA) was used for statistical analysis.
Results
Patient Characteristics
Fourteen uveal melanoma patients with metastatic disease were enrolled in dendritic cell vaccination studies. Patient characteristics are shown in Table 1 . The mean age was 52 years; 9 patients were men and 5 were women. One patient had metastases confined to extrahepatic locations. All other patients had liver metastases, of which the liver was the sole site of metastasis in 5 patients. Six patients had received prior treatment for their metastatic disease, mostly consisting of surgery or dacarbazine (chemotherapy). Lactate dehydrogenase, (if elevated, a negative prognostic factor in metastatic uveal melanoma), was elevated at baseline in 3 of 14 patients. Median time between diagnosis of the primary tumor and metastatic disease was 20.4 months. Four patients had synchronous metastasis at presentation ( Table 2 ).
| Patient | Gender | Age (y) | LTD (mm) | Chromosome 3 | Treatment of Primary | No. of Metastases | Site of Metastatic Disease | LDH (U/L) | LDM (mm) | Prior Treatment for Metastases |
|---|---|---|---|---|---|---|---|---|---|---|
| I-C14 | M | 54 | 14 | Disomy | E | 1 | Liver | 312 | 33 | No |
| III-B7 | M | 54 | 13 | Monosomy | E | 2 | Liver | 277 | 16 | No |
| III-B8 | M | 40 | 13 | Monosomy | E | 3 | Liver | 1289 | Diffuse | No |
| IV-A4 | M | 51 | 13 | Monosomy | E | >5 | Liver | 417 | 41 | No |
| IV-A10 | F | 54 | n.a. | Monosomy | No | >5 | Liver, lung | 432 | 17 | No |
| IV-B11 | M | 65 | n.a. | n.t. | RT | >10 | Liver, lymph node, lung | 640 | 182 | C1 |
| IV-D3 | F | 42 | n.a. | Monosomy | RT/L | >10 | Liver, lymph node, lung, pancreas | 344 | 19 | S, C1 |
| V-A3 | M | 52 | n.a. | n.t. | Ru | >5 | Liver, bone | 517 | 56 | RFA, P/T |
| VI-B6 | M | 53 | 15 | Disomy | E, RT | >5 | Liver, lung, bone, soft tissue | 434 | 40 | S |
| VI-DE3 | M | 62 | 23 | Hyperdiploidy | E, RT | >5 | Liver, lung | 360 | 17 | No |
| VI-DE4 | M | 35 | 16 | n.t. | E | >5 | Liver, lymph node, soft tissue | 320 | 47 | No |
| VIII-A1 | F | 49 | 12 | Monosomy | E | >5 | Liver | 424 | 16 | No |
| VIII-A4 | F | 46 | 12 | Disomy | Ru/Th | >5 | Skin, large intestine, soft tissue | 440 | 25 | S |
| VIII-DE2 | F | 70 | 12 | Monosomy | E | >10 | Liver, lymph node, lung, adrenal gland | 447 | 53 | C1 |
| Patient | Time to Metastases (mo) | No. of Vaccinations | PFS (mo) | Later Treatment for Metastases | OS from Primary Diagnosis (mo) | OS from Apheresis (mo) | Best Clinical Response |
|---|---|---|---|---|---|---|---|
| I-C14 | 44 | 3 | 6 | S (×2) | 130 a | 84 a | SD |
| III-B7 | 0 | 3 | 2 | C1 | 10 | 7 | PD |
| III-B8 | 8 | 3 | 2 | C1 | 11 | 3 | PD |
| IV-A4 | 10 | 3 | 5 | C1 | 32 | 19 | SD |
| IV-A10 | 2 | 6 | 14 | No | 55 | 52 | SD |
| IV-B11 | 103 | 3 | 5 | No | 145 | 9 | SD |
| IV-D3 | 1 | 3 | 6 | C2, S | 53 | 26 | SD |
| V-A3 | 27 | 3 | 2 | Ipi | 55 | 4 | PD |
| VI-B6 | 68 | 3 | 4 | RT | 85 | 13 | SD |
| VI-DE3 | 32 | 3 | 5 | C1, Ipi, AKTi | 71 | 38 | SD |
| VI-DE4 | 90 | 3 | 5 | C1, C2/WKi, P/Ifos, Ipi | 120 | 27 | SD |
| VIII-A1 | 0 | 3 | 2 | C1 | 12 | 10 | PD |
| VIII-A4 | 97 | 9 | 28 | PKCi | 136 a | 32 a | SD |
| VIII-DE2 | 20 | 9 | 22 | RT | 52 | 26 | SD |
All tumors were confirmed histopathologically as uveal melanoma. Histopathologic examination results of the primary tumor were available in 9 patients who were treated with enucleation. Based on cell type, 8 primary tumors were classified as epithelioid or mixed and 1 as spindle. The median largest tumor diameter of the primary tumor was 13 mm. One tumor was located in the ciliary body (VI-DE3) and 11 were located in the choroid (2 unknown primary location in the ciliary body or choroid). In 12 of 14 patients, metastatic disease was confirmed by histopathologic analysis. All uveal melanoma tumor cells tested, 6 primary tumors and 8 metastases, showed positive results for gp100 expression. Additionally, 11 of 12 uveal melanoma tumor cells tested also expressed tyrosinase.
Cytogenetic Results
Uveal melanomas of 11 patients were analyzed for chromosomal changes by using cytogenetic and FISH analyses and were classified for gain and loss in chromosome 3 ( Table 1 ). Analyses were performed on primary tumors in 5 patients, on metastases in 4 patients, and on both in 2 patients. Not enough tumor material was available to analyze the remaining 3 patients. Clonal chromosomal abnormalities were present in 8 of 11 tumors tested. Seven tumors showed monosomy 3, 3 patients showed disomy, and 1 patient had a tumor showing hyperdiploidy of chromosome 3. No discrepancies were seen in the patients where both the primary tumor and a metastasis were tested.
Immunologic Responses
To test the capacity of the patients in this study to generate an immune response with vaccination, dendritic cells were loaded with a control antigen. Peripheral blood mononuclear cells collected after each vaccination were analyzed for the presence of control antigen-specific T cells. Almost all patients (12 of 14) showed a cellular response to control antigen in the first cycle. In 7 of 13 patients tested, control antigen-specific IgG antibodies were detected after vaccination ( Table 3 ). These results indicate that the vaccine induced de novo immune responses. To determine the presence of tumor antigen-specific CD4+ and CD8+ T cells, tetramer analyses for 1 tyrosinase and 2 gp100 epitopes were performed after 3 vaccinations. In peripheral blood, tetramer-positive CD4+ T cells, indicative of tumor recognition by T-helper cells, could be seen in 1 of 2 HLA-DRB*01:04-positive patients tested, which were also detectable in the blood before dendritic cell vaccination.
| Patient | Control Antigen-Specific T-Cell Response (Blood) | Control Antigen-Specific Antibody Response (Blood) | Tumor Antigen-Specific CD8+ T-Cell Response (Skin Test) | Tumor Antigen-Specific CD8+ T-Cell Response (Blood) | Tumor Antigen-Specific CD4+ T-Cell Response (Blood) |
|---|---|---|---|---|---|
| I-C14 | + | − | − | − | n.t. |
| III-B7 | + | − | − | − | n.t. |
| III-B8 | + | n.t. | n.t. | − | n.t. |
| IV-A4 | + | + | + | − | + a |
| IV-A10 | + | + | − | − | − |
| IV-B11 | + | + | + | − | n.a. b |
| IV-D3 | + | + | − | − | n.a. b |
| V-A3 | + | − | − | − | n.t. |
| VI-B6 | + | − | − | − | n.t. |
| VI-DE3 | + | + | − | + | n.t. |
| VI-DE4 | − | + | − | + | n.t. |
| VIII-A1 | + | − | n.a. | n.a. c | n.t. |
| VIII-A4 | + | + | n.a. | n.a. c | n.t. |
| VIII-DE2 | + | − | − | − | n.t. |
a Tumor antigen-specific CD4+ T-cell response also was detectable in the blood before dendritic cell vaccination.
b HLA-DRB*01:04–negative patients.
In 3 patients (protocol VI), blood mononuclear cells were restimulated in vitro over 2 weeks with the 3 antigenic peptides, before screening all microcultures for the presence of CD8+ tetramer-positive cells. This procedure allowed estimation of the frequencies of tumor antigen-specific CD8+ T cells in blood that proliferate in vitro in response to tumor antigen. Two patients showed a significant increase (≥5-fold) of the frequency of gp100-specific CD8+ T cells.
Antigen-specific CD8+ T cells were detected in delayed-type hypersensitivity skin tests in 2 of 11 HLA-A*02:01-positive patients ( Figure 2 ; Table 3 ). In patient IV-B11, functionality of the antigen-specific CD8+ T cells was tested, and they proved to be fully functional and to produce high levels of interleukin-2 and interferon-γ on antigen-specific stimulation.




