Purpose
To observe morphologic changes and the distribution of dendritic cells in pterygium using in vivo laser scanning confocal microscopy (LSCM).
Design
Prospective comparative study.
Methods
Twenty-six eyes of 26 patients with pterygium and 17 eyes of 17 healthy subjects were recruited. Using LSCM, in vivo images of the pterygium and adjacent clear cornea were captured. The density of basal corneal epithelial cells and keratocytes in the anterior and posterior stroma and the density of dendritic cells in the pterygium and adjacent clear cornea were determined. In the controls, the central cornea and nasal bulbar conjunctiva were imaged. The density of basal corneal epithelial cells, keratocytes, and dendritic cells was evaluated.
Results
Morphologic alterations of the sub-basal nerve plexus were observed in pterygium. The density of basal corneal epithelial cells and anterior keratocytes in pterygium was 5359.0 ± 543.1 cells/mm 2 and 407.4 ± 188.7 cells/mm 2 respectively, which was significantly lower than that in the controls ( P < .001). The density of dendritic cells in the clear corneas of pterygia was 60.3 ± 25.5 cells/mm 2 , which was significantly higher than the 23.6 ± 11.1 cells/mm 2 in the central corneas of controls ( P < .001). The dendritic cell density in the pterygium was significantly higher than the density in the nasal bulbar conjunctiva of controls ( P < .001).
Conclusions
Histopathologic alterations and increased dendritic cells were evident in pterygium and the adjacent clear cornea by in vivo LSCM. In vivo LSCM was found to be an effective method of observing the morphologic alterations of pterygium.
A pterygium is a wing-shaped, fibrovascular conjunctival outgrowth that centripetally invades the clear cornea. It is a common ocular surface condition that can cause ocular discomfort, cosmetic deformity, and aberrations in visual function. Chronic exposure to ultraviolet-B (UVB) light has been attributed as the major cause of pterygia. Growing evidence implicates many other factors in the development of pterygia, including anti-apoptotic and immunologic mechanisms, cytokines, growth factors, extracellular matrix modulators, genetic factors, and viral infections. Evidence has been inconclusive and the pathogenesis of pterygia remains unclear.
Previous studies relied primarily on surgically excised tissue samples, which have provided few details about the pterygium and underlying cornea interface and do not allow for analysis of the structural alterations in the clear cornea adjacent to the pterygium. A method in which the in situ histologic characteristics of both the pterygium and the adjacent cornea could be evaluated would undoubtedly provide more information about the pathogenesis and characteristics of pterygia.
In vivo laser scanning confocal microscopy (LSCM) is a method widely used to observe morphologic changes of the cornea on a cellular level. Recently, 2 groups have performed studies using LSCM to observe pterygia in situ. The studies from Papadia and associates observed the central cornea of pterygium patients and found decreased epithelial cell density and increased dendritic-like cells, while the study from Zhivov and associates described the morphologic characteristics of pterygium. These studies demonstrate the potential uses of LSCM in pterygium research and suggest the importance of dendritic-like cells in the pathogenesis of pterygium.
Up to this point, in vivo studies of pterygia remain limited. The purpose of our work was 2-fold. First, we sought to perform a comprehensive investigation of the morphologic characteristics of not only the pterygium, but also the adjacent cornea using in vivo LSCM. Secondly, we sought to provide a quantified study of the distribution of dendritic cells and of the changes in density of corneal epithelial cells and keratocytes in patients with pterygium.
Materials and Methods
Study Subjects
Twenty-six eyes of 26 patients consecutively examined at the general outpatient clinic of the Eye and ENT Hospital of Fudan University were recruited for this prospective study. Seventeen right eyes of 17 age-matched healthy subjects were included as controls. Patients with known systemic diseases associated with ocular surface disease, a history of ocular surgery, systemic or topical drug use, or contact lens use were excluded from the study.
Slit-Lamp Biomicroscopy
Slit-lamp biomicroscopy was performed on each subject. Photographs taken at the slit lamp were used to measure the length and width of the pterygium.
In Vivo Laser Scanning Confocal Microscopy Examination
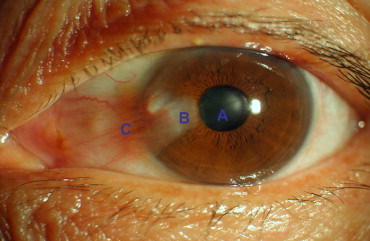
The Heidelberg Retina Tomograph/Rostock Cornea Module (Heidelberg Engineering GmbH, Dossenheim, Germany) was used in this study. A 60× water-immersion objective lens (Olympus Europa GmbH, Hamburg, Germany) is used with a 670-nm diode laser as a light source to allow a scanning area of 384 × 384 μm, with lateral and vertical resolutions of 1 μm and up to 800× magnification. Just prior to examination, 1 drop of 0.4% oxybuprocaine hydrochloride (Benoxil; Santen Pharmaceutical Co., Ltd, Osaka, Japan) was applied to the lower conjunctival sac. In vivo LSCM was performed on each eye with pterygium in 3 areas ( Figure 1 ): clear cornea adjacent to the pterygium (A), pterygium head (B), pterygium body (C). Images were captured at all 3 points in the same sequence for all subjects. The central cornea, nasal limbus, and nasal bulbar conjunctiva were examined in healthy subjects. The images were recorded at 1 point along the z-axis as a single scan or in the movie-motion mode.
Analysis of the In Vivo Laser Scanning Confocal Microscopy Images
The morphologic characteristics of the pterygium, cornea, and the sub-basal nerve plexus were evaluated. In the clear cornea adjacent to the pterygium, the density of basal epithelial cells, keratocytes in the anterior and posterior stroma, and dendritic cells was determined. The density of dendritic cells in the pterygium head and pterygium body was also determined. In the central cornea of control subjects, the density of dendritic cells, basal corneal epithelial cells, and keratocytes in the anterior and posterior stroma was determined. The density of dendritic cells was also calculated in the nasal bulbar conjunctiva. Three clear images (without motion blur or compression lines) from different observation points were selected to use for cell number counting. The cells were counted using Cell Count software (Heidelberg Engineering GmbH) in manual mode. The data are expressed as density (cells/mm 2 ) ± standard deviation.
Statistical Analysis
Statistical analysis was performed using SPSS 11.5 for Windows (Chicago, Illinois, USA). The independent-sample t test was used to compare the age difference between the 2 groups. The Mann-Whitney test was used to compare the means in cell density between the 2 groups. The Kruskal-Wallis test was performed to compare the dendritic cell densities of the cornea and the head and body of the pterygium. A P value of less than .05 was considered statistically significant.
Results
The pterygium group included 8 male and 18 female subjects with a mean age of 60.9 ± 9.9 years. The average pterygium length was 3.5 ± 1.6 mm, with an average width of 5.9 ± 1.6 mm. The control group included 7 male and 10 female subjects with a mean age of 56.2 ± 13.2 years. The mean age of the 2 groups was not significantly different ( P = .216).
In Vivo Laser Scanning Confocal Microscopy Images of Pterygium
In vivo LSCM images of the pterygia demonstrated tightly arranged epithelial cells with dark-appearing borders, light-gray bodies, and punctiform hyper-reflective nuclei. Inflammatory cells, primarily dendritic cells, were lightly to heavily distributed throughout the epithelium of the pterygium in most patients. Goblet cells, appearing in groups as large hyper-reflective oval-shaped cells, were also observed in the epithelium of the pterygium. Microcysts with hyper-reflective contents and hypo-reflective round borders were also found occasionally in the epithelial layer or sub-epithelium. The pterygium stroma had a dense fibrovascular structure with hyper-reflective blood cells and minimal infiltration of inflammatory cells.
In Vivo Laser Scanning Confocal Microscopy Images of Clear Cornea Adjacent to the Pterygium
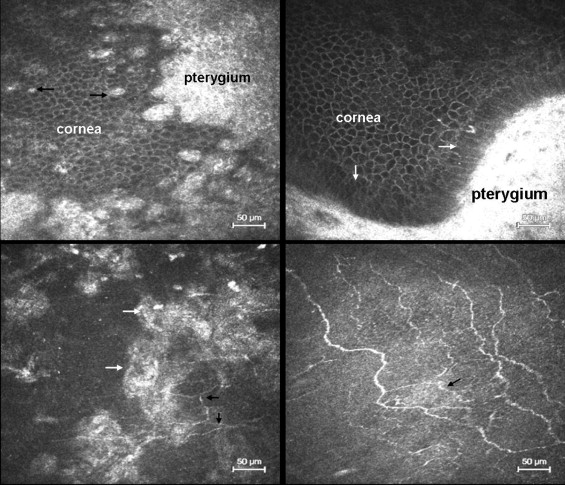
Basal corneal epithelial cells appeared as tightly packed cells with bright borders and dark cytoplasm. They were present in a uniform density of 5359.0 ± 543.1 cells/mm 2 in the clear cornea adjacent to the pterygium and 6463.1 ± 960.0 cells/mm 2 in the central cornea of healthy controls. The basal epithelial cell density in the control group was significantly higher than that in the pterygium group ( P < .001). At the pterygium-cornea junction, hyper-reflective epithelial cells of the pterygium were found scattered within the basal corneal cell layer ( Figure 2 , Top left), and the corneal basal cells located at the border were in some cases arranged in a columnar fashion ( Figure 2 , Top right). Hyper-reflective deposits were sporadically found at the level of the Bowman membrane and the anterior stroma ( Figure 2 , Bottom left). The keratocyte density in the anterior stroma was 407.4 ± 188.7 cells/mm 2 in the pterygium patients, which was significantly lower than the density of 1032.5 ± 212.5 cells/mm 2 in the central cornea of the control group ( P < .001). The keratocyte density in the posterior stroma of pterygium patients and controls was 442.8 ± 57.8 cells/mm 2 and 516.7 ± 105.0 cells/mm 2 respectively. This difference was not statistically significant ( P = .111). Morphologic irregularities, including tortuosity, breakage, and localized bulge and loop formation, were commonly seen in the sub-basal nerve plexus of pterygium subjects ( Figure 2 , Bottom right).
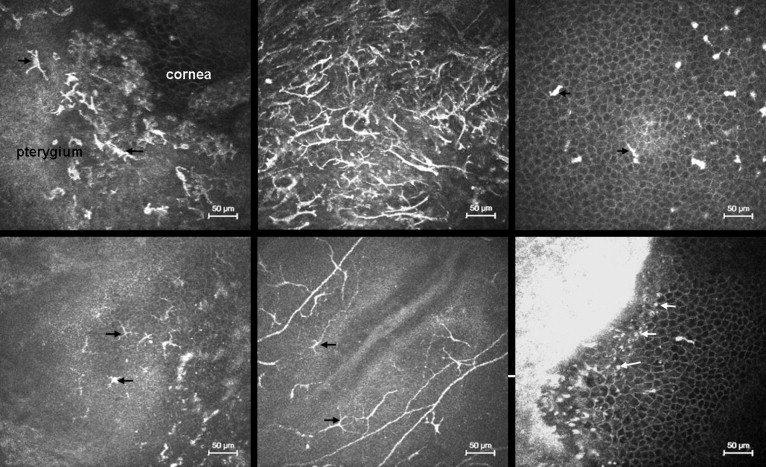
Distribution and Density of Dendritic Cells
In healthy subjects, dendritic cells were found in the central cornea of 52.9% of eyes (9/17), with a density of 23.6 ± 11.1 cells/mm 2 , and scattered dendritic cells were found in the nasal bulbar conjunctiva of 64.7% of eyes (11/17), with a density of 24.1 ± 17.6 cells/mm 2 . In the pterygium group, dendritic cells were found in the basal corneal epithelium in 57.7% of eyes (15/26), with a density of 60.3 ± 25.5 cells/mm 2 , which was significantly higher than the density in healthy controls ( P < .001). In the pterygium head, dendritic cells were noted in 61.5% of eyes (16/26), with a density of 89.4 ± 35.9 cells/mm 2 . In the pterygium body, dendritic cells were evident in 76.9% of eyes (20/26), with a density of 104.2 ± 44.5 cells/mm 2 . The dendritic cell density in the body of the pterygium was significantly higher than the density in the nasal bulbar conjunctiva of healthy controls ( P < .001). The dendritic cell density in the pterygium body and head was significantly higher than that in the basal corneal epithelium ( P < .05). The majority of the dendritic cells in the pterygium displayed the typical branching dendritic phenotype. In many cases, the dendritic cells were so numerous that they took on a wire net appearance ( Figure 3 , Top left and middle). In the clear cornea adjacent to the pterygium, most of the dendritic cells observed within the basal epithelial layer lacked dendrites ( Figure 3 , Top right). However, the dendritic cells observed in the sub-epithelium and sub-basal nerve plexus demonstrated an obvious dendritic appearance ( Figure 3 , Bottom left and middle). At the pterygium and cornea junction, presumed inflammatory cells were found infiltrating the corneal epithelium ( Figure 3 , Bottom right) and occasional inflammatory cells were also observed in the anterior stroma of the cornea.