Purpose
To evaluate the efficacy and safety of intracameral triamcinolone acetonide (TA) in glaucoma surgery.
Design
Prospective randomized clinical trial.
Methods
setting : Institutional—Wills Eye Hospital. study population : Patients undergoing trabeculectomy (with or without cataract surgery) or tube shunt surgery. intervention : Patients were randomized to receive intracameral TA or balanced salt solution at the end of surgery. Follow-up time was 6 months. main outcome measures : Intraocular pressure, visual acuity, inflammation measured by slit-lamp examination and laser flare meter, cataract grading, bleb appearance, dry eye scores, use of supplemental medical therapy, surgical success, and rate of complications.
Results
Seventy-seven patients were enrolled in the study, including 37 in the TA group and 40 in the control group. There were no significant differences in success rates between the 2 groups ( P = .60). Intraocular pressure and medication use were similar between the groups for each follow-up visit. Dry eye scores were lower in the TA group at month 1 ( P = .042), while flare scores were higher in the TA group on day 1 ( P = .015) but lower at month 1 ( P = .044). The complication rates were higher in the TA group on day 1 ( P = .04). All other outcome measures were similar for both groups.
Conclusions
Intracameral TA did not affect the success rates or change the complication rates of glaucoma surgery.
Modulation of wound healing is an important determinant of the success of glaucoma surgery. Failure of glaucoma-filtering surgery has long been known to be related to fibroblastic proliferation and cellular inflammation. The use of intraoperative antimetabolites has greatly increased the success of glaucoma filtering surgery, and the benefits of anti-inflammatory medications in the postoperative management of glaucoma surgery are well established. Topical corticosteroid drops result in lower postoperative intraocular pressure (IOP) and a higher rate of thin, cystic bleb formation after trabeculectomy surgery. Few studies have examined the role of intraoperative corticosteroid use during glaucoma surgery.
Triamcinolone acetonide (TA; Triesence; Alcon, Fort Worth, Texas, USA) is a sterile, nonpreserved injectable corticosteroid ophthalmic suspension. The use of TA in ocular surgery is well established in the form of intravitreal and sub-Tenon injections. Intracameral injection of TA has been studied in the pediatric population during cataract surgery, with positive results. Other studies have shown a good safety profile for the intracameral use of TA during cataract surgery, with no significant postoperative IOP elevation.
This study aimed to determine the efficacy and safety of intracameral TA in patients undergoing trabeculectomy, tube shunt, or combined cataract and glaucoma filtering surgeries, with or without adjunctive mitomycin C.
Methods
This prospective, randomized, interventional study enrolled eligible patients from April 2009 to July 2011 at the Wills Eye Hospital in Philadelphia, Pennsylvania.
The Institutional Review Board of the Wills Eye Hospital reviewed and approved the study protocol, and written permission was obtained from all patients. The trial was registered, and the information is publicly available at http://www.clinicaltrials.gov ( NCT00853905 ).
Patients
Adult patients (>18 years) with primary open-angle glaucoma (POAG); primary angle closure glaucoma (PACG); or pseudoexfoliation, pigmentary, traumatic, neovascular, or uveitic glaucoma (no inflammation for at least 6 weeks) were enrolled. The glaucoma surgeon determined whether these patients needed a standard trabeculectomy, tube shunt, or triple procedure (combined cataract extraction, intraocular lens implantation, and trabeculectomy), with or without adjunctive mitomycin C.
Exclusion criteria included patients under 18 years old; patients with any other type of glaucoma; anterior dysgenesis syndrome; elevated episcleral venous pressure from any cause, including active thyroid-related immune orbitopathy, carotid-cavernous fistula, Sturge-Weber syndrome, orbital tumors, or orbital congestive disease; any corneal pathology that would preclude measurement of cells and flare in the anterior chamber, including corneal dystrophies or any other opacities; history of ocular trauma or chemical burn; chronic ocular inflammatory disease; history of allergy to the active compound; and pregnancy or breastfeeding.
Patients were allocated into 3 groups based on the planned surgery: Group 1: trabeculectomy; Group 2: tube shunt surgery; Group 3: triple procedure. Patients were randomized to receive intraoperative TA vs balanced salt solution (BSS).
Surgical Technique and Postoperative Care
Patients underwent standard trabeculectomy, tube shunt, or triple procedure. After conjunctival reapproximation was complete at the end of the procedure, the surgeon reformed the anterior chamber with BSS.
For patients in the control group, viscoelastic was used to maintain adequate IOP if needed and this completed the procedure. For patients in the TA group, following reformation of the anterior chamber with BSS, the surgeon injected 0.1 cc TA into the anterior chamber using a tuberculin syringe with a 30 gauge cannula through the previously created paracentesis wound. The TA settled as a thick layer onto the iris and over the pupil. Next, the surgeon irrigated the anterior chamber with BSS until the pupillary border was visible underneath the remaining layer of TA over the iris and the IOP was within normal limits. Figure 1 summarizes the surgical technique used for intracameral TA injection. In order to standardize the amount of medication left in the anterior chamber, prior to surgery, the physician saw color photographs showing adequate, too little, and too much TA. Approximately 2 mg (0.05 mL) of TA was left in the anterior chamber following irrigation with BSS. When injection of viscoelastic material was necessary, the surgeon placed it in the anterior chamber.
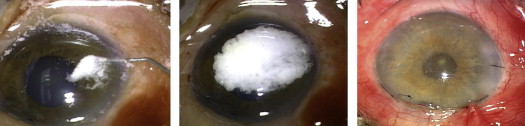
All patients received the same medication regimen of gatifloxacin 0.3% (Zymar; Allergan, Inc, Irvine, California, USA) 1 drop 4 times a day during the first week and prednisolone 1% (Falcon Pharmaceuticals, LTD, Fort Worth, Texas, USA) 1 drop 4 times a day for the first week, followed by a tapering course over an additional 5 weeks.
Data Collection and Outcome Measures
Baseline parameters included age, sex, race, glaucoma diagnosis, previous surgery, current glaucoma medication, and duration of glaucoma therapy. Complete ophthalmic examination was performed and flare in the anterior chamber was measured using the Kowa FM-500 laser flare meter (Kowa Optimed Europe Ltd, Dusseldorf, Germany).
Patients were followed on postoperative day 1, week 1 (days 5–9), month 1 (days 25–35), month 3 (days 80–100), and month 6 (days 140–220). At each visit, the following parameters were recorded: best-corrected visual acuity, bleb appearance, IOP, lens opacities, observation of cell and flare in the anterior chamber, and the need for glaucoma medications. The patients completed a questionnaire detailing their level of comfort on each visit.
Prior to surgery and at each postoperative visit, IOP was measured using a calibrated Goldmann applanation tonometer. Two measurements were recorded and averaged. If the measurements differed by more than 2 mm Hg, a third pressure was taken and the average of the 3 was the final IOP.
The amount of anterior chamber inflammation was measured by the Kowa FM 500 laser flare meter on the preoperative visit and at the 1-month postoperative visit. The laser flare meter took 10 measurements per eye. It discarded the lowest and highest measurements and averaged the remaining 8 for a final reading with standard deviation. The measurement was expressed as photons per millisecond. Slit-lamp biomicroscopy graded the amount of inflammation to determine the correlation between the laser flare meter and slit-lamp examination.
Postoperative bleb appearance was evaluated at each postoperative visit, based on the Indiana Bleb Appearance Grading Scale (IBAGS). This system includes evaluation of bleb height, horizontal extent, vascularity, and amount of leak. Cataract was evaluated at baseline and at the 3- and 6-month postoperative visits using the Lens Opacities Classification System III (LOCS III). LOCS III evaluates 4 features based on a set of standardized photographs: nuclear opalescence, nuclear color, cortical cataract, and posterior subcapsular cataract.
IOP-lowering medications were added according to physician discretion, and the type of medication was recorded. The rate of complications was recorded, including shallow anterior chambers, hypotony, wound leaks, and choroidal detachments.
Efficacy was determined on the basis of patient comfort and clinical parameters: IOP, amount of anterior chamber inflammation, bleb morphology, and success of the surgery at postoperative months 3 and 6, defined as postoperative IOP ≤21 mm Hg and IOP reduction by 20%. Failure was defined as an inability to meet the aforementioned criteria for success, IOP less than 5 mm Hg, or a need for reoperation for glaucoma. These criteria were adopted from the Tube-Versus-Trabeculectomy Study. Safety was evaluated by assessing early and late surgical complications, formation of cataractous lens changes, and the presence of IOP spikes.
Statistical Analysis
Continuous variables were analyzed using medians, ranges, and categorical variables. The primary outcome, IOP, was analyzed using mixed-effects linear regression with fixed effects for treatment arm, time, treatment-by-time interaction, procedure type, and baseline IOP. Square root transformation was used to better meet assumptions of normality and constant variance across time.
The study assumed a first-order autoregressive covariance structure to account for correlation among repeated measurements from the same subject. It used a similar model to analyze comfort and inflammation scores. It included fixed effects for treatment arm, time, treatment-by-time interaction, and procedure type. It assumed a first-order autoregressive covariance structure. Prior to analysis, laser flare meter readings were log-transformed. Rates of complications and medication usage were evaluated using logistic regression. Generalized estimating equation methods were used to account for correlation among repeated measures. Owing to low variability in scores, opacity measures were compared using a nonparametric Wilcoxon rank sum test at months 3 and 6. P values were not adjusted for multiple comparisons.
The sample size of 77 participants provides 86% power to detect a decrease in failure rate from 30% to 5%, and 61% power to detect a decrease in the failure rate from 30% to 10%.
Randomization was performed electronically through www.graphpad.com . All analyses were performed using SAS version 9.3 (SAS Institute, Cary, North Carolina, USA).
Results
The study enrolled 77 patients. Randomization assigned 37 patients to receive intracameral TA injection and 40 patients to the control group. The baseline characteristics of study patients appear in Table 1 . No significant differences in any of the demographic or clinical features were observed between the groups at baseline. Of the 22 total tube shunt surgeries performed, 11 (6 TA, 5 control) were a Baerveldt BG 101–350 Glaucoma Implant (Abbott Laboratories, Inc, Abbott Park, Illinois, USA), 6 (2 TA, 4 control) were an Ahmed Glaucoma Valve Model FP7 (New World Medical Inc, Rancho Cucamonga, California, USA), and 5 (2 TA, 3 control) were a Molteno3 implant (Molteno Ophthalmic Limited, Dunedin, New Zealand).
| TA (N = 37) | Control (N = 40) | |
|---|---|---|
| Age, median (range) | 68 (47–88) | 69 (42–88) |
| Sex, n (%) | ||
| Male | 17 (46%) | 23 (58%) |
| Female | 20 (54%) | 17 (43%) |
| Race, n (%) | ||
| White | 17 (46%) | 15 (38%) |
| African-American | 16 (43%) | 23 (58%) |
| Hispanic | 2 (5%) | 1 (3%) |
| Other | 2 (5%) | 1 (3%) |
| DDLS, median (range) | 8 (0–10) (n = 36) | 8 (3–10) (n = 37) |
| IOP, median (range) | 24.5 (13–40) | 23.8 (8.5–56) |
| Visual acuity (logMAR), median (range) | 0.2 (0–1.3) | 0.4 (0–2.5) |
| Flare meter (average), median (range) | 1.0 (0.2–21.6) | 1.2 (0.1–169.5) |
| Flare meter (SD), median (range) | 1.1 (0.1–9) | 1.1 (0–30.5) |
| Diagnosis, n (%) | ||
| POAG | 25 (68%) | 28 (70%) |
| PACG | 6 (16%) | 3 (8%) |
| SOAG | 6 (16%) | 6 (15%) |
| SACG | 0 (0%) | 3 (8%) |
| Procedure, n (%) | ||
| Trabeculectomy | 20 (54%) | 20 (50%) |
| Tube shunt | 10 (27%) | 13 (33%) |
| Phaco-trabeculectomy | 7 (19%) | 7 (18%) |
| Medical history, n (%) (categories not exclusive) | ||
| Diabetes | 11 (30%) | 14 (35%) |
| High cholesterol | 15 (41%) | 15 (38%) |
| Hypertension | 28 (76%) | 30 (75%) |
| Asthma | 3 (8%) | 1 (3%) |
| Arthritis | 5 (14%) | 4 (10%) |
Table 2 shows model-adjusted estimates of mean IOP by group at each follow-up time. IOP was higher in the TA arm on day 1, but the difference did not reach statistical significance. No other differences were observed. Table 3 reports mean bleb appearance grading, dry eye/comfort, and inflammation scores by treatment arm and time. Dry eye scores were lower in the TA arm at month 1, while flare scores were higher in the TA arm on day 1 but lower at month 1. No other significant differences were observed.
| Time | TA | Control | P Value |
|---|---|---|---|
| Day 1 | 16.1 (13.1,19.05) | 12.8 (10.7,15.0) | .073 |
| Week 1 | 11.7 (9.1,14.5) | 11.8 (9.8,13.9) | .96 |
| Month 1 | 16.1 (13.0,19.4) | 14.4 (12.2,16.8) | .39 |
| Month 3 | 15.1 (12.2,18.4) | 12.8 (10.7,15.1) | .23 |
| Month 6 | 14.6 (11.7,17.8) | 13.6 (11.5,16.0) | .62 |
| Measure | Time | TA | Control | P Value |
|---|---|---|---|---|
| IBAGS, H | Day 1 | 1.7 (1.4,2.1) | 2.1 (1.7,2.5) | .070 |
| Week 1 | 1.7 (1.4,2.1) | 1.9 (1.5,2.3) | .38 | |
| Month 1 | 2.2 (1.8,2.6) | 1.9 (1.5,2.3) | .086 | |
| Month 3 | 2.0 (1.6,2.4) | 1.8 (1.4,2.2) | .51 | |
| Month 6 | 1.9 (1.5,2.3) | 2.1 (1.6,2.5) | .38 | |
| IBAGS, E | Day 1 | 2.5 (2.1,2.9) | 2.6 (2.3,2.9) | .54 |
| Week 1 | 2.3 (1.9,2.7) | 2.4 (2.1,2.8) | .47 | |
| Month 1 | 2.6 (2.2,3.0) | 2.3 (1.9,2.7) | .092 | |
| Month 3 | 2.4 (2.0,2.8) | 2.2 (1.9,2.6) | .52 | |
| Month 6 | 2.4 (2.0,2.8) | 2.3 (1.9,2.7) | .45 | |
| IBAGS, V | Day 1 | 2.4 (2.0,2.9) | 2.5 (2.1,2.9) | .76 |
| Week 1 | 2.2 (1.7,2.6) | 2.2 (1.8,2.7) | .76 | |
| Month 1 | 1.9 (1.5,2.4) | 1.7 (1.2,2.1) | .24 | |
| Month 3 | 1.6 (1.1,2.1) | 1.5 (1.0,1.9) | .58 | |
| Month 6 | 1.5 (1.0,1.9) | 1.1 (0.6,1.6) | .16 | |
| Pain | Day 1 | 0.4 (0.2,0.6) | 0.4 (0.2,0.6) | .87 |
| Week 1 | 0.3 (0.1,0.5) | 0.4 (0.2,0.6) | .40 | |
| Month 1 | 0.2 (0.0,0.4) | 0.2 (−0.0,0.4) | .85 | |
| Month 3 | 0.1 (−0.1,0.3) | 0.1 (−0.1,0.3) | .89 | |
| Month 6 | 0.2 (−0.0,0.4) | 0.2 (−0.0,0.3) | .78 | |
| FB sensation | Day 1 | 1.1 (0.8,1.4) | 0.8 (0.5,1.0) | .068 |
| Week 1 | 1.1 (0.9,1.4) | 0.8 (0.5,1.0) | .053 | |
| Month 1 | 0.6 (0.3,0.9) | 0.8 (0.5,1.0) | .37 | |
| Month 3 | 0.3 (0.0,0.6) | 0.4 (0.1,0.7) | .55 | |
| Month 6 | 0.4 (0.1,0.7) | 0.5 (0.3,0.8) | .57 | |
| Dry eye | Day 1 | 0.3 (0.1,0.6) | 0.1 (−0.1,0.4) | .21 |
| Week 1 | 0.6 (0.4,0.9) | 0.3 (0.0,0.5) | .054 | |
| Month 1 | 0.3 (0.0,0.5) | 0.6 (0.4,0.9) | .042 | |
| Month 3 | 0.4 (0.1,0.6) | 0.4 (0.2,0.7) | .79 | |
| Month 6 | 0.7 (0.4,0.9) | 0.7 (0.5,0.9) | .96 | |
| Blurred vision | Day 1 | 1.4 (1.0,1.7) | 1.2 (0.8,1.5) | .46 |
| Week 1 | 1.3 (0.9,1.6) | 1.2 (0.9,1.6) | .94 | |
| Month 1 | 1.2 (0.9,1.5) | 1.1 (0.7,1.4) | .66 | |
| Month 3 | 1.0 (0.6,1.3) | 1.0 (0.7,1.4) | .78 | |
| Month 6 | 1.1 (0.7,1.4) | 1.1 (0.7,1.5) | .95 | |
| Tearing | Day 1 | 0.7 (0.4,1.0) | 0.8 (0.6,1.1) | .38 |
| Week 1 | 0.5 (0.2,0.7) | 0.8 (0.5,1.0) | .101 | |
| Month 1 | 0.7 (0.4,1.0) | 0.8 (0.5,1.0) | .68 | |
| Month 3 | 0.5 (0.2,0.8) | 0.5 (0.2,0.7) | .92 | |
| Month 6 | 0.7 (0.5,1.0) | 0.5 (0.3,0.8) | .24 | |
| Itching | Day 1 | 0.6 (0.4,0.9) | 0.6 (0.4,0.9) | .97 |
| Week 1 | 0.8 (0.6,1.1) | 0.6 (0.3,0.8) | .19 | |
| Month 1 | 0.5 (0.2,0.8) | 0.5 (0.2,0.7) | .87 | |
| Month 3 | 0.6 (0.3,0.8) | 0.6 (0.3,0.8) | .89 | |
| Month 6 | 0.8 (0.5,1.0) | 0.5 (0.3,0.8) | .20 | |
| Cells | Day 1 | 2.1 (1.5,2.8) | 1.8 (1.3,2.3) | .42 |
| Week 1 | 2.0 (1.4,2.6) | 1.4 (0.9,1.9) | .14 | |
| Month 1 | 1.5 (0.9,2.0) | 1.2 (0.7,1.7) | .55 | |
| Month 3 | 0.6 (−0.0,1.1) | 0.7 (0.2,1.3) | .63 | |
| Month 6 | 0.7 (0.1,1.2) | 0.6 (0.0,1.1) | .83 | |
| Flare | Day 1 | 1.9 (1.3,2.4) | 1.0 (0.5,1.4) | .015 |
| Week 1 | 1.5 (1.0,2.0) | 1.3 (0.8,1.7) | .48 | |
| Month 1 | 0.4 (−0.2,0.8) | 1.0 (0.6,1.4) | .044 | |
| Month 3 | 0.6 (0.0,1.1) | 0.4 (−0.1,0.9) | .64 | |
| Month 6 | 0.3 (−0.2,0.8) | 0.2 (−0.2,0.7) | .86 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


