Purpose
To explore the effect of body position (sitting vs supine) on intraocular pressure (IOP) in children, as assessed by the Icare PRO and the Tono-Pen.
Design
Prospective clinical study.
Methods
Children with known or suspected glaucoma and those without glaucoma were recruited from the Duke Eye Center pediatric clinic. Subjects underwent tonometry in both eyes while upright (sitting), after instillation of topical anesthetic, with either the Icare PRO or the Tono-Pen first, and then the second instrument (order randomized). Goldmann applanation tonometry (GAT) was then performed by a clinician masked to the previous measurements. The subjects were then placed in the supine position for 5 minutes, and tonometry using the Icare PRO and the Tono-Pen was obtained, in the same order used when they were the sitting position.
Results
Enrolled were 47 children (94 eyes). Mean seated IOP for GAT, Icare PRO and Tono-Pen were 16.4 ± 4.2, 17.5 ± 3.5, and 18.0 ± 3.9 mm Hg, respectively. The mean supine IOP for the Icare PRO and Tono-Pen were 18.4 ± 4.5 and 18.8 ± 4.2 mm Hg, respectively. This rise was +0.9 ± 2.3 mm Hg for Icare PRO ( P = 0.01) and +0.7 ± 1.8 mm Hg for Tono-Pen ( P = 0.009), respectively.
Conclusion
In children, Icare PRO tonometry correlates well with GAT in the sitting position, and with the Tono-Pen in both the sitting and supine positions. IOP rises when a child changes position from sitting to supine when measured by the Icare PRO or the Tono-Pen. However, the increase, which is less than 1 mm Hg, seems clinically insignificant and is unlikely to alter glaucoma management in children.
Measurement of intraocular pressure (IOP) is critical in the management of pediatric glaucoma. When accurate IOP measurement in infants and young children with known or suspected glaucoma cannot be accomplished in the clinic, examination under anesthesia may be required. The clinical introduction of rebound tonometry has positively impacted the management of pediatric glaucoma. The Icare (Icare Finland, Helinski, Finland) is a portable handheld rebound tonometer that does not require the use of topical anesthetic. It has been well received in the pediatric population and has been demonstrated to give reproducible results, to correlate fairly well with Goldmann applanation tonometry (GAT), and to decrease the number of examinations requiring anesthesia.
Although the original Icare rebound tonometer is commercially available and has been approved by the Food and Drug Administration for use in the United States, this device can be used only when the patient is in an upright position. A newer Icare rebound tonometer, the Icare PRO (Icare), which can be used in both the upright and the horizontal positions for IOP measurement, is commercially available in Europe but has not yet been approved by the Food and Drug Administration in the United States. Like its predecessor, the Icare PRO uses a probe attached to a magnetic solenoid probe to rebound off the cornea. Unlike the earlier Icare, the Icare PRO uses a plastic probe and has a built-in inclination sensor that allows it to take readings with the instrument held horizontal (tip pointing downward), without loss of the probe. The Icare PRO also allows for storage of data (time of measurement, right or left eye and IOP) for later download. The Icare PRO computes an average of 6 sequential IOP measurements and has a color display that shows the IOP and its reliability as green, yellow or red in descending order. The Icare PRO can therefore be used for tonometry in any subject—infant, child or adult—in the supine position, whether in the office or in the operating room under anesthesia.
GAT measurements (obtained with the patient in the sitting position) are considered the gold standard for decision making in adults and in older children with glaucoma, despite the difficulties in acquiring these measurements in many children. Further, recent studies in adults have demonstrated changes in the measured IOP with changes in body position; the IOP rises when the adult lies down from a sitting position. There is little information about how IOP in children is altered by changes in body position. The Icare PRO allows for evaluation of this parameter in children and for interinstrument comparison of this effect by using different tonometers.
This study compares Icare PRO rebound tonometry to Tono-Pen (Medtronic Ophthalmics, Jacksonville, Florida, USA) tonometry in children with known and suspected glaucoma and also in normal subjects, measured with the child in both the upright (sitting) and supine positions (against GAT as standard). We have demonstrated that IOP measurements using Icare rebound tonometry do not change appreciably after instillation of topical anesthetic in children (personal data; accepted for publication). Additionally, repeated measurements of IOP using the Icare tonometer do not cause clinically-significant changes in measured IOP in children (personal data; accepted for publication).
Methods
Following Institutional Review Board approval by the Duke University Health System, this prospective study was conducted in the pediatric glaucoma clinic of one of the authors (SFF) at the Duke Eye Center. Written informed consent was obtained from a parent or guardian for all enrolled subjects. Written assent was obtained from subjects 12 years of age and older. The children were recruited during routine clinic visits and the group included only those who were known or expected to be cooperative for GAT, Tono-Pen and Icare tonometry. Excluded were children with corneal abnormalities or pronounced nystagmus known to preclude accurate GAT. Because the study subjects were carefully selected and most were established patients, we were able to include a few whose nystagmus did not preclude GAT.
Demographic information collected included age, sex, visual acuity, type of glaucoma (if present) and other ophthalmic diagnoses, number of prior ocular surgeries, number of current topical medications, and the presence of nystagmus, strabismus, or corneal abnormalities. Demographic data is summarized in the Table .
| No. of Subjects (%) | |
|---|---|
| Sample Size | 47 subjects/94 eyes (100%) |
| Sex: male/female | 20/27 (42.7%/57.4%) |
| Ethnicity: | |
| African American | 11 (23.4%) |
| Asian | 2 (4.3%) |
| White | 32 (68.1%) |
| Hispanic | 1 (2.1%) |
| Other | 1 (2.1%) |
| Nystagmus: no/yes | 38/9 (81%/19%) |
| Diagnoses | |
| Glaucoma: | |
| Aphakic | 18 (19.1%) |
| Primary congenital | 11 (11.7%) |
| Juvenile open angle | 2 (2.1%) |
| Uveitic | 10 (10.6%) |
| Glaucoma suspect | 9 (9.6%) |
| Other | 7 (7.4%) |
| Normal | 37 (39.3%) |
The subjects were randomized with respect to the tonometer initially used to obtain IOP measurements (Icare PRO vs Tono-Pen). The subjects were seated in the examining chair and topical anesthetic was placed into both eyes (proparacaine hydrochloride ophthalmic solution 0.5%). Two sequential measurements (right eye first followed by left eye, then repeated) were obtained using either the Icare PRO or the Tono-Pen, based on the randomization. Another 2 sequential IOP measurements were then obtained using the second tonometer. Only reliable measurements were recorded. If necessary, a third or fourth IOP measurement was obtained to ensure high reliability of the measurements. Fluorescein sodium and benoxinate hydrochloride ophthalmic solution 0.25%/0.4% was then instilled into both eyes, the corneas were examined for any defects, and 2 GAT measurements were obtained by the same examiner (SFF), who was masked to prior IOP measurements. The subject was then placed in the supine position with eyes closed for a period of 5 minutes, following which 2 IOP measurements were obtained using the Icare PRO and Tono-Pen, in the same instrument order as for upright testing for that subject. Figure 1 depicts the Icare PRO being used in the upright and supine positions. One Icare PRO and one Tono-Pen were used throughout the study and were in good working order. For purposes of simplicity, throughout the remainder of the text, upright position refers to the child’s sitting upright in the examining chair, whereas supine position refers to the child’s lying down (supine) and looking up toward the ceiling.
For analysis of data, the IOPs from the right and left eyes were averaged together for each subject (n = 47 total). Subjects’ characteristics were tabulated by using number and percent for categorical data and by using median, range and standard deviation (SD) for continuous variables. Comparisons between groups for continuous variables were completed with the paired t test as appropriate. All statistical tests were 2-sided, and the threshold for significance was set at α = 0.05. All values are reported as mean ± SD unless otherwise noted. Linear regression analysis and Bland-Altman analysis were performed using GraphPad Prism for Windows, v 5.01 (GraphPad Software, San Diego, California, USA).
Results
Included were 47 subjects (94 eyes) with a mean age of 12.7 years (range, 8–17). Demographic and clinical data are recorded in the Table . Of the eyes, 48 (51%) had established diagnoses of glaucoma, 9 eyes (10%) were being followed as glaucoma suspects, and 37 eyes (39%) were normal.
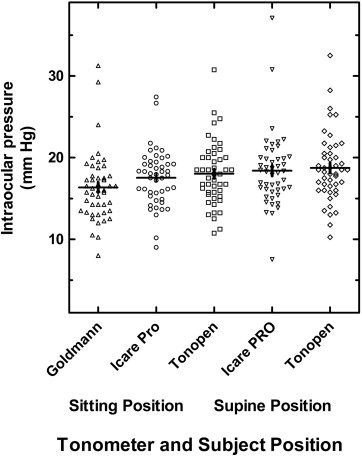
Clinic examiners were able to obtain Icare PRO, Tono-Pen and GAT measurements of acceptable reliability in both the upright and supine positions in all enrolled subjects. The median (range) IOPs measured by GAT was 16 mm Hg (8–31). The mean IOPs measured in the sitting position using GAT, Icare PRO and Tono-Pen were 16.4 ± 4.2, 17.5 ± 3.5, and 18.0 ± 3.9 mm Hg, respectively. The mean IOPs measured in the supine position using the Icare PRO and Tono-Pen were 18.4 ± 4.5 and 18.8 ± 4.2 mm Hg, respectively ( P > 0.05). Figure 2 depicts the IOP measurements obtained for all study subjects, using the 3 different tonometers and in the 2 different body positions.