Purpose
To evaluate the risk factors, recurrence rates, retreatments, and long-term patient outcomes following proton beam therapy for uveal melanoma.
Design
Retrospective interventional case series.
Methods
All patients treated with primary proton beam therapy for uveal melanoma at the oncology service at Charité-Berlin and Helmholtz-Zentrum-Berlin between May 1998 and December 2008 were reviewed for local recurrence. Of 982 patients, 982 eyes matched the inclusion criteria. The data were obtained from electronic health records, operative reports, discharge letters, and radiation planning. Comparisons of fundus photographs and ultrasound measurements were performed to assess the growth pattern of the tumor and to determine the success of retreatment, in the case that a globe-retaining therapy was undertaken.
Results
Of 982 patients, 35 patients (3.6%) developed local recurrence. The median follow-up was 60.7 months (6.0–170.4 months). Local control rate was 96.4% and the overall eye retention rate was 95.0% in this cohort. Local recurrence was correlated with a higher risk for metastasis and reduced survival. Largest tumor diameter was identified as the sole statistically significant risk factor for local recurrence ( P = .00001). All globe-retaining retreatment approaches for local recurrence, including proton beam therapy, brachytherapy, and transpupillary thermotherapy used for recurrences at the tumor margins, showed good local tumor control and similar metastasis-free survivals.
Conclusions
This study showed that each globe-retaining retreatment approach can result in satisfying local tumor control. In case of early detection of local recurrence, preservation of the globe can be warranted. Therefore, regularly performed follow-ups should be ensured.
The 3 objectives of uveal melanoma treatment are saving the patient’s life, preservation of the globe, and preservation of its function, in descending order. With the decision for globe-retaining treatment, local tumor control becomes greatly important. Today’s most accepted globe-retaining therapies are brachytherapy with episcleral plaques (eg, 125 I or 106 Ru), teletherapy with heavy charged particles (proton beam and helium ion beam therapy), and CyberKnife radiotherapy. Transpupillary thermotherapy can be used for small choroidal melanoma. All approaches bear more or less risk of local recurrence. Proton beam therapy showed local tumor recurrence rates ranging from 2% to 8.4% after 5 years. Risk factors for failure of local control after proton beam therapy include a large tumor diameter, small tumor volume, iris root involvement, and safety margins inferior to 1 mm. Recurrence may take the form of marginal, diffuse, distant, or extrascleral extension. Currently, enucleation is still the most used treatment for recurrent melanoma, although irradiation (eg, proton beam therapy) is a suitable option for retreatment. As there is no standard for retreatment, the treatment approach depends on the extent and the location of the recurrence and remains a case-by-case decision. This study therefore concentrates on classifying the risk factors for local recurrences after proton beam therapy and the local and systemic outcome of different retreatment approaches.
Methods
The study was approved by the institutional review board of the Charité-Universitätsmedizin Berlin, Berlin, Germany, and was in accordance with the tenets of the Declaration of Helsinki. All patients with initial diagnosis of choroidal or ciliary body melanoma that were treated with primary proton beam therapy at the Ocular Oncology Service at Charité Berlin and the Helmholtz Center Berlin between May 1998 and December 2008 were reviewed for local recurrence. In the framework of our treatment planning, the indication for using primary proton beam therapy is a centrally located tumor close to sensitive structures or a peripheral tumor beyond the arcades exceeding 6 mm.
The dose for proton beam therapy in our study was 60 cobalt gray equivalent in total (cobalt gray equivalent: 1 cobalt gray equivalent = 1.1 Gy, taking a radiobiological effectiveness of 1.1 into account), given in 4 fractions of 15 cobalt gray equivalent on 4 sequential days. Prior to irradiation, 2.5-mm-diameter tantalum clips were sutured to the sclera to demarcate the margins of the tumor. Three-dimensional modeling of the tumor (clinical target volume) was calculated based on localization measurements via ultrasound, funduscopy, and computed tomography scans. Treatment planning and dose calculation were performed with the treatment planning systems EYEPLAN version 1.2 and (since 2006) OCTOPUS version 4.4 (German Cancer Research Center, DKFZ and Hahn-Meitner-Institute, Berlin). The “clinical target volume” is extended by a standard 2.5 mm margin to compensate for patient motion, set-up errors, range uncertainties, and lateral dose build-up. During treatment, the head of the patient was immobilized by a combination of an individually molded face mask and a bite block to ensure the correct position. The patient had to focus on an external light source. In case of movement, the therapy could immediately be interrupted.
Color fundus photographs taken throughout the examinations (before proton beam therapy [montage-photograph] until last follow-up) were compared to assess the growth pattern of the tumor in combination with ultrasound measurements. Comparisons of fundus photographs and ultrasound measurements were also performed to determine the success of retreatment, in the case that a globe-retaining therapy was undertaken.
The local recurrences were divided into 4 groups, depending on their growth pattern: marginal, central, distant, and diffuse. Local recurrence was defined as marginal if the tumor showed only a marginal growth with an increased diameter, central if the tumor increased only in thickness (after an interval of 12 months, to exclude the expected swelling during the first months after proton beam treatment), diffuse if diameter and height showed tumor growth, and distant if local recurrence occurred distant from the primary tumor.
Statistics
For statistical analysis, patients were divided into 2 groups, a local control group and a local recurrence after proton beam therapy group. Statistical analysis was performed using SPSS 20.0 (SPSS, Inc, Chicago, Illinois, USA). A Mann-Whitney U test, Fisher exact test, χ 2 test, and factorial analysis of variance (ANOVA) were used to assess statistical differences between groups. Interrelationships between variables were assessed by calculating correlation coefficients according to Spearman/Pearson. Stepwise models for multiple regression analysis with odds ratios were used to evaluate different independent risk factors of the event of local recurrence (1 dependent and multiple independent variables), while models for multivariate regression analysis were used to evaluate risk factors in the development of local recurrences (multiple independent and dependent variables). Kaplan-Meier survival analysis was used for examining the distribution of time-to-event variables. Visual acuity is described as logMAR. In relation to the MARAN protocol, light perception (LP) was added as 2.1 logMAR, hand motion (HM) as 2.0 logMAR, and counting fingers (CF) as 1.9 logMAR.
Results
We included 982 patients, of whom 35 patients (3.6%) developed local recurrence after proton beam therapy. The median follow-up was 60.7 months (6.0–170.4 months). The local tumor control rate was 96.4%. The Kaplan-Meier estimate for local recurrence was 2% (± 1%) at 1 year, 3% (± 1%) at 3 years, 4% (± 1%) at 5 years, and 6% (± 1%) at 10 years. After proton beam therapy, the median time local recurrence occurred was 17.5 months (1.3–88.7 months).
Time of overall survival differed significantly ( P < .05) between 49.4 months (11.5–152.1 months) in the local recurrence group and 61.7 months (0.1–170.4 months) in the local control group. The Kaplan-Meier estimate for survival was 99% (± 1%) at 1 year, 90% (± 1%) at 3 years, 80% (± 1%) at 5 years, and 60% (± 1%) at 10 years in the local recurrence group compared to 99 % (± 1%) at 1 year, 95% (± 1%) at 3 years, 89% (± 1%) at 5 years, and 80% (± 1%) at 10 years in the local control group ( Figures 1 and 2 ).
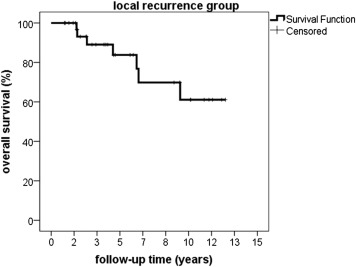
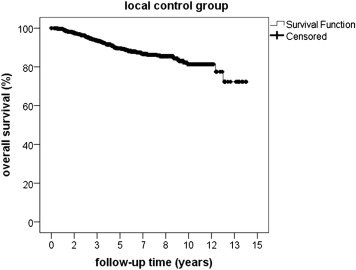
Metastasis occurred in 10 patients of the local recurrence group (10/35, 28.6%) after a median time of 53.9 months (12.3–147.9 months) after proton beam therapy and 14 months (2.6–128.6 months) after the development of local recurrence—compared to 145 patients (145/947, 15.3%) of the local control group who developed metastasis after a median interval of 31.6 months (0.4–154.1 months) after proton beam therapy, P < .03.
The risk of developing metastasis was statistically significantly increased in the local recurrence group, P < .03. Median time of metastasis-free survival was 49.4 months (11.5–148.8 months) and 60.7 months (6.0–170.4 months) in the local recurrence group and the local control group, respectively. The Kaplan-Meier estimate for metastasis-free survival calculated from the day after finishing proton beam therapy was 97% (± 1%) at 1 year, 84% (± 1%) at 3 years, 76% (± 1%) at 5 years, and 50% (± 1%) at 10 years in the local recurrence group; and 99% (± 1%) at 1 year, 90% (± 1%) at 3 years, 85% (± 1%) at 5 years, and 77% (± 1%) at 10 years in the local control group. Statistical analysis showed a significant difference between both groups, indicating a prolonged metastasis-free survival in patients without local recurrence, P = .037 ( Figures 3 and 4 ).
Evaluation of Recurrence Risk Factors
Initial tumor volume
Median initial tumor volume was 362 mm 3 (31–2011 mm 3 ) in the local recurrence group vs 276 mm 3 (1.0–3063.0 mm 3 ) in the local control group. No statistical significance was found between groups, nor was there any significant correlation with local recurrence.
Initial tumor diameter
Median initial largest tumor diameters ranged between 12.5 mm (5.1–17.6 mm) and 12.0 mm (3.0–26.1 mm) in the local recurrence group and the local control group, respectively. Statistical univariate analysis revealed a strong correlation between tumor diameter and occurrence of local recurrence, P < .0001. Further evaluation of patients with large largest tumor diameter, defined as largest tumor diameter >10 mm, revealed that especially tumors with flat edges are at high risk for local recurrence owing to underestimation of clinical target volume.
Initial tumor thickness
Median initial tumor thickness was 5.1 (1.8–14.8 mm) in the local recurrence group and 4.4 mm (1.3–15.3 mm) in the local control group. No statistical significance was found between these groups, nor was there any significant correlation with local recurrence.
Distance to sensitive structures
Of 982 patients, 320 patients (32.6%) presented with a tumor directly adjacent to the optic disc or fovea, 320 (32.6%) had tumors at a distance to a sensitive structure of 0.1–2.5 mm, 176 (17.9%) at a distance of 2.6–5 mm, 119 (12.1%) at a distance of 5.1–10 mm, and 47 (4.8%) at a distance of 10.1–21 mm. All tumors that developed local recurrence were located at the posterior pole (31 patients) except those with ciliary body involvement (4 patients). The median distance was 0.5 mm (0–6.0 mm) to the optic disc and 0.4 mm (0–7.5 mm) to the fovea. In the local control group median distance to optic disc and fovea were 1.5 mm (0–21.0 mm) and 0.9 mm (0–18.9 mm), respectively. There was a statistically significant difference ( P < .005) for distances to sensitive structures between both groups but no correlation related to the development of local recurrence ( Table ).
| Anatomic Features | Initial Tumor Volume (mm 3 ) | Initial Tumor Diameter (mm) | Initial Tumor Thickness (mm) | Distance to Optic Disc (mm) | Distance to Fovea (mm) |
|---|---|---|---|---|---|
| Local recurrence group (n = 35 patients) | |||||
| Median | 362 | 12.5 | 5.1 | 0.5 | 0.6 |
| Min | 32 | 5.1 | 1.8 | 0 | 0 |
| Max | 2011 | 17.6 | 14.8 | 6.0 | 7.5 |
| Local control group (n = 947 patients) | |||||
| Median | 276 | 12.0 | 4.4 | 1.5 | 1.9 |
| Min | 1 | 3.0 | 1.3 | 0 | 0 |
| Max | 3063 | 26.1 | 15.3 | 21.0 | 18.9 |
2007 Tumor-Node-Metastasis (TNM) classification
Of the 35 patients with local recurrence 5 (14.3%) were classified as T1, 14 (40%) as T2, 11 (31.4%) as T3, and 5 (14.3%) as T4. Of the 947 patients with good tumor control, 260 (27.4%) were classified as T1, 340 (35.9%) as T2, 278 (29.4%) as T3, and 69 (7.3%) as T4. There was no statistical significance found between groups, and no significant correlation with local recurrence.
Safety margin
The clinical target volume is expanded by a 1.0 mm safety margin in all directions. The resulting volume is called planning target volume. According to ICRU Reports 50 and 62, this margin should compensate for uncertainties in patient set-up, small movements of the treated eye during irradiation, and all other geometric uncertainties. In the direction of the proton beam a margin of 1.5 mm distal and proximal to the planning target volume is used to cover range uncertainties of the proton beam to cover uncertainties of the density of the patient’s eye. To compensate for the lateral dose fall-off, a lateral margin of 1.5 mm between the patient collimator and the planning target volume is needed. All these margins combined result, by coincidence, in a 2.5 mm margin around the clinical target volume. The margin of the clinical target volume was ≥2.5 mm in all but 5 patients (5/35, 14.3%) in the local recurrence group vs 9 patients (9/947, 1.0%) in the local control group, P < .005. Of the 5 patients who developed local recurrence, 4 patients were treated with a reduced margin by 0.5 mm and 1 patient by 1.0 mm. In the local control group the safety margin was reduced by 0.5 mm in 2 patients, by 1.0 mm in 5 patients, and by 1.5 mm in 2 patients. Statistical univariate analysis showed a statistically significant correlation between the reduction of safety margin and the development of local recurrence, P < .05. Three patients 3/35, (8.5%) were treated with an enlarged safety margin in the local recurrence group and 24 patients (24/947, 2.5%) in the local control group for unknown tumor spread or unknown infiltration of the optic nerve. There was no statistical significance found between groups and no significant correlation with local recurrence.
Ciliary body involvement
Of all 982 patients, 80 patients presented with ciliary body involvement. Four of those patients (5%) developed local recurrence and were classified as being of the local recurrence group. Conversely, 76 patients with ciliary body involvement (95%) showed good local tumor control. No statistical significance was found between these groups, nor was there any significant correlation with local recurrence.
Multivariate Regression Analysis
Statistical multivariate regression analysis of the recurrence risk factors identified the initial largest tumor diameter as the sole statistically significant recurrence risk factor, P = .00001. All other features such as safety margin, close distances or involvement of papilla and fovea, initial tumor thickness, initial tumor volume, or ciliary body involvement were statistically unrelated to recurrence.
Treatment Approaches for Local Recurrence
Treatment of the 35 eyes with local recurrence comprised enucleation in 19 eyes (54%), brachytherapy in 8 eyes (23%), transpupillary thermotherapy in 7 eyes (20%), and proton beam therapy in 1 eye (3%). The treatment approach was based on an individual case-by-case decision. Transpupillary thermotherapy was only used for local recurrence at the tumor margins. No re-recurrence was detected. There was no statistically significant difference in metastasis-free and overall survival dependent on globe-retaining retreatment approaches but a reduced metastasis-free survival in patients treated with enucleation ( Figure 5 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


