Purpose
To investigate the short-term effects of intravitreal ranibizumab on retinal vessel functionality in patients with retinal vein occlusion (RVO).
Design
Prospective, interventional case series.
Methods
We enrolled 11 eyes of 11 consecutive treatment-naïve patients with macular edema secondary to RVO. All patients underwent a complete ophthalmic evaluation, including optical coherence tomography and dynamic and static retinal vessel analysis, using the Dynamic Vessel Analyzer (Imedos, Jena, Germany) before (baseline) and 1 week and 1 month after administration of intravitreal ranibizumab. Investigations of RVO patients were compared to 11 eyes of age- and sex-matched control subjects.
Results
In RVO patients, dynamic analysis showed a significant increase of mean venous dilation from +2.46% ± 1.03% at baseline to +3.96% ± 1.3% at 1 week ( P = .001). At 1 week mean maximum venous and arterial dilations did not differ from those of control subjects. Static analysis showed a mean overall significant decrease of central retinal artery equivalent and central retinal vein equivalent from baseline to 1 week (from 174.8 ± 22.5 measurement units [MU] to 167.2 ± 26.7 MU [ P = .04], and from 228.4 ± 20.7 MU to 217.3 ± 22.8 [ P = .0002]). Mean central retinal artery equivalent in healthy control subjects was 175.9 ± 10.45 MU, not significantly different from baseline, week 1, and month 1 of RVO eyes. Conversely, mean central retinal vein equivalent was 195.5 ± 9.91 MU in healthy control subjects, significantly different from baseline, week 1, and month 1 of RVO eyes.
Conclusions
Using the Dynamic Vessel Analyzer in patients with RVO, we found that intravitreal ranibizumab increased vein dilation (dynamic analysis) and had a vasoconstrictive effect on both arteries and veins (static analysis).
Retinal vein occlusion (RVO) is the second most prevalent blinding vascular retinal disorder. The pathogenesis of RVO is not completely known. The main types of RVO are central RVO (CRVO) and branch RVO (BRVO), depending on the site of obstruction. One of the most important sight-threatening complications of RVO is macular edema (ME). This is probably the result of increased vascular pressure behind the occlusion that may lead to leakage of fluid across the vascular wall to the adjacent retinal tissue. Furthermore, the damage of the endothelium in the affected vein may induce a low-grade, chronic inflammation of the retinal microvasculature and an upregulation of inflammatory mediators that break the blood-retina barrier and perpetuate ME.
Vascular endothelial growth factor (VEGF) is a potent, diffusible, endothelial-specific mitogen that is released in response to hypoxia. VEGF acts by stimulating the angiogenesis and the vascular hyperpermeability of retinal capillaries, resulting in ME by disruptive effects on the endothelial zona occludens, induction of fenestrations in endothelial cells, fragmentation of endothelium, and degenerative changes in endothelial basement membranes.
As elevated intraocular levels of VEGF have been found in patients with RVO, there is a strong basis for the hypothesis that anti-VEGF agents may be beneficial in the treatment of vascular leakage and ME.
The comprehension of the pathogenesis of ME secondary to RVO has led to new therapies. Ranibizumab (Lucentis; Genentech, Inc, San Francisco, California, USA) is an anti-VEGF fragment that acts by neutralizing the biologic activity of all isoforms of VEGF. It is relevant to determine whether a variation in the retinal vessel diameter occurs after anti-VEGF injection because it has an effect directly on the endothelium.
Recent studies have reported that treatment with intravitreal injection of an anti-VEGF may change the ocular blood flow. Bonnin and associates measured a reduced velocity of the retrobulbar blood flow using color Doppler imaging in patients with age-related macular degeneration treated with intravitreal ranibizumab injection, with subsequent return to baseline levels suggesting that anti-VEGF treatment has a reversible effects on native retinal vascular supply after its discontinuation. Papadopoulou and associates reported that, in patients with age-related macular degeneration, the retinal arterioles constrict in response to intravitreal ranibizumab injection. So far, only 1 study examined the effects of intravitreal ranibizumab on the retinal circulation in patients with ME secondary to RVO. Sacu and associates demonstrated vasoconstriction in the veins and arteries of BRVO eyes measured by retinal vessel analyzer and a significant reduction in flow velocities in the retrobulbar central retinal artery measured using color Doppler imaging after intravitreal injections of ranibizumab.
Therefore, to improve our understanding of the vascular modification attributable to ranibizumab in patients with RVO, we performed an analysis of vascular functionality using the Dynamic Vessel Analyzer (Imedos, Jena, Germany), which allows the noninvasive evaluation of retinal vessels both at steady state (static) and upon stimulation (dynamic).
Methods
Study Participants
To perform this prospective interventional case series study, an informed consent was obtained from all subjects in agreement with the Declaration of Helsinki for research involving human subjects. The study was approved by the Ethics Committee of the Scientific Institute San Raffaele.
Consecutive treatment-naïve patients with single-eye RVO-related ME (study eye), presenting between January 7 and March 30, 2014, at the Retina Service of the Department of Ophthalmology of the University Scientific Institute San Raffaele in Milan, were enrolled in this prospective, interventional study. Criteria for inclusion were: (1) ME (central macular thickness [CMT] >300 μm, as measured by spectral-domain optical coherence tomography [SD OCT]) secondary to RVO; (2) age older than 18 years; and (3) best-corrected visual acuity (BCVA) between 20/200 and 20/25 (Snellen equivalent) in the study eye (to ensure reliability of the dynamic testing). The exclusion criteria were: (1) any ocular surgery in the study eye in the past 6 months; (2) previous laser photocoagulation; (3) previous intravitreal injection of corticosteroids or anti–VEGF; (4) diabetes mellitus with diabetic retinopathy; (5) history of ocular inflammation; (6) significant media opacities (to ensure proper execution of examination); and (7) uncontrolled systemic hypertension or other systemic diseases (in particular, we selected only patients with systolic blood pressure between 110 mm Hg and 140 mm Hg and diastolic blood pressure between 60 mm Hg and 90 mm Hg). During the period of patient recruitment, age- and sex-matched healthy control subjects were also recruited. Exclusion criteria were any eye pathology other than a need for glasses or contact lenses, and smoking.
Study Protocol
At baseline, patients with RVO underwent blood pressure and heart rate measurement; a complete ophthalmic evaluation, including assessment of distance BCVA using Early Treatment Diabetic Retinopathy Study charts, tonometry, slit-lamp biomicroscopy, indirect fundus ophthalmoscopy, and fluorescein angiography; SD OCT (Spectralis HRA+OCT; Heidelberg Engineering, Heidelberg, Germany) with automated CMT measurements (generated by using a 19-horizontal-line protocol, 6 × 6 mm area, each consisting of 1024 A-scans per line); customized high-resolution enhanced depth imaging (EDI) SD OCT scan; and dynamic and static analysis with the Dynamic Vessel Analyzer. The same day, all patients received 0.05 mL intravitreal ranibizumab (0.5 mg).
Follow-up examinations were performed at 1 week and 1 month and included distance BCVA, tonometry, slit-lamp biomicroscopy, indirect fundus ophthalmoscopy, SD OCT, EDI SD OCT, and Dynamic Vessel Analyzer examination. All patients, at each time point, also underwent repeated measurements of blood pressure and heart rate.
During the study period, healthy control subjects also underwent a complete ophthalmic evaluation, which included SD OCT and EDI SD OCT scans and Dynamic Vessel Analyzer examination, as well as blood pressure and heart rate measurement.
High-Resolution Enhanced Depth Imaging Spectral-Domain Optical Coherence Tomography Assessment
The method to obtain EDI OCT images has been previously described. A 9-mm high-resolution horizontal line scan through the fovea was performed. The line scan was saved for analysis after 100 frames were averaged, using the automatic averaging and eye tracking features of the proprietary device. The subfoveal choroidal thickness was obtained on the horizontal line scan: a single operator manually measured the distance between the hyporeflective line corresponding to the sclerochoroidal interface and the outer portion of the hyperreflective line corresponding to the retinal pigment epithelium.
Dynamic Vessel Analysis
The Dynamic Vessel Analyzer is a useful technique to evaluate retinal blood vessels, as it easily, noninvasively, and accurately measures the response of retinal vessels to flicker light. Flicker light stimulation enables the retina to regulate blood flow in response to neural activity. The increase of neural activity induced by flicker stimulation leads to retinal arterial and venous dilation because of the release of vasodilating factors, such as nitric oxide, from neural and endothelial cells.
Abstention from alcohol and caffeine-containing products was requested from participants in the 12 h period prior to the study. Measurements were performed at the same time point in the early afternoon hours before the injection (baseline) and after 1 week and 1 month. Topical tropicamide was used to obtain pupil dilation and examinations were conducted in a dimly lit room. Patients had to stay focused on the tip of a fixation bar during the test while the fundus was examined under green light with an average luminance of 130 cd/m 2 (ILT1700 Research Radiometer; International Light Technologies, Peabody, Massachusetts, USA). The Dynamic Vessel Analyzer creates a great contrast between retinal blood vessels and the adjacent tissue using a green light illumination (530–600 nm), while in contrast most surrounding tissue reflects light in this range. A charge-coupled device camera detects this reflected light and uses it to quantify the width of the vessels. The Dynamic Vessel Analyzer contains software to track the eye movements, so once a focus image of the fundus was obtained, a fixation target was located in a single zone (eg, a vessel branch) over the entire 30-degree visual field.
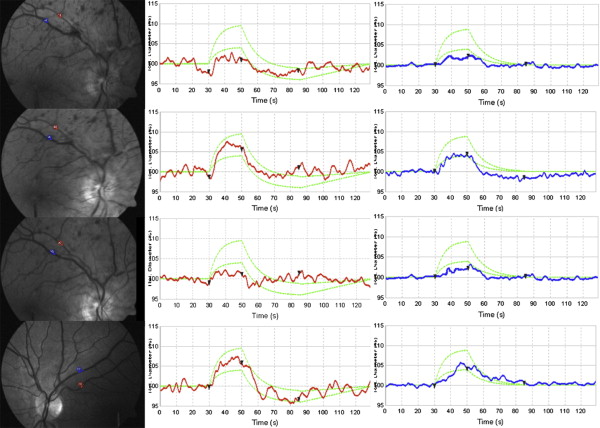
To perform the dynamic analysis in patients with CRVO and in healthy controls, a superior or inferior temporal venous and arterial segment located between one half and 2 disc diameters from the optic disc margin and at least 1 vessel diameter from any bifurcation or close vessel was chosen and marked manually with a probe (blue for the vein and red for the artery) ( Figure 1 ). The same procedure was performed in patients with BRVO; in this case the venous branch occluded with corresponding arterial segment was chosen. The complete examination had a duration of 350 s, which included 3 cycles of flicker/nonflicker light. The Dynamic Vessel Analyzer creates flicker with an optoelectronic shutter that interrupts the light source with a bright-to-dark ratio of 25:1 at a frequency of 12.5 Hz, to maximize vasodilation and blood flow during flicker. Selected vessel diameters were first recorded for 50 s, then a flicker stimulation was applied for 20 s (to allow vessel dilation), followed by a nonflicker period for 80 s; the sequence was repeated 3 times. Vessel diameters were calculated and expressed in measurement units (MU); vessel dilation was measured by calculating the percentage increase in vessel diameter relative to baseline after 20 s of flicker stimulation, and averaging the 3 measurement cycles.
Static Vessel Analysis
In each subject a 50-degree fundus photograph was taken using the FF450 retinal camera (Zeiss AG, Jena, Germany), contained in the Dynamic Vessel Analyzer system. VISUALIS and VesselMap Software (Imedos Systems, Ltd, Jena, Germany) allowed analysis of these photographs ( Figure 2 ). Principles of static vessel analysis have been described earlier.
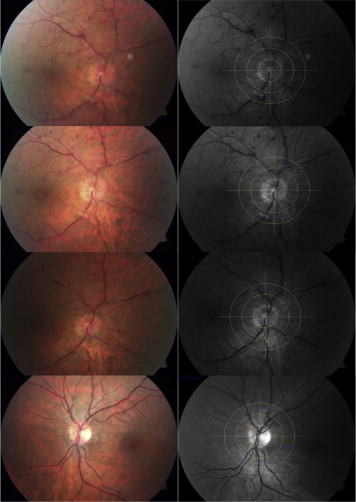
Briefly, using an optic disc–centered image, the papilla is marked and the software creates an area of one-half to one disc diameter from its center to measure all vessels. Arterial and venous vessels are differentiated and selected manually ( Figure 2 ).
In all subjects we calculated the central retinal artery equivalent, which relates to the diameter of the central retinal artery; the central retinal vein equivalent, which relates to the diameter of the central retinal vein; and the arteriovenous ratio, which represents the central retinal artery equivalent/central retinal vein equivalent ratio. In patients with RVO and in healthy controls we computed and compared the whole central retinal artery equivalent, central retinal vein equivalent, and arteriovenous ratio and the central retinal artery equivalent, central retinal vein equivalent, and arteriovenous ratio of each single (affected/occluded and unaffected/nonoccluded) quadrant (superior nasal, inferior nasal, superior temporal, and inferior temporal).
Statistical Analysis
Statistical calculations were performed with GraphPad Prism version 6.0 (GraphPad Software, San Diego, California, USA). All variables were tested for normal distributions, according to the Kolmogorov-Smirnov test. The data were summarized with the mean ± standard deviation. The effects of ranibizumab were assessed by 1-way analysis of variance followed by post hoc analysis performed using planned comparison with Bonferroni correction for multiple comparisons. Univariate linear regression models and Pearson correlation were used to study the relationship among variables.
The differences between RVO patients and healthy control subjects were analyzed using the nonparametric Mann-Whitney U test or Kruskal-Wallis test, as appropriate.
All tests were 2-sided and a P value less than .05 was considered significant.
Results
We included a total of 11 eyes of 11 patients with ME secondary to RVO (10 men, 1 woman; mean age 56.8 ± 13.3 years).The duration of symptoms before inclusion ranged between 1 and 6 months. Mean baseline BCVA and CMT of study eyes, including 6 eyes with CRVO and 5 eyes with BRVO, were 0.37 ± 0.15 logMAR and 485.7 ± 154.3 μm, respectively; mean intraocular pressure (IOP) was 14.7 ± 1.4 mm Hg.
Mean BCVA significantly increased to 0.21 ± 0.11 at 1 week ( P = .0001) and to 0.17 ± 0.12 at 1 month ( P < .0001). Mean IOP did not change at both week 1 and month 1 (14.8 ± 0.8 mm Hg, P = .9, and 14.4 ± 0.8 mm Hg, P = .9, respectively).
Mean CMT significantly decreased to 273.5 ± 47.94 μm at 1 week ( P < .0001) and to 255.3 ± 61.54 μm at 1 month ( P < .0001). The mean subfoveal choroidal thickness, as measured by EDI OCT, decreased from 292.2 ± 43.24 μm at baseline to 259.5 ± 48.07 μm at 1 week ( P = .1) and to 240.02 ± 55.9 μm at 1 month ( P = .009).
A total of 11 eyes of 11 age- and sex-matched control subjects (10 men, 1 woman; age 53.6. ± 10.7 years) met the inclusion criteria and were included for analysis. Mean BCVA was 0.00 logMAR, significantly different compared to baseline, week 1, and month 1 BCVA of RVO eyes ( P = .003, P = .005, and P = .008, respectively). Mean CMT was 218.1 ± 8.1 μm, significantly different compared to baseline, week 1, and month 1 CMT of RVO eyes ( P < .0001, P = .01, and P = .019, respectively). Mean IOP was 14.3 ± 1 mm Hg ( P = .4, P = .2, and P = .8, compared to, respectively, baseline, week 1, and month 1 IOP in RVO eyes). The mean subfoveal choroidal thickness, as measured by EDI OCT, was 235.4 ± 34.29 μm, significantly different from baseline, but not from week 1 and month 1 subfoveal choroidal thickness of RVO eyes ( P = .008, P = .2, and P = .8, respectively).
In RVO patients, dynamic vessel analysis of retinal veins showed a mean venous dilation change from +2.46% ± 1.03% at baseline to +3.96% ± 1.3% at 1 week ( P = .001) and to +2.46% ± 1.58% at 1 month ( P = .9) ( Table ). The retinal arteries showed mean dilation changes from +2.28% ± 1.39% at baseline to +3.55% ± 1.98% at 1 week ( P = .2), and to +2.41% ± 1.25% at 1 month ( P = .9) ( Table ). Four out of 11 eyes underwent repeated examination at 2 months (after subsequent treatment). These 4 eyes revealed a similar trend in mean vessel dilation change (data not shown). No difference was detected between CRVO and BRVO eyes (data not shown).
| Baseline a | 1 Week | 1 Month | Controls b | |
|---|---|---|---|---|
| Dilation Art, % | +2.19 ± 1.34 | +3.38 ± 2.05 | +2.41 ± 1.25 | +4.06 ± 1.42 |
| P | .01 b | .237 a .3 b | .9 a .008 b | |
| Dilation Ven, % | +2.46 ± 1.03 | +3.96 ± 1.3 | +2.46 ± 1.58 | +4.89 ± 1.98 |
| P | .002 b | .001 a .2 b | .9 a .004 b | |
| CRAE, MU | 174.8 ± 22.5 | 167.2 ± 26.7 | 169.4 ± 26.3 | 175.9 ± 10.4 |
| P | .8 b | .04 a .6 b | .2 a .8 b | |
| CRVE, MU | 228.4 ± 20.7 | 217.3 ± 22.8 | 215.8 ± 25.8 | 195.5 ± 9.91 |
| P | .0001 b | .0002 a .008 b | <.0001 a .016 b | |
| AVR | 0.76 ± 0.08 | 0.77 ± 0.1 | 0.78 ± 0.1 | 0.90 ± 0.02 |
| P | .0008 b | .9 a .004 b | .9 a .005 b | |
| CRAE occl, MU | 102.3 ± 19.7 | 92.7 ± 21.6 | 99.6 ± 20.1 | 106.4 ± 12.6 |
| P | .4 b | .003 a .006 b | .9 a .09 b | |
| CRVE occl, MU | 136.4 ± 24.5 | 123.5 ± 24.7 | 126.8 ± 24.3 | 120.5 ± 19.7 |
| P | .007 b | <.0001 a .5 b | .0002 a .2 b | |
| AVR occl | 0.76 ± 0.16 | 0.76 ± 0.18 | 0.78 ± 0.12 | 0.89 ± 0.14 |
| P | <.0001 b | .9 a <.0001 b | .9 a .0005 b | |
| CRAE nonoccl, MU | 101.8 ± 14.2 | 100 ± 15.6 | 103.2 ± 15 | 106.4 ± 12.6 |
| P | .4 b | .9 a .2 b | .9 a .7 b | |
| CRVE nonoccl, MU | 126.1 ± 12 | 123.2 ± 13.4 | 124.1 ± 10.9 | 120.5 ± 19.7 |
| P | .09 b | .4 a .3 b | .9 a .1 b | |
| AVR nonoccl | 0.81±0.15 | 0.82 ± 0.17 | 0.83 ± 0.14 | 0.89 ± 0.14 |
| P | 0.1 b | .9 a .15 b | .9 a .4 b |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


