Objective
To investigate the prevalence of polypoidal choroidal vasculopathy (PCV) in patients with myopic choroidal neovascularization (CNV) using indocyanine green angiography (ICGA).
Design
Retrospective cross-sectional study.
Methods
A total of 297 eyes (255 patients) who presented with treatment-naive myopic CNV between January 2005 and December 2011 at Yonsei University Medical Center in Seoul, South Korea, were reviewed. Fluorescein angiography (FA) images obtained from the patients were analyzed to detect CNV presence and classify disease type. ICGA images were reviewed to detect polypoidal lesions. The main outcome measure was the prevalence of polypoidal lesions in patients with myopic CNV.
Results
All 297 eyes with myopic CNV were type 2 CNV, and mean age at diagnosis was 47.32 ± 14.69 years. The mean refractive error was −11.95 ± 5.88 diopters, and the mean axial length was 29.39 ± 2.02 mm in the affected eyes. Among the myopic CNV eyes, 141 eyes (118 patients) were older than 50 years of age (mean 60.48 ± 7.34 years). No eyes with myopic CNV showed polypoidal lesions on ICGA at initial presentation. After treatments for myopic CNV, 243 eyes (206 patients) completed at least 12 months of follow-up, and 86 eyes (35.4%) showed at least one recurrence of CNV during follow-up. The follow-up imaging studies, FA, and ICGA, showed no polypoidal lesions associated with recurred myopic CNV.
Conclusions
ICGA analysis demonstrated no polypoidal component in myopic eyes with CNV.
Polypoidal choroidal vasculopathy (PCV) is a peculiar disease entity characterized by abnormal branching microvascular networks and terminal polypoidal protrusions as shown on indocyanine green angiography (ICGA). PCV shares common features with typical age-related macular degeneration (AMD), including both risk factors and genetic predispositions. Optical coherence tomography (OCT) has localized polypoidal lesions with abnormal branching vascular networks to within the Bruch membrane, under the retinal pigment epithelium (RPE). This suggests that PCV is a variant of type 1 choroidal neovascularization (CNV), in which the neovascular complex is confined to the sub-RPE space. However, PCV has its own clinical characteristics, which are distinguished from typical exudative AMD. Several studies have documented choroidal hyperpermeability and choroidal vascular congestion in PCV eyes. By ICGA, multifocal choroidal hyperfluorescence and dilated choroidal vessels are identified in PCV eyes. The choroidal thickness of PCV eyes is also significantly thicker than in typical exudative AMD eyes and in normal controls.
Pathologic myopia is prevalent in Asian countries. In pathologic myopia, various degenerative changes, including chorioretinal atrophy, lacquer crack, staphyloma formation, and diminished choroidal vasculature with altered blood flow, occur with eyeball elongation. Myopic CNV is a vision-threatening complication that affects the macula in eyes with pathologic myopia.
Both PCV and pathologic myopia are prevalent in Asian countries. In addition, the aqueous levels of vascular endothelial growth factor (VEGF) in eyes with active PCV and myopic CNV were similar and were significantly lower than those of AMD. It is known that PCV can develop on the borders of inferior staphylomas associated with tilted disc syndrome. PCV also can appear as type 2 classic CNV on fluorescein angiography (FA) when fibrinoid subretinal material presents over polypoidal lesions, and additional ICGA evaluation is therefore necessary for differential diagnosis. However, in our clinical setting, we generally do not detect polypoidal lesions in myopic CNV eyes. Furthermore, it is not known whether PCV can develop in eyes with pathologic myopia other than tilted disc syndrome with inferior staphyloma. If polypoidal lesions develop in pathologic myopia eyes, it will be detected as presumed myopic CNV. Thus, in this study, we evaluated the prevalence of PCV in eyes with myopic CNV by ICGA.
Methods
Subjects
We retrospectively reviewed the medical records of 297 eyes of 255 patients with treatment-naive myopic CNV who presented at Yonsei University Medical Center between January 2005 and December 2011. Informed consent was obtained from all eligible participants at the time of angiographic examination, regarding the purpose of the examinations and possible complications due to angiographic dyes. This retrospective study was approved by the Institutional Review Board of Yonsei University Medical Center (approval number 4-2012-0171) and adhered to the tenets of the Declaration of Helsinki.
Inclusion criteria were (1) patients initially diagnosed with myopic CNV; and (2) absence of concurrent ocular diseases in the enrolled eyes that might compromise FA and ICGA images in the study eye, such as retinal vein occlusion and diabetic retinopathy. Exclusion criteria were (1) any history of prior treatment for CNV, such as laser photocoagulation, submacular surgery, or photodynamic therapy; and (2) the absence of FA and ICGA data.
Definitions for this study were pathologic myopia as (1) a refractive error exceeding −6.5 diopters or an axial length exceeding 26.5 mm; and (2) fundus changes indicative of pathologic myopia, including lacquer cracks, atrophic patches, or chorioretinal atrophy. Myopic CNV was defined as (1) eyes exhibiting the characteristic features of pathologic myopia described above; (2) relatively localized, focal late-phase leakage on FA; and (3) absence of features suggesting AMD such as drusen. PCV was defined as the presence of abnormal branching vascular networks with terminating polypoidal lesions on ICGA. Recurrence of myopic CNV was defined as evidence of leakage from a previously closed CNV on FA images.
Type 1 occult CNV was defined as poorly defined forms of CNV on FA; fibrovascular pigment epithelial detachment (stippled hyperfluorescence within 1 to 2 minutes after fluorescein injection with persistent staining or leakage in the overlying subretinal space by 10 minutes), or late-phase leakage from an undetermined source (late speckled hyperfluorescence lacking well-demarcated borders, with pooling of the eye in the overlying subretinal space). The location of type 1 occult CNV was confined to the subRPE space by OCT images. Type 2 classic CNV was defined as well-demarcated hyperfluorescence with progressive pooling of dye leakage in the overlying subretina space in the late phase. The neovascular membrane of type 2 classic CNV proliferates above the RPE in the subneurosensory space as shown by OCT.
Treatments for myopic CNV included photodynamic therapy (PDT) and anti-VEGF therapy. During the earlier period of the study, patients were treated with PDT. With the introduction of anti-VEGF agents, both PDT and anti-VEGF therapy were used for myopic CNV, depending on the physician’s discretion. After late 2007, anti-VEGF therapy has been the mainstay of treatment for both treatment-naive and recurrent myopic CNV.
Examination
All patients underwent complete ophthalmologic evaluation, including color fundus photography, FA, and ICGA at baseline. OCT images were obtained using a time domain OCT device (Stratus Carl Zeiss Meditec, Dublin, CA, USA) for patients before 2009, and a spectral domain OCT device (SD OCT; Spectralis; Heidelberg Engineering, Heidelberg, Germany) from 2009 onward. FA and ICGA data were collected for all enrolled patients during a baseline evaluation using a Heidelberg retina angiograph (HRA-II; Heidelberg Engineering, Dossenheim, Germany). All ICGA images were reviewed by 2 independent examiners (H.M.K., H.J.K.) to determine the presence of polypoidal lesions, and good agreements were made. The greatest linear dimension of myopic CNV was measured and calculated by the early phase FA image at 1 minute, using embedded software programs in the HRA system.
Patient characteristics retrieved from their medical charts included age at initial diagnosis, gender, refractive errors, and axial length at the time of diagnosing myopic CNV. Refractive error in all patients was measured by an auto-refractometer without cycloplegia. The spherical equivalent of refractive error in diopters was used in the statistical analyses. The greatest linear dimension of myopic CNV was also retrieved from image analysis.
Statistical analysis was performed using SPSS 18.0 for Windows (SPSS, Chicago, IL, USA).
Results
Myopic Choroidal Neovascularization at Initial Presentation
Among 297 eyes (255 patients) with myopic CNV, 232 eyes (192 patients, 75.3%) were female. Mean refractive errors and axial lengths were −11.95 ± 5.88D (range −39.00D to −5.25D) and 29.39 ± 2.02 mm (range 26.7 mm to 34.4 mm), respectively. Mean age at diagnosis was 47.32 ± 14.69 years, and 141 eyes (47.5%; 118 patients) were from patients aged over 50 years (mean age 60.48 ± 7.34 years).
Angiographic Imaging Studies at Initial Presentation
All myopic CNV eyes were type 2 CNV, and the mean greatest linear dimension of the lesions was 1794.25 ± 1157.14 μm. ICGA analysis showed no polypoidal component associated with CNV in any of the eyes. Representative eyes are shown in Figure 1 and Figure 2 .
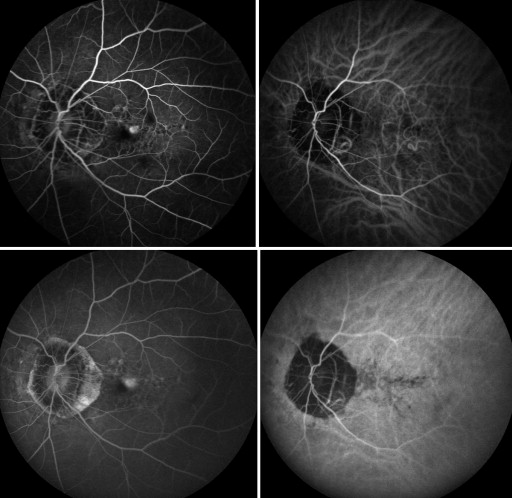
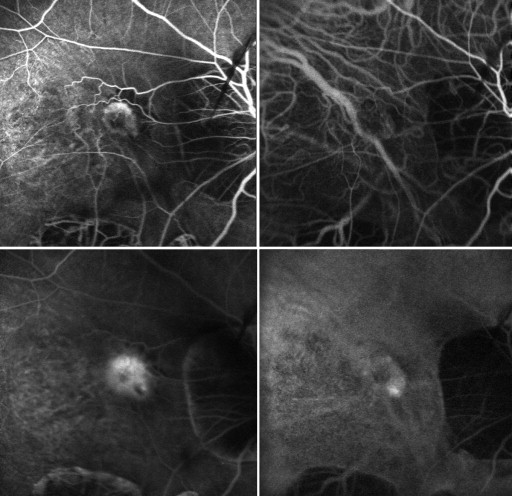
Recurrences
Among 297 eyes, 243 eyes (81.8%) completed at least 1 year of follow-up after initial treatment. Mean follow-up duration was 58.67 ± 38.42 months. In 86 eyes (35.4%) we saw at least 1 recurrence, with the mean number of recurrences being 1.86 ± 0.67 times. The mean interval between first remission and first recurrence was 18.74 ± 17.33 months. ICGA imaging studies at recurrence showed no polypoidal lesions associated with recurred myopic CNV.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


