Purpose
To identify predictive factors for visual outcome and need for retreatment after treating myopic choroidal neovascularization (CNV) with ranibizumab.
Design
A prospective interventional case series.
Methods
Sixty-seven eyes of 67 patients with myopic CNV were treated with 3 intravitreal ranibizumab injections given monthly. Best-corrected visual acuity (BCVA) and optical coherence tomography–determined central macular thickness (CMT) were recorded monthly during follow-up. Fluorescein angiography changes and the number of injections needed were also assessed.
Results
Mean follow-up was 15.9 months. Mean BCVA improved by 7.8 letters after the first injection, 12.5 letters after 3 injections, and 12 letters by end follow-up. In 53 eyes (79.1%), BCVA improved; 40.3% gained more than 15 letters. No differences were detected in visual outcome between treatment-naïve and previously treated patients. Myopic CNV area and greatest linear dimension had diminished at the study end. The mean reduction in CMT was 93.6 μm. The mean number of injections given was 4.2. A total of 53.7% of eyes received only 3 injections. Through regression analysis, baseline BCVA ( P = .006) and myopic CNV location ( P = .026) were significantly correlated with BCVA at the end of follow-up. Myopic CNV location ( P = .023) and prior treatment ( P = .047) were significantly linked to the number of injections given. No major complications arose.
Conclusion
An initial treatment regimen of 3 monthly ranibizumab injections seems effective and safe to treat myopic CNV. Baseline BCVA and myopic CNV location emerged as predictive factors for visual outcome. A need for retreatment was associated with myopic CNV location and prior treatment.
Choroidal neovascularization (CNV) is one of the most important causes of vision loss and myopic CNV is the leading cause of vision impairment in patients younger than 50 years. The only approved treatment for myopic CNV available today is photodynamic therapy (PDT) with verteporfin (Visudyne; Novartis Ag, Basel, Switzerland). Although the results of the VIP study group demonstrated the stability of visual acuity in eyes treated with PDT as compared with placebo at 12 months of follow-up, this stability did not persist in the 24-month follow-up examination.
CNV in age-related macular degeneration (AMD) has also been traditionally treated by vascular occlusion using PDT. Recently, however, this strategy has been replaced with the use of anti-angiogenic drugs that bind vascular endothelial growth factor (VEGF). Ranibizumab (Lucentis; Novartis, Basel, Switzerland) is a humanized antibody fragment against VEGF-A that has proved effective in the treatment of AMD. Ranibizumab offers the advantage over the anti-VEGF drug bevacizumab (Avastin; Genentech, South San Francisco, California, USA) that its smaller molecular weight determines its greater retinal penetration and facilitates its intravitreal use. However, despite this new treatment modality for AMD having been also used to treat myopic CNV, there is still no general consensus as to the best treatment regimen for myopic patients. Although fewer studies have examined the effects of ranibizumab than bevacizumab in myopia treatment, both these agents have resulted in excellent visual outcomes without associated complications. Some of the authors of these studies recommend a single intravitreal injection followed by treatment as needed (pro re nata; PRN). In contrast, despite its limitations, the PRONTO study indicates good anatomic and visual outcomes of an initial loading dose of ranibizumab to treat CNV in AMD and indicates that these effects persist for 2 years without significant complications.
This prospective study evaluates the treatment of myopic CNV with ranibizumab by identifying possible predictive factors that could affect final visual acuity and the need for retreatment. The initial intravitreal dose selected was similar to that recommended for the treatment of CNV in exudative AMD.
Methods
This prospective, consecutive, nonrandomized pilot study was conducted on 67 eyes of 67 patients with subfoveal or non-subfoveal myopic CNV.
The inclusion criteria were minimum spherical equivalent (SE) of −6 diopters (D) with retinal signs of pathologic myopia, a best-corrected visual acuity (BCVA) of 24 letters or more, and active myopic CNV. The latter was defined as angiographic leakage and/or intraretinal edema and/or subretinal fluid determined by optical coherence tomography (OCT). We also included patients who had been treated a minimum of 3 months previously with PDT or 2 months previously with sodium pegaptanib (Macugen; EyeTech Pharmaceuticals/Pfizer Inc, New York, New York, USA).
The exclusion criteria were presence of active CNV secondary to a disease other than pathologic myopia, concomitant disease in the study eye that could compromise the patient’s vision or fixation, patients with previous thromboembolic episodes, allergy to fluorescein, and fertile women not using contraception.
A complete ophthalmic examination was initially performed, including BCVA determination using ETDRS charts (letters), anterior segment slit-lamp examination, intraocular pressure (IOP) measurement, detailed dilated fundus examination with special attention to the macular area and retinal periphery, retinography, digital fluorescein angiography (FA), and OCT Stratus (V4.01; Carl Zeiss Meditec, Inc, Dublin, California, USA) determinations of central macular thickness (CMT) and retinal architecture. OCT and FA were performed and the data interpreted by 2 retinal specialists in an unmasked fashion. When needed, the opinion of a third specialist was sought. CNV area and greatest linear dimension (GLD) were measured in the first angiogram phase. To detect angiographic fluorescein leakage, late-phase (6–8 minutes) and early-phase (1–2 minutes) images were compared.
Patients were scheduled for 3 loading doses of intravitreal ranibizumab at baseline and at 1 and 2 months. Patients were retreated with single ranibizumab injections in case of anatomic activity, defined as intraretinal edema or subretinal fluid detected in the OCT and/or myopic CNV leakage in the FA. Patients were also retreated if they underwent a visual loss of at least 5 letters, compared to the best BCVA obtained at any visit, along with the above anatomic findings. Retreatments were suspended in cases of cystoid macular edema showing no evidence of change after 3 consecutive intravitreal ranibizumab injections, as long as there was no deterioration in symptoms or BCVA.
After the onset of treatment, monthly follow-up examinations consisted of BCVA and IOP determination, retinography, and OCT. FA was performed at 3 and 12 months and at the end of follow-up. Additional FA was conducted if there was visual loss or worsening of patient symptoms without evidence of deterioration observed in the OCT, or if an increased CNV area was revealed by retinography. Progression of chorioretinal atrophy was also assessed at 12 months and at the end of follow-up. Ocular or systemic adverse effects were also recorded in each follow-up session.
Statistical Analysis
The Student paired t test or multivariate analysis of variance were used to compare continuous variables (expressed as means ± standard deviation [SD]). Categorical variables were compared using the χ 2 test or McNemar test for paired data in the intent-to-treat population with last observation carried forward. Possible factors predictive of BCVA at the end of follow-up were identified by linear regression and those predictive of a need for retreatment by logistic regression analysis. The factors included in these analyses were age (in years), gender (male/female), spherical equivalent (D), prior PDT and/or sodium pegaptanib treatment, myopic foveal atrophy (none, mild, moderate, severe), foveal pigment (none, mild, moderate, severe), fibrosis (none, mild, moderate, severe), myopic CNV GLD (μm) and area (mm 3 ), myopic CNV location (subfoveal/non-subfoveal), initial cystoid macular edema (μm), baseline BCVA (letters), and CMT (μm). The level of significance was set at P < .05. All statistical tests were performed using SPSS version 15.0 software (SPSS Inc., Chicago, Illinois, USA).
Results
The mean age of the 67 patients included in our study (17 men, 50 women) was 59 (± 12.8) years. The average SE (± SD) was −11.1 (± 3.3) D. Three of 67 eyes (4.5%) had undergone refractive surgery and 13 eyes (19.4%) cataract surgery. Forty-two of the eyes (62.7%) had received prior treatment. Forty eyes (59.7%) showed subfoveal myopic CNV. Angiographically, all lesions were predominantly classic. Mean follow-up was 15.9 months (range, 6–27 months).
Mean baseline BCVA (± SD) was 53.4 (± 12.4) letters. This starting BCVA rose to 61.2 (± 12.8) letters after the first intravitreal ranibizumab injection and to 65.9 (± 12.8) after the third injection or loading dose. Mean BCVA was 65.3 (± 13.5) letters at 12 months (n = 59), 68.3 (± 17.8) letters at 21 months (n = 17), and 78.6 (± 5.6) letters at 24 months (n = 9). At the end of follow-up (6–24 months), mean BCVA was 65.4 (± 16.1) letters. The gains in BCVA produced were 7.8 (± 9.8) letters after the first injection and 12.6 (± 11.6) letters after all 3 injections. Average changes in BCVA recorded at 12 months, at 24 months, and at the end of the follow-up were 12.4 (± 13) letters, 19.1 (± 9.1) letters, and 12 (± 14.6) letters, respectively. In each follow-up session, mean BCVA values recorded were significantly improved over baseline ( P < .05) except for values determined at 25, 26, and 27 months of follow-up. Figure 1 shows the changes in mean BCVA produced during treatment follow-up.
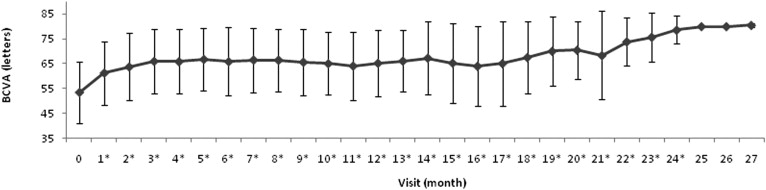
At the end of follow-up, 9 eyes (13.4%) had lost more than 1 letter, 4 of which lost more than 15 letters. Five eyes (7.5%) showed the same BCVA and 53 eyes (79.1%) showed a gain, 27 eyes (40.3%) improving by more than 15 letters.
When we compared visual outcomes between eyes subjected to prior treatment and those treated for the first time, mean BCVAs rose from 54.7 (± 12.7) letters at baseline to 70.4 (± 13.1) letters at the end of follow-up for the 25 naïve eyes and from 52.6 (± 12.3) letters to 62.5 (± 17.1) letters in the 42 eyes undergoing prior treatment, indicating no significant difference ( P = .08).
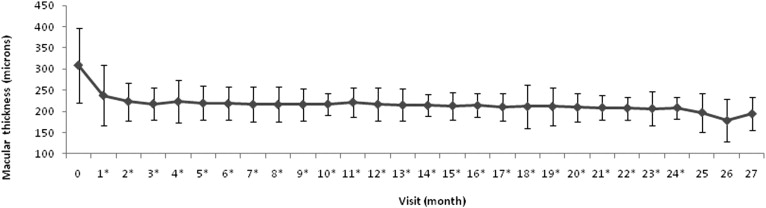
Figure 2 illustrates the mean changes in CMT as determined by OCT produced during follow-up. Thus, mean CMT decreased from 308.5 (± 87.4) μm at baseline to 217.9 (± 37.8) μm at 3 months, 216.8 (± 39.3) μm at 12 months, and 214.8 (± 35.7) μm at the end of follow-up. The average reductions in CMT recorded were 70.9 (± 55.9) μm after the first injection and 90.5 (± 77.9) μm after the loading dose. At 12 months, the CMT was 95.4 (± 74.8) μm thinner and the final average reduction was 93.7 (± 78.6) μm. The differences in CMT detected at each visit over baseline were significant ( P = .001), with the exception of those recorded in visits at 25, 26, and 27 months.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


