Purpose
To assess intraoperative variation of corneal thinnest point in keratoconic patients undergoing riboflavin/ultraviolet type A (UV-A) cross-linking treatment using noncontact time-domain optical pachymetry.
Design
Prospective, noncomparative interventional study.
Methods
Ten patients underwent epithelium-off riboflavin/UV-A corneal cross-linking at Siena University Hospital; corneal thickness was measured using optical coherence tomography at the following times: preoperatively with epithelium on; after removal of a 9-mm-diameter disc of epithelium; immediately after instillation of 2 drops of riboflavin 0.1%–dextran T 500 20% solution; after repeated instillation of riboflavin 0.1%–dextran T 500 20% solution every 2.5 minutes, at 10, 20, and 30 minutes of soaking time; at the end of the treatment after 30 minutes of UV-A exposure with the riboflavin biofilm in situ; and finally after washing the riboflavin biofilm.
Results
The most significant decrease in thinnest point measurement was detected meanly in the first 10 minutes of corneal soaking. In this interval, the minimum recommended in various studies, a mean reduction of −79.28 μm (−17.61% of initial thinnest point value after removal of the epithelium) was recorded. No adverse events were recorded.
Conclusions
The study demonstrates a statistically significant intraoperative reduction in corneal thinnest point value during epithelium-off cross-linking procedure using standard riboflavin 0.1%–dextran 20% solutions. Intraoperative optical pachymetry evaluation during cross-linking should be recommended before starting UV-A irradiation, and if a thickness under 380 μm is detected, the stroma should be reexpanded with hypotonic dextran-free riboflavin solutions.
Keratoconus is a degenerative noninflammatory ectatic disease of the cornea characterized by biomechanical instability of stromal collagen leading to a reduction in corneal thickness, a variation in posterior and anterior corneal curvature, and a loss of visual acuity attributable to irregular astigmatism with or without a reduction in corneal transparency.
Riboflavin–ultraviolet A (UV-A)-induced corneal collagen cross-linking is increasingly proposed as a therapeutic approach to progressive keratoconus and for progressive ectasia of the cornea in general. The aim of the method is to slow down or block progress, avoiding or delaying recourse to keratoplasty.
The surgical technique of standard cross-linking involves removal of the epithelium to favor penetration of riboflavin into the stroma and the anterior chamber, ensuring sufficient efficacy while protecting the endothelium and posterior eye structures. A controversial aspect of the technique is how long the stroma should be imbibed with the solution (usually riboflavin 0.1% and dextran T 500 20%) in order to ensure a sufficient intrastromal concentration of vitamin B 2 and its presence in the anterior chamber to maximize the efficacy of treatment and shielding.
Since the literature does not suggest unequivocal imbibition times, the cross-linking procedure is still not fully standardized. Indeed, certain authors continue to recommend and use an imbibition time of 5 minutes after removal of the epithelium, as originally proposed by Wollensak and associates in 2003 (first international report), repeated by Caporossi and associates in 2006 (second international report). Subsequently, Spoerl and associates proposed an imbibition time of 30 minutes, while the Siena School proposed a standard time of 10-15 minutes. Actually, no significant differences in clinical results of cross-linking in relation to imbibition time have ever been demonstrated, making the time beyond the first necessary 10 minutes still an open question of subjective experience.
However, a study by our group on intrastromal concentration of riboflavin, measured by high-precision liquid chromatography (HPLC), demonstrated that 10 minutes of imbibition is more than enough to ensure a sufficient concentration of riboflavin in the stroma. This aspect is not secondary, since Kymionis and associates demonstrated by intraoperative ultrasound (US) pachymetry that, during impregnation of the stroma with standard 0.1% riboflavin solution containing 20% dextran of 500 000 Da molecular weight (MW), a significant intraoperative reduction in stromal thickness occurs, exposing the endothelium and eye structures behind it to radiation damage. However, ultrasound pachymetry has certain limits because it is a nonrepeatable contact method in identifying the thinnest point of the keratoconic cornea. The present prospective observational clinical study aimed to assess intraoperative variation in corneal thickness during cross-linking treatment by time-domain noncontact optical pachymetry mapping, involving readings at regular, significant intervals during corneal soaking time and at the end of treatment (after 30 minutes of UV-A irradiation). An in vivo intraoperative pachymetric map seeks to determine the best time to interrupt imbibition and safely begin UV-A exposure.
Patients and Methods
The study design was an open prospective clinical study.
Ten patients (8 male and 2 female) with progressive keratoconus undergoing riboflavin–UV-A-induced corneal collagen cross-linking (CXL) with removal of epithelium (10 eyes) were enrolled in the study after the approval of the Ethics Committee of Siena University Hospital. Inclusion criteria were: age between 16 and 28 years (average age 21.5 years); Krumeich stage I or II progressive keratoconus; documented clinical, topographic, and pachymetric deterioration; minimum corneal thickness greater than 400 μm measured by optical pachymetry at the thinnest point; and absence of stromal opacities, concomitant infections, autoimmune diseases, and severe dry eye. Pachymetric readings before, during, and after the operation were made with a Visante OCT (Zeiss, Jena, Germany) time-domain optical coherence tomography (OCT) noncontact optical system inside the operatory room. Statistical analysis was performed using a Wilcoxon test. All analyses were done using SPSS v16.0. A P value ≤.05 was considered to be statistically significant.
Main Outcome Measures
Pachymetric measurements of corneal thickness were made at the following times: preoperative (epithelium-on), outside the operating room before the operation; epithelium-off, immediately after removal of an 9-mm-diameter disc of epithelium with a blunt metal spatula and instillation of a drop of oxybuprocaine to anesthetize the naked stroma; immediately after instillation of 2 drops of sterile riboflavin 0.1%–dextran T 500 20% solution (Ricrolin; Sooft, Montegiorgio AP, Italy), to assess any effects of the solution on corneal thickness (reflection of light, meniscus thickness); at 10 minutes, after repeated instillation of solution every 2.5 minutes; at 20 minutes, after repeated instillation of solution every 2.5 minutes; at 30 minutes, after repeated instillation of solution every 2.5 minutes; at the end of treatment, after 30 minutes of exposure to UV-A and instillation of solution every 2.5 minutes, with riboflavin biofilm in situ; at the end of treatment, after washing the riboflavin biofilm with a balanced saline solution (BSS).
In all patients, CXL was performed according to the standard protocol (Dresden modified): exposure to the UV-A source (Caporossi, Baiocchi, Mazzotta, Vega X-Linker; C.S.O., Florence, Italy) in 6 5-minute steps (total 30 minutes), irradiating an area of 8 mm in diameter (power 3 mW/cm 2 ); instillation of a solution containing 0.1% riboflavin and 20% dextran every 2.5 minutes; corneal washing with sterile BSS, and antibiotic medication with levofloxacin and cyclopentolate eye drops, followed by application of a therapeutic soft corneal contact lens (Schalcon S.p.A., Rome, Italy). Patients were given topical antibiotic therapy (ofloxacin eye drops), nonsteroidal anti-inflammatory drug (diclofenac eye drops), and artificial tears (sodium hyaluronate 0.2%) to be applied 4 times a day until removal of the contact lens on day 4. Then a topical steroid was prescribed (fluorometholone 0.2% drops 4 times a day for 4 weeks, subsequently tapered to zero over 4-6 weeks). Artificial tears were maintained at 1 drop 3 times per day for 3 months after removal of the corneal lens.
Results
The results for the average thinnest point provided by the Visante OCT are reported below and summarized in the Table .
| Average Corneal Thickness TP ± SD (μm) | Δ Thickness ± SD | % | |
|---|---|---|---|
| Preop epi-on | 473.29 ± 16.98 | – | – |
| Epi-off | 450.29 ± 15.71, P = .06 | 0 | 0 |
| Immediately after Ricrolin | 438.71 ± 19.02, P = .10 | −11.57 ± 11.54 | −2.57 |
| 10 min imbibition Ricrolin | 371.00 ± 19.07, P = .031 | −79.28 ± 9.25 | −17.61 |
| 20 min imbibition Ricrolin | 335.71 ± 19.00, P = .031 | −114.57 ± 11.83 | −25.44 |
| 30 min imbibition Ricrolin | 305.86 ± 23.29, P = .039 | −144.43 ± 18.85 | −32.07 |
| After 30 min UV-A, biofilm in situ | 346.71 ± 24.12, P = .031 | −103.57 ± 19.69 | −23.00 |
| After 30 min UV-A, without biofilm | 359.71 ± 10.03, P = .031 | −90.57 ± 16.75 | −20.11 |
Average minimum corneal thickness (thinnest point) thickness measured with the epithelium in situ was 473.29 ± 16.98 μm (range 437-526 μm). After epithelial removal, average thickness was 450.29 ± 15.71 μm (range 413-492 μm) ( P = .06), equal to 95.14% of the initial thickness, with an average decrease of 23 ± 8.86 μm (4.86% of the initial thickness). Average thickness immediately after the instillation of 2 drops of Ricrolin solution (riboflavin 0.1% and dextran T 500 20%) and 1 drop of oxibuprocaine chlorhydrate was 438.71 ± 19.02 μm (range 402-504 μm) ( P = .10), with a not statistically significant change of −11.57 ± 11.54 μm (2.57% of average thickness after epithelial removal) ( Figure 1 ).
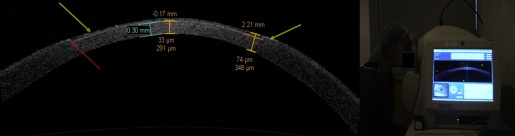
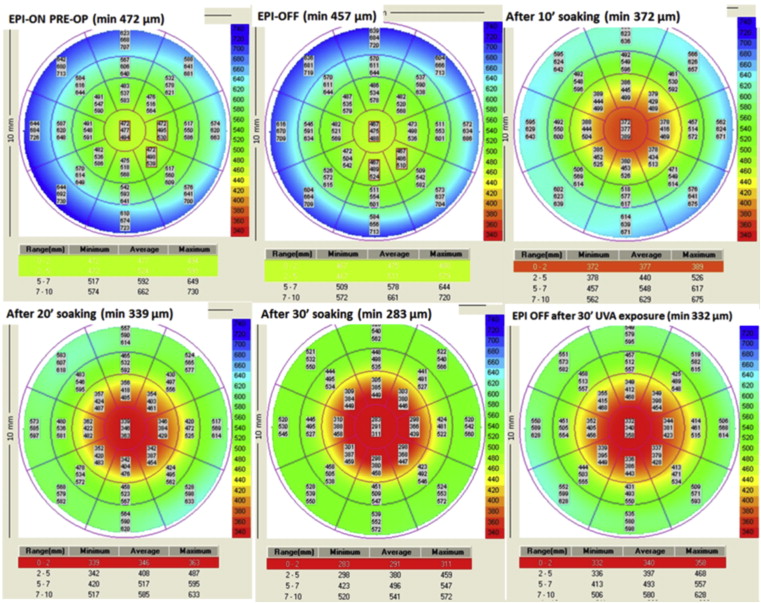
Average minimum corneal thicknesses measured after 10, 20, and 30 minutes of imbibition with Ricrolin, instilled every 2.5 minutes, were as follows: after 10 minutes of imbibition: 371.00 ± 19.07 μm (range 335-440 μm), P = .031; after 20 minutes of imbibition: 335.71 ± 19.07 μm (range 305-414 μm), P = .031; after 30 minutes of imbibition: 305.86 ± 23.29 μm (range 280-410 μm), P = .039. Respective decrements were −79.28 ± 9.25 μm after 10 minutes (−17.61%); −114.57 ± 11.83 μm after 20 minutes (−25.44%); and −144.43 ± 18.85 μm after 30 minutes (−32.07%) ( Figure 2 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


