Purpose
To investigate the influence of vitreomacular adhesion on the efficacy of pro re nata (PRN) ranibizumab monotherapy and verteporfin photodynamic therapy (PDT) combination therapy for neovascular age-related macular degeneration.
Design
Post hoc analysis of prospective randomized 12-month multicenter clinical trial data.
Methods
patient population : Total of 255 treatment-naïve patients with subfoveal choroidal neovascularization. observation procedure : Assessment of the vitreomacular interface on monthly optical coherence tomography with division of patients into the following categories according to continuous 1-year grading: posterior vitreous detachment (n = 154), dynamic release of vitreomacular adhesion (n = 32), stable vitreomacular adhesion (n = 51). main outcome measures : Mean best-corrected visual acuity (BCVA) letter and central retinal thickness changes at month 12 in the vitreomacular interface groups.
Results
Mean BCVA changes at month 12 were +3.5 (posterior vitreous detachment), +4.3 (release of vitreomacular adhesion), and +6.3 (vitreomacular adhesion) in patients receiving monotherapy ( P = .767), and +0.1 (posterior vitreous detachment), +6.6 (release of vitreomacular adhesion), and +9.2 (vitreomacular adhesion) in patients receiving combination therapy ( P = .009). Mean central retinal thickness changes were −113 μm (posterior vitreous detachment), −89 μm (release of vitreomacular adhesion), and −122 μm (vitreomacular adhesion) in monotherapy ( P = .725) and −121 μm (posterior vitreous detachment), −113 μm (release of vitreomacular adhesion), and −113 μm (vitreomacular adhesion) in combination therapy ( P = .924). Mean ranibizumab retreatments during 12 months were 4.9 (posterior vitreous detachment), 6.6 (release of vitreomacular adhesion), and 5.3 (vitreomacular adhesion) in monotherapy ( P = .018) and 4.7 (posterior vitreous detachment), 5.2 (release of vitreomacular adhesion), and 5.8 (vitreomacular adhesion) in combination therapy ( P = .942).
Conclusion
This study adds evidence that the vitreomacular interface status impacts functional outcomes and retreatment requirements. Patients with posterior vitreous detachment achieve acceptable results with fewer injections in PRN monotherapy, but lose potential vision gain with PDT. Patients with other vitreomacular interface configurations may potentially achieve optimized vision outcomes by combination of antiangiogenic treatment and vaso-occlusive PDT.
Intravitreal administration of anti–vascular endothelial growth factor (anti-VEGF) agents is the current first-line therapy in the management of neovascular age-related macular degeneration (AMD). Anti-VEGF agents effectively block the relevant signal cascade involved in the pathogenesis of choroidal neovascularization (CNV), leading to reduction of vascular leakage and restoration of visual acuity. However, at the individual patient level, the magnitude and durability of the anatomic and, particularly, functional response to anti-VEGF therapy are markedly heterogeneous, which makes individualized dosing recommendations difficult. Despite meta-analysis of the Comparison of AMD Treatment Trials (CATT) and Inhibition of VEGF in Age-related Choroidal Neovascularisation (IVAN) trials suggesting inferiority of discontinuous pro re nata (PRN) vs continuous monthly treatment, most clinicians aim to treat patients as little as possible, but as much as required to control the chronic and progressive disease course. Moreover, treatment results in real-life scenarios usually fail to reach the level of vision improvement reported from clinical trials. Although some microstructural characteristics that are predictive of treatment response have been identified, such as the presence of intraretinal cysts, the exact disease mechanisms responsible for individual response patterns are poorly understood. Therefore, identification of reliable morphologic parameters that could successfully guide individualized treatment represents an unmet medical need.
In this context, the condition of the vitreomacular interface is gaining scientific interest, while modern imaging strategies enhance the investigation of the vitreous itself. Typically, patients in the AMD age group show a complete posterior vitreous detachment, defined as separation of the posterior vitreous cortex from the retina and liquefaction as well as anteposition of the vitreous body. A minority of patients (20%–30%) present with persistent vitreomacular adhesion. As the vitreomacular interface configuration is a dynamic condition with potential transitions of one state to another, about 40% of patients with initial vitreomacular adhesion experience a release of vitreomacular adhesion and conversion to posterior vitreous detachment during several months of anti-VEGF treatment, although release may already occur after a single intravitreal injection.
Several pilot studies reported an influence of vitreomacular interface configuration on treatment efficacy in anti-VEGF therapy for neovascular AMD, with less favorable outcomes for patients with vitreomacular adhesion. The first large prospective study showed distinct response patterns of intravitreal ranibizumab in eyes with posterior vitreous detachment. Equivalence of monthly vs quarterly treatment was proven, as well as functional superiority of monthly vs quarterly treatment in patients with vitreomacular adhesion or dynamic vitreous release.
Apart from anti-VEGF therapy, the other approved therapeutic approach to neovascular AMD is verteporfin photodynamic therapy (PDT). PDT targets the neovascular lesion selectively via photochemical vaso-occlusion with minimal effect on neurosensory retina and adjacent structures, and it remains the treatment of choice for selected subtypes of CNV, such as polypoidal choroidal vasculopathy, which is exceedingly frequent in Asian populations, and CNV secondary to central serous chorioretinopathy. Since PDT itself triggers the release of VEGF and other pro-proliferative factors, the potential synergistic effect of PDT and anti-VEGF treatment in neovascular AMD provided the rationale for the large-scale SUMMIT clinical trial program evaluating combination of PDT with ranibizumab therapy. However, these studies showed no benefit of combination therapy in terms of vision outcome and inconclusive results regarding a potential reduction in treatment frequency. Nevertheless, recent studies reported a beneficial effect of PDT in recalcitrant cases of neovascular AMD, suggesting that—in addition to its role in the treatment of polypoidal choroidal vasculopathy–PDT may provide a useful therapeutic tool in selected AMD patients, and adding weight to the current concept of personalized therapy.
This analysis focuses on a study based exclusively on a PRN regimen and compares the impact of the vitreomacular interface condition on anti-VEGF monotherapy vs combination therapy with PDT, with the aim to evaluate visual outcome and retreatment frequency. A standardized analysis of prospective multicenter trial data was conducted by an independent central reading center. Monthly optical coherence tomography (OCT) examinations were analyzed and correlated with vision response and retreatment rate.
Methods
This post hoc analysis of prospective multicenter clinical trial data was conducted in compliance with the tenets of the Declaration of Helsinki and the International Conference on Harmonization of Good Clinical Practice guidelines. The ethics committee at the Medical University of Vienna, Austria, prospectively approved the current study. At each participating multicenter study site, prospective ethics committee or institutional review board approval was obtained. All patients provided written informed consent before enrollment into the trial. The study is registered at www.clinicaltrials.gov ( NCT00433017 ).
Monitoring and Treatment Protocol
All patients were participants of the MONT BLANC study, a randomized, single-masked multicenter phase II study in patients with primary subfoveal CNV secondary to AMD. Detailed information on trial design, inclusion and exclusion criteria, and primary as well as secondary outcomes have been published. In brief, MONT BLANC was designed to compare the efficacy of ranibizumab (Lucentis; Genentech Inc, South San Francisco, California, USA) monotherapy vs combination therapy of ranibizumab and PDT. Patients were randomly assigned to receive either PRN combination treatment regimen (verteporfin PDT 6 mg/m 2 and ranibizumab 0.5 mg) (Arm 1) or PRN ranibizumab monotherapy (sham infusion [5% dextrose] PDT and ranibizumab 0.5 mg) (Arm 2). On day 1, verteporfin or sham infusion was followed by laser application at standard fluence (wavelength, 689 nm; irradiance, 600 mW/cm 2 ) for 83 seconds (light dose, 50 J/cm 2 ). Intravitreal ranibizumab 0.5 mg (10 mg/mL) was administered a minimum of 1 hour after the start of verteporfin PDT. Two consecutive ranibizumab injections were performed at months 1 and 2. After this loading phase, verteporfin PDT and ranibizumab were administered according to predefined retreatment criteria at intervals of 90 and 30 days, respectively, as described in detail previously. Retreatment parameters included a 100 μm increase in OCT-determined central retinal thickness from the lowest previous value, presence of subretinal fluid or new hemorrhage, best-corrected visual acuity (BCVA) decrease of 5 or more letters, and leakage on fluorescein angiogram.
All patients underwent standardized monthly monitoring according to protocol, including measurement of BCVA by certified examiners according to the Early Treatment Diabetic Retinopathy Study protocol, slit-lamp examination, and fundus biomicroscopy. At each visit, eyes were imaged by certified examiners using Stratus OCT (Carl Zeiss Meditec, Dublin, California, USA) after pupil dilation and before the administration of treatment. The scanning protocol included the typical standard scanning modi for clinical trials at the Vienna Reading Center and consisted of 1 “6-mm cross-hair scan” (2 sections perpendicular to each other with a resolution of 512 A-scans per section) and 1 “fast macular thickness map scan” (6 6-mm radial sections with a resolution of 128 A-scans per section) at each visit.
Evaluation of the Vitreomacular Interface
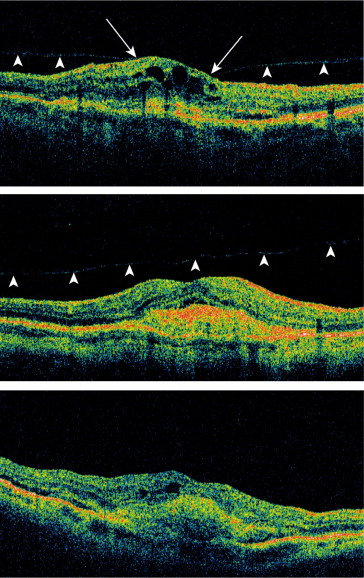
Analysis of the vitreomacular interface was performed on raw, masked OCT datasets at the Vienna Reading Center using a validated grading scheme as reported in detail previously. Briefly, trained and certified readers graded the vitreomacular interface configuration at each visit into 1 of the following states: (1) vitreous completely attached; (2) focal vitreomacular adhesion; (3) vitreous border antepositioned without contact to the macula; (4) vitreous border not visible. If vitreomacular traction was detected, the patient was excluded from further analysis since presence of vitreomacular traction was part of the MONT BLANC study exclusion criteria. The vitreomacular interface gradings from each visit were integrated after completion of the initial grading, and each patient was assigned to 1 of the following categories reflecting the vitreomacular interface configuration over the entire 12-month study period: (1) stable vitreomacular adhesion; (2) progressive release of vitreomacular adhesion; (3) posterior vitreous detachment. Representative grading examples for the 3 categories are provided in Figure 1 .
Statistical Analysis
Vitreomacular interface groups were compared by analysis of variance and χ 2 tests at baseline with respect to age, BCVA, central retinal thickness, intraretinal cysts, subretinal fluid, and pigment epithelial detachment, respectively. Further analyses were performed on differences to baseline. Differences after the loading phase and at the end of the first year were analyzed by 2-factor analysis of variance with vitreous groups and treatment groups as factors. Further differences were tested by linear contrasts. Corrections were made for multiple comparisons but not for multiple endpoints. P values less than .05 were considered statistically significant.
Results
Patient Disposition
Of the 255 patients included in the trial, 237 patients had complete vitreomacular interface data over 12 months available according to protocol. The ranibizumab monotherapy arm (n = 123) received a mean of 5.6 ranibizumab treatments following the loading dose. In the combination therapy arm (n = 114), a mean of 5.2 ranibizumab injections and 1.8 verteporfin PDT retreatments were administered during the PRN phase.
Vitreomacular Interface Characteristics
Posterior vitreous detachment was the most common vitreomacular interface configuration (n = 154, 65%), followed by vitreomacular adhesion (n = 51, 22%) and release of vitreomacular adhesion (n = 32, 13%). The distribution of vitreomacular interface configurations among the treatment arms as well as baseline characteristics were balanced as shown in Tables 1 and 2 .
| Posterior Vitreous Detachment N = 154 | Vitreomacular Adhesion N = 51 | Release of Vitreomacular Adhesion N = 32 | Total N = 237 | |
|---|---|---|---|---|
| Monotherapy, % (n) | 64.2 (79) | 21.1 (26) | 14.6 (18) | 100 (123) |
| Combination therapy, % (n) | 65.8 (75) | 21.9 (25) | 12.3 (14) | 100 (114) |
| Posterior Vitreous Detachment | Vitreomacular Adhesion | Release of Vitreomacular Adhesion | P | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Combination Therapy | Monotherapy | P | Combination Therapy | Monotherapy | P | Combination Therapy | Monotherapy | P | ||
| Age (mean ± SD) | 77 ± 7.3 | 72 ± 8.0 | 76 ± 6.3 | <.001 | ||||||
| 78 ± 6.8 | 76 ± 7.7 | .103 | 71 ± 9.0 | 73 ± 6.9 | .474 | 77 ± 5.6 | 75 ± 6.9 | .506 | ||
| Best-corrected visual acuity (mean ± SD) | 55 ± 12.8 | 53 ± 10.8 | 57 ± 12.5 | .494 | ||||||
| 55 ± 12.6 | 54±13.0 | .592 | 54 ± 12.4 | 52 ± 9.1 | .570 | 53 ± 15.7 | 59 ± 9.1 | .216 | ||
| Central retinal thickness (mean ± SD) | 338 ± 119.8 | 344 ± 102.2 | 334 ± 103.3 | .915 | ||||||
| 337 ± 100.5 | 339 ± 136.6 | .636 | 325 ± 86.7 | 363 ± 114.3 | .244 | 334 ± 79.2 | 333 ± 120.1 | .575 | ||
| Intraretinal cysts (%) | 53.9 | 56.9 | 40.6 | .314 | ||||||
| 53.3 | 54.4 | 1.000 | 48.0 | 65.4 | .264 | 35.7 | 44.4 | .725 | ||
| Subretinal fluid (%) | 57.8 | 68.6 | 75.0 | .111 | ||||||
| 64.0 | 51.9 | .144 | 76.0 | 61.5 | .368 | 71.4 | 77.8 | .703 | ||
| Pigment epithelial detachment (%) | 59.1 | 64.7 | 68.8 | .520 | ||||||
| 57.3 | 60.8 | .744 | 68.0 | 61.5 | .771 | 57.1 | 77.8 | .267 | ||
Vision Outcomes by Vitreomacular Interface Configuration
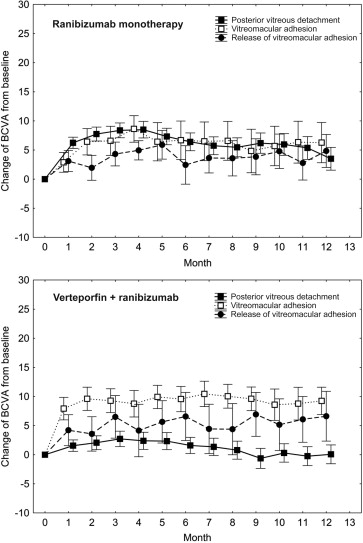
In the ranibizumab monotherapy arm, mean BCVA gains from baseline to the end of the loading phase at month 3 were +8.4 for posterior vitreous detachment, +4.7 for release of vitreomacular adhesion, and +6.6 for vitreomacular adhesion ( P = .420). During PRN maintenance therapy from month 3 until month 12, mean letter changes were −5.0 for posterior vitreous detachment, −0.4 for release of vitreomacular adhesion, and +0.6 for vitreomacular adhesion ( P = .070). In the ranibizumab plus verteporfin combination arm, mean letter gains at the end of the loading phase were +2.7 for posterior vitreous detachment, +6.5 for release of vitreomacular adhesion, and +9.3 for vitreomacular adhesion, showing superior outcomes for vitreomacular adhesion and release of vitreomacular adhesion with statistical significance at P = .042. This difference was maintained during PRN maintenance until month 12, with mean changes of −3.1 letters for posterior vitreous detachment, +0.8 for release of vitreomacular adhesion, and +0.0 for vitreomacular adhesion ( P = .212). Figure 2 shows BCVA results over time, comparing the vitreomacular interface groups in the monotherapy and combination therapy arms.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


