Purpose
To analyze retrospectively the efficacy of intravitreal ranibizumab injections for the management of choroidal neovascularization (CNV) in patients with angioid streaks.
Design
Nonrandomized, double-center, retrospective, interventional case series.
Methods
A consecutive series of patients affected with CNV associated with angioid streaks were treated with intravitreal ranibizumab injections (0.5 mg/0.05 mL). Best-corrected visual acuity, fundus photography results, optical coherence tomography (OCT) results, and fluorescein angiography results were examined before and after treatment. The primary end point was the percentage of eyes with stable or improved visual acuity at the end of follow-up. Secondary end points were the percentage of eyes with stable or decreased macular thickness on optical coherence tomography and the percentage of eyes with persistent leakage on fluorescein angiography at the last follow-up examination.
Results
Thirty-five eyes of 27 patients were treated with repeated intravitreal ranibizumab injections (mean, 5.7 injections; range, 2 to 14 injections) for a mean of 24.1 months (range, 6 to 37 months). At the end of follow-up, visual acuity was stabilized or improved in 30 (85.7%) of 35 eyes. Macular thickness had stabilized or decreased in 18 (51.5%) of 35 eyes. At the last follow-up examination, on fluorescein angiography, no further leakage was observed in 23 (65.7%) of 35 eyes.
Conclusions
In this large series of angioid streaks-associated CNV, ranibizumab injections allowed stabilization of visual acuity. Ranibizumab seems to be a safe therapeutic option in CNV associated with angioid streaks.
Angioid streaks are the clinical signs of extended disruptions of the Bruch membrane, which becomes calcified and fragile, allowing ingrowth of choroidal neovascularization (CNV) into the subretinal space. Angioid streaks are a major source of visual impairment, especially in middle-aged active patients. An estimated 72% to 86% of angioid streaks cases have CNV at presentation, which can develop as a result of Bruch membrane disruptions. The spontaneous evolution of angioid streaks-associated CNV is poor and frequently results in legal blindness, given that both eyes are affected in more than 70% of cases. Angioid streaks also can be associated with systemic diseases, such as pseudoxanthoma elasticum (PXE). Historically, angioid streaks-associated CNV has been treated with laser photocoagulation, photodynamic therapy with verteporfin, and rarely, transpupillary thermotherapy or surgical removal, all of which have poor results. Recently, certain studies have shown the benefits of intravitreal injections of anti-vascular endothelial growth factor (VEGF) in angioid streaks associated CNV in small series using bevacizumab. Herein we report a large retrospective series of patients affected with CNV associated with angioid streaks treated by ranibizumab, another anti-VEGF agent approved by the Food and Drug Administration for treatment of all types of CNV in patients with age-related macular degeneration.
Methods
All consecutive patients affected with subfoveal CNV associated with angioid streaks and treated with intravitreal ranibizumab at the Eye University of Creteil (Paris XII) or at the Ecole Militaire Retinal Centre between February 2007 and March 2010 were included in this retrospective study. Patients followed-up for fewer than 6 months were excluded from the analysis.
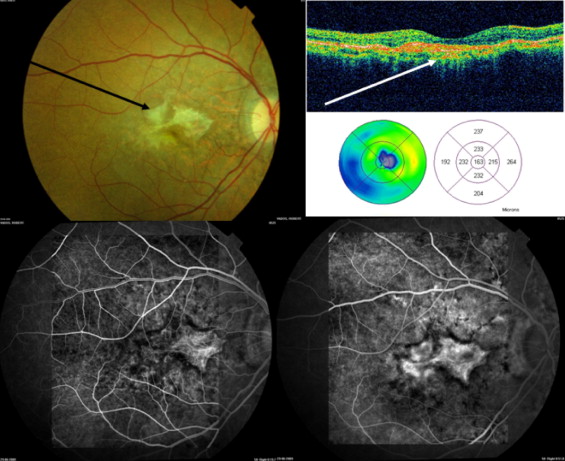
We recorded data about age at presentation, gender, medical history, ocular symptoms and history, duration of the disease (CNV), prior ocular treatment, interval between the last treatment and the first injection of ranibizumab, and association with PXE (biopsy proven). Best-corrected visual acuity (BCVA) was measured using Early Treatment Diabetic Retinopathy Study charts. For statistical analysis, visual acuities were converted to logarithm of the minimal angle of resolution charts. Patients were examined at the baseline visit, 1 month after the first injection, and then monthly with measurement of best-corrected visual acuity, fundus biomicroscopy, and optical coherence tomography (OCT). Fluorescein angiography (FA) was performed systematically at baseline and again if active CNV was suspected on clinical examination or on OCT ( Figure 1 ) . FA was performed systematically at the baseline visit, then after the third, sixth, ninth, and twelfth injections. FA was performed using a Topcon fundus camera with a TRC-50 IX system (Topcon, Tokyo, Japan). OCT (with mapping) was performed with a Stratus OCT3 version 4.0 (Carl Zeiss Meditec, Dublin, California, USA). Indocyanine green angiography was performed systematically at baseline and then again if doubts subsisted about the diagnosis of CNV on fluorescein angiography. Indocyanine green angiography was performed with the Heidelberg Retinal Analyzer (Heidelberg Engineering, Inc, Heidelberg, Germany). All macular thickness data were based on the central macular map obtained with OCT3. During the follow-up, all patients were examined by the same physician (G.M.). The decision to retreat the patient was taken if one or more of the following signs was present: decreased visual acuity (5 letters or more), recent macular hemorrhage from CNV, leakage or growth of CNV on FA or indocyanine green angiography, presence of subretinal or intraretinal fluid on OCT or a 10% or more increase in macular thickness in comparison with that at the last visit examination. In the absence of signs of CNV activity, the patient underwent the same examinations 1 month later to test for active disease. Tolerance of the injections also was assessed.
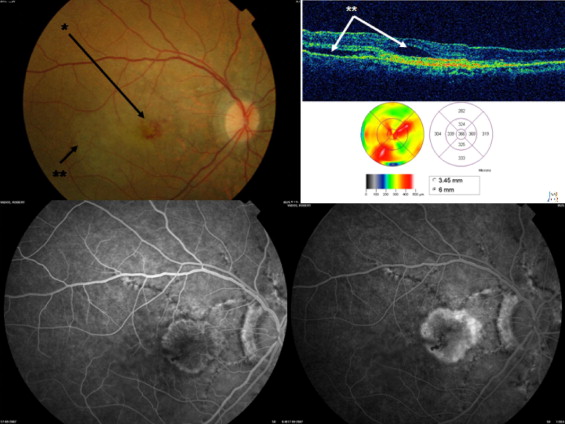
Significant changes in visual acuity were defined as a 3-line or more change from baseline BCVA. Posttreatment BCVA was classified as either stable or improved or as worse versus baseline. Posttreatment changes in macular thickness were defined, respectively, to OCT results as a 10% or more change versus baseline macular thickness or to subretinal or intraretinal fluid. The FA results were evaluated on the basis of whether CNV leakage was detected.
Ranibizumab injections were performed according to the Guidelines of the French Ophthalmologic Society. On the day of injection, patients lying in a sterile room received topical oxybuprocaine (Novesina; Novartis Pharmaceuticals, Basel, Switzerland; 3 eyedrops) followed by povidone–iodine drops on the conjunctival and corneal surfaces. Lids were kept open using a locking eyelid speculum. Ranibizumab 0.5 mg in a volume of 0.05 mL was injected with a 30-gauge needle into the vitreous cavity, 4 mm posterior to the limbus. Topical antibiotics were administered 4 times daily for the 4 days after each injection.
The total number of injections given and the follow-up time from baseline to the last visit were recorded for each patient. The primary end point was the percentage of patients whose BCVA stabilized or improved between baseline and the last follow-up visit. The secondary end points were the percentage of patients with stable or decreased macular thickness on Stratus OCT3 and the percentage of patients without leakage as demonstrated by FA at the last examination.
For statistical analysis, categorical variables were compared using the Fischer exact test, and quantitative data were compared using the Student t test. P < .05 was considered to be the threshold for significance.
Results
Thirty-five eyes of 27 patients (16 women and 11 men) with angioid streaks-associated subfoveal CNV (retrofoveal, juxtafoveal, or perifoveal) were treated with intravitreal ranibizumab ( Table 1 ). The mean age at initial treatment was 63.7 years (range, 30 to 86 years). All patients underwent a skin biopsy, and PXE was proven histologically in 11 (40.7%) of 27 patients. The mean time between the first diagnosis of CNV and treatment with ranibizumab was 39.4 months (range, 0 to 114 months). Ten (28.6%) of 35 eyes were CNV-treatment naïve, and 25 (71.4%) of 35 eyes already had been treated for CNV before the first ranibizumab injection. Previous treatments consisted of laser photocoagulation, photodynamic therapy (PDT), or both (14 eyes) or intravitreal injections (11 eyes: 7 treated with triamcinolone, 3 with pegaptanib sodium, and 1 with both; Table 1 ). In these 25 patients, the average time between the most recent previous treatment and the first ranibizumab injection was 11.3 months (range, 1 to 75 months). For the 11 of 25 patients who had received intravitreal injections previously, these treatments were administered at least 4 months before ranibizumab ( Table 1 ).
| Patient No. | Age (yrs) | Sex | Eye No. | Duration of Disease (CNV) at First Injection | Previous Treatment | Delay between Last Treatment and IR (mos) |
|---|---|---|---|---|---|---|
| 1 | 79 | F | 1 | 41 | 3 PDT + 2 IVT pegaptanib sodium | 4 (IVT 4) |
| 2 | 42 | F | 2 | 6 | 2 PDT | 6 |
| 3 | 70 | F | 3 | 44 | 5 PDT+ 1 LP + 1 IVT triamcinolone | 27 (IVT 42) |
| 3 | 70 | F | 4 | 0 | 0 | |
| 4 | 70 | M | 5 | 54 | 3 PDT | 54 |
| 5 | 43 | F | 6 | 11 | 1 PDT+ 1 IVT triamcinolone + 1 LP | 10 (IVT 10) |
| 6 | 71 | F | 7 | 28 | 2 PDT | 1 |
| 7 | 73 | M | 8 | 15 | 2 PDT + 2 LP | 1 |
| 8 | 78 | M | 9 | 80 | 6 PDT + 1 LP | 1 |
| 9 | 79 | F | 10 | 84 | 1 radiotherapy + 4 PDT + 1 IVT triamcinolone | 1 (IVT 30) |
| 9 | 79 | F | 11 | 40 | 0 | |
| 10 | 54 | M | 12 | 30 | 2 LP (extrafoveal CNV) | 1 (IVT 24) |
| 10 | 54 | M | 13 | 76 | 6 PDT + 4 LP + 2 IVT triamcinolone | 75 |
| 11 | 51 | F | 14 | 15 | 0 | |
| 12 | 30 | F | 15 | 29 | 0 | |
| 13 | 79 | F | 16 | 0 | 0 | |
| 14 | 59 | M | 17 | 60 | 0 | |
| 15 | 78 | F | 18 | 96 | 2 PDT | 1 |
| 16 | 59 | F | 19 | 19 | 0 | |
| 17 | 60 | M | 20 | 55 | 2 LP + 9 PDT + 3 IVT triamcinolone | 4 (IVT 4) |
| 18 | 76 | F | 21 | 41 | 0 | |
| 19 | 64 | F | 22 | 111 | 1 LP + 1 PDT | 1 |
| 19 | 64 | F | 23 | 114 | 1PDT + 1 IVT pegaptanib sodium | 3 (IVT 20) |
| 20 | 86 | F | 24 | 25 | 3 PDT | 10 |
| 21 | 65 | F | 25 | 25 | 5 PDT+ 2 IVT triamcinolone | 2 (IVT 16) |
| 22 | 48 | M | 26 | 119 | 1 LP + 3 PDT | 24 |
| 22 | 48 | M | 27 | 6 | 1 PDT | 1 |
| 23 | 72 | F | 28 | 42 | 0 | |
| 24 | 58 | F | 29 | 43 | 4 PDT +1 IVT triamcinolone + 2 IVT pegaptanib sodium | 1 (IVT 5) |
| 24 | 58 | F | 30 | 43 | 7 PDT + 1 IVT triamcinolone | 1 (IVT 27) |
| 25 | 70 | M | 31 | 0 | 1 PDT | 1 |
| 25 | 70 | M | 32 | 0 | 1 PDT | 1 |
| 26 | 59 | M | 33 | 0 | 2 PDT + 1 IVT pegaptanib sodium | 12 (IVT 12) |
| 27 | 48 | M | 34 | 0 | 0 | |
| 27 | 48 | M | 35 | 2 | 1 PDT | 2 |
The mean duration of follow-up for this cohort of patients was 24.1 ± 3.0 months (range, 6 to 37 months). The mean number of intravitreal ranibizumab injections was 5.7 ± 1.2 (range, 2 to 14 injections). Mean BCVA at baseline was 20/43 (range, 20/400 to 20/20). Seven patients had a baseline BCVA of 20/20, associated with juxtafoveal or perifoveal CNV. Post-treatment improvement in visual acuity (gain of ≥ 3 lines) occurred in 4 (11.4%) of 35 eyes, no change (± 3 lines) in visual acuity was observed in 26 (74.3%) of 35 eyes, and loss of visual acuity (≥ 3 lines) occurred in 5 (14.3%) of 35 eyes ( Figure 2 ) . Finally, BCVA was stabilized or improved in 30 (85.7%) of 35 eyes.
At baseline, 12 (34.3%) of 35 eyes had a BCVA of 20/200 or less. At the last visit, 8 (22.9%) of 35 eyes had a BCVA of 20/200 or less. At baseline, 17 (48.6%) of 35 eyes had a BCVA 20/40 or more. At the last visit, 15 (42.9%) of 35 eyes had a BCVA of 20/40 or more. At the end of follow-up, mean BCVA was 20/46 (range, 20/400 to 20/20). The mean variation in visual acuity was −0.17 ± 1.0 line (range, −10 to +7 lines), corresponding to +0.017 ± 0.10 line using logarithm of the minimal angle of resolution charts (no statistical difference, P = .73; Table 2 ).
| Eye No. | No. of IR Treatments | Duration of Follow-up (mos) | Initial VA | Final VA | Variation of VA (EDTRS Lines) | Initial OCT Macular Thickness (μm) | Final OCT Macular Thickness (μm) | Variation of Macular Thickness (%) | Leakage on FA at Last Follow-up Examination |
|---|---|---|---|---|---|---|---|---|---|
| 1 | 14 | 34 | 20/20 | 20/32 | −2 | 147 | 195 | +33 | No |
| 2 | 3 | 6 | 20/400 | 20/200 | +3 | 296 | 200 | −32 | No |
| 3 | 3 | 19 | 20/400 | 20/400 | 0 | 510 | 1000 | +96 | No |
| 4 | 8 | 9 | 20/50 | 20/64 | −1 | 334 | 359 | +7 | Yes |
| 5 | 8 | 22 | 20/40 | 20/80 | −3 | 211 | 422 | +100 | Yes |
| 6 | 7 | 36 | 20/20 | 20/20 | 0 | 235 | 255 | +9 | No |
| 7 | 8 | 14 | 20/32 | 20/40 | −1 | 297 | 272 | −8 | No |
| 8 | 4 | 20 | 20/40 | 20/32 | +1 | 272 | 344 | +26 | No |
| 9 | 3 | 25 | 20/200 | 20/120 | +2 | 168 | 183 | +9 | Yes |
| 10 | 4 | 27 | 20/200 | 20/80 | +4 | 229 | 293 | +28 | Yes |
| 11 | 3 | 22 | 20/64 | 20/120 | −3 | 226 | 324 | +43 | No |
| 12 | 5 | 30 | 20/400 | 20/160 | +4 | 174 | 196 | +13 | No |
| 13 | 7 | 26 | 20/32 | 20/40 | −1 | 336 | 215 | −36 | No |
| 14 | 3 | 20 | 20/40 | 20/40 | 0 | 176 | 213 | +21 | No |
| 15 | 4 | 23 | 20/25 | 20/20 | +1 | 392 | 402 | +3 | No |
| 16 | 4 | 10 | 20/25 | 20/20 | −1 | 322 | 254 | −21 | No |
| 17 | 3 | 16 | 20/20 | 20/20 | 0 | 253 | 249 | −2 | No |
| 18 | 6 | 26 | 20/20 | 20/32 | −2 | 280 | 290 | +4 | No |
| 19 | 8 | 32 | 20/20 | 20/25 | −1 | 157 | 246 | +57 | Yes |
| 20 | 3 | 25 | 20/40 | 20/25 | +2 | 151 | 182 | +21 | Yes |
| 21 | 3 | 10 | 20/50 | 20/50 | 0 | 247 | 251 | +2 | No |
| 22 | 2 | 24 | 20/32 | 20/25 | +1 | 179 | 234 | +31 | No |
| 23 | 2 | 10 | 20/80 | 20/80 | 0 | 137 | 234 | +71 | Yes |
| 24 | 4 | 8 | 20/200 | 20/160 | +1 | 264 | 195 | −26 | No |
| 25 | 4 | 31 | 20/200 | 20/200 | 0 | 257 | 189 | −26 | No |
| 26 | 12 | 37 | 20/250 | 20/200 | −1 | 191 | 432 | +126 | Yes |
| 27 | 11 | 28 | 20/20 | 20/20 | 0 | 230 | 281 | +22 | No |
| 28 | 3 | 30 | 20/200 | 20/200 | 0 | 389 | 242 | −38 | Yes |
| 29 | 12 | 36 | 20/64 | 20/40 | +2 | 229 | 209 | −9 | No |
| 30 | 2 | 36 | 20/200 | 20/200 | 0 | 246 | 398 | +62 | Yes |
| 31 | 3 | 29 | 20/50 | 20/120 | −4 | 441 | 232 | −47 | No |
| 32 | 9 | 29 | 20/20 | 20/200 | −10 | 231 | 301 | +30 | No |
| 33 | 4 | 29 | 20/250 | 20/400 | −2 | 284 | 455 | +60 | No |
| 34 | 14 | 30 | 20/32 | 20/120 | −6 | 358 | 245 | −32 | Yes |
| 35 | 6 | 35 | 20/400 | 20/80 | +7 | 204 | 185 | −9 | Yes |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


