Purpose
To compare outcomes of illuminated microcatheter–assisted circumferential trabeculotomy vs combined mitomycin C–augmented trabeculotomy-trabeculectomy for primary congenital glaucoma.
Design
Prospective, randomized trial.
Methods
Of the 30 patients (60 eyes) analyzed with bilateral primary congenital glaucoma aged ≤2 years, 1 eye of each patient was randomized to: illuminated microcatheter–assisted trabeculotomy (Group I) or combined trabeculotomy with trabeculectomy augmented with mitomycin C (Group II). Primary outcome measure was intraocular pressure (IOP) reduction. Categorization into absolute success (IOP ≤15 mm Hg without medication) and qualified success (IOP ≤15 mm Hg with medication) was done. Secondary outcomes included change in corneal diameter and clarity, optic disc status, refraction, need for anti-glaucoma therapy, and occurrence of complications.
Results
Mean age of patients was 6.63 ± 5.74 months. IOP fell by 49.3% (22.25 ± 10.88 to 11.56 ± 3.33 mm Hg) in Group I and 46.6% in Group II (21.73 ± 8.89 to 11.60 ± 3.03 mm Hg) ( P < .001 in both). At 1 year, absolute success was achieved in 86.7% (26/30) and 90.0% (27/30) in Groups I and II, respectively ( P >.99) and qualified success in 93.3% (28/30) in both groups ( P = 1). There was significant improvement in corneal clarity ( P < .001) and cup-to-disc ratio ( P ≤ .01) in both groups at 1 year. Though incidence of hyphema was significantly more in Group I ( P = .0001), no vision-threatening complications occurred in either group.
Conclusions
Illuminated microcatheter–assisted circumferential trabeculotomy achieved comparable surgical outcomes to mitomycin C–augmented combined trabeculotomy-trabeculectomy and may be recommended as the initial surgical procedure for primary congenital glaucoma.
The principal pathology in primary congenital glaucoma is an abnormal development of the anterior chamber angle leading to an obstruction to the aqueous outflow. Surgery remains the definitive treatment in these cases. Angle surgeries like goniotomy and trabeculotomy target restoration of the physiological outflow by removing this obstruction. However, many patients present with corneal changes that preclude proper visualization of angle structures, and hence goniotomy is often not a feasible option in such cases. On the contrary, ab externo trabeculotomy can be performed in hazy corneas. However, goniotomy and conventional trabeculotomy treat the angle partially and may have to be attempted a number of times to achieve the desired intraocular pressure (IOP) control.
Some studies have shown better IOP-lowering effects of combined trabeculotomy-trabeculectomy over either procedure alone, thus making it the preferred procedure for management of primary congenital glaucoma. According to current consensus, standard filtering surgery done for congenital and developmental glaucoma involves the use of mitomycin C. This is because children demonstrate a more exuberant fibrotic reaction in response to surgical trauma when compared to adults, which leads to early bleb failure. However, the long-term complications associated with the use of mitomycin C and the presence of a filtering bleb can sometimes be sight threatening.
To overcome the limitations of conventional surgeries, circumferential (360-degree) trabeculotomy was described, initially using a suture. However, this procedure had a risk of misdirection of the leading end of suture into the subretinal or suprachoroidal space. Illuminated microcatheter–assisted circumferential trabeculotomy has the success potential similar to suture trabeculotomy with an added advantage of trans-scleral visualization, which minimizes the risk of false passage. Previously published retrospective studies on illuminated microcatheter–assisted trabeculotomy in congenital glaucoma have shown high success rates with encouraging results. However, there are no prospective studies comparing illuminated microcatheter–assisted trabeculotomy with combined mitomycin C–augmented trabeculectomy-trabeculotomy. The purpose of this study was to compare the safety and efficacy of illuminated microcatheter–assisted circumferential trabeculotomy with mitomycin C–augmented combined trabeculotomy-trabeculectomy in the treatment of primary congenital glaucoma.
Subjects and Methods
Study Design
A prospective randomized controlled trial was conducted at the Glaucoma Services of Dr Rajendra Prasad Centre for Ophthalmic Sciences, All India Institute of Medical Sciences (AIIMS), New Delhi, a tertiary eye care center at a university hospital. Eligible patients were recruited in the study between August 2012 and May 2013. A written informed consent was obtained from the parents of eligible subjects after thoroughly explaining the risks and benefits of each procedure. The study included patients with bilateral primary congenital glaucoma aged ≤2 years, after confirming the disease and its baseline severity on an examination under anesthesia (EUA). Those patients who had unilateral disease, secondary glaucoma, previously operated eyes, and parents not willing for consent and follow-up were excluded from the study. The study was approved by the Institute Ethics Committee (Ref. no. IESC/T-268/29.06.2012) and was registered under the Clinical Trials Registry–India (CTRI/2014/05/004603). The trial conformed to the tenets of Declaration of Helsinki.
A random number table was generated using nQuery Advisor + nTerim 2.0 software (Science Plus Group, Groningen, The Netherlands). Each eye of the subjects was randomized to 1 of the 2 procedures, wherein 1 eye (right/left) underwent illuminated microcatheter–assisted circumferential trabeculotomy (Group I) while the other eye received combined trabeculotomy-trabeculectomy augmented with mitomycin C (Group II), decided upon by numbering obtained from the random number table.
Examination
A detailed history with emphasis on family history was elicited for all patients by a single nonmasked observer. Recruited patients underwent a detailed preoperative evaluation on EUA to record the baseline characteristics. EUA was performed under sevoflurane anesthesia using spontaneous ventilation. Intraocular pressure was measured in the early phase of anesthesia after induction using a hand-held Perkins applanation tonometer (Perkins Tonometer MK2; Clement Clarke Ophthalmic, Harlow, UK). In cases of corneal edema/opacity precluding applanation tonometry owing to distorted mires, IOP was measured using Tonopen Avia (Reichert Technologies, Depew, New York, USA). Gonioscopy was performed with a Koeppe goniolens if the corneal clarity was adequate enough to visualize the angle structures under the operating microscope. Horizontal white-to-white corneal diameter was measured using Castroviejo calipers. Corneal clarity was divided into 3 grades. Grade 1 was visibility of iris details, grade 2 was visibility of iris without clarity of details, and grade 3 was nonvisibility of iris. Fundus examination for optic disc status (measurement of vertical cup-to-disc ratio) was done using direct or indirect ophthalmoscopy (using a 20 diopter [D] lens) as permitted by the media clarity. In the event of nonvisibility of fundus, ultrasound B-scan was performed to rule out any intraocular pathology and to detect excavation of the optic nerve head.
Surgical Procedure
Surgery was performed under general anesthesia by a single nonmasked surgeon (T.D.). In 1 eye of each patient, as per randomization, circumferential trabeculotomy was performed using an illuminated microcatheter (Glaucolight; DORC International, Zuidland, The Netherlands) (Group I). Glaucolight is a fiberoptic-based device with an external diameter of 150 microns (40 Gauge). It has an atraumatic tip with flexibility for 360-degree catheterization and an LED light source for trans-scleral visualization to minimize chances of false passage.
The illuminated microcatheter–assisted trabeculotomy was performed under strict aseptic precautions. A corneal stay suture was applied using spatulated 8-0 polyglactin. A fornix-based localized peritomy was performed in the superior quadrant (12 o’clock) and hemostasis was achieved using wet field cautery. A 4 × 4-mm superficial scleral flap was raised followed by creation of a deep scleral flap to identify and deroof the Schlemm canal. After injection of viscoelastic at the cut end of the Schlemm canal, a microcatheter was introduced and advanced slowly within the Schlemm canal with direct trans-scleral visualization of the microcatheter tip. The catheter was retrieved from the other cut end of the Schlemm canal in case of successful 360-degree catheterization. Paracentesis was performed and air was injected to maintain the anterior chamber. Circumferential trabeculotomy was performed by pulling the 2 ends of the catheter like a purse string. In cases of incomplete catheterization owing either to tip obstruction or to misdirection, either a cohesive viscoelastic (Healon OVD; Abbott Medical Optics Inc, California, USA) was injected into the canal to dilate the canal and an attempt was made to pass the catheter from the other end. If 360-degree passage was not possible, partial trabeculotomy (conventional ab externo trabeculotomy) was performed using Harms trabeculotome. Scleral flaps were closed using 10-0 monofilament nylon suture and peritomy was closed by continuous interlocking 8-0 round-bodied absorbable polyglactin sutures.
In the same sitting, combined trabeculotomy-trabeculectomy with mitomycin C (Group II) was performed for the other eye under strict aseptic precautions with another sterile set of instruments. A corneal stay suture was passed using 8-0 polyglactin suture. A superior-quadrant (12 o’clock) fornix-based localized peritomy was similarly fashioned and hemostasis was achieved using wet field cautery. Subconjunctival mitomycin C (0.4 mg/mL) was applied for 3 minutes followed by thorough saline wash. A 4 × 4-mm superficial scleral flap was raised. Under high magnification, a radial incision was given starting from the blue zone up to the white zone until aqueous was seen to ooze out from the cut ends of the Schlemm canal. The Harms trabeculotome was then rotated through each end of the canal to perform a manual trabeculotomy. Paracentesis was performed and air was instilled in the anterior chamber. Trabeculectomy was performed by creating a full-thickness internal ostium in the corneoscleral tissue anterior to the site of the trabeculotomy followed by a broad-based iridectomy to prevent blockade of the ostium by iris tissue. The scleral flap was closed using one 10-0 monofilament nylon suture. The peritomy was closed by continuous interlocking 8-0 absorbable polyglactin sutures. A standard postoperative regimen was prescribed in both groups, which included topical cycloplegics along with steroid-antibiotic drops in tapering doses for 8 weeks.
Postoperative Follow-up
Postoperative follow-up was performed at day 1 and day 7 to observe for anterior chamber shallowing/hyphema or any other inadvertent events. EUAs were performed at 1 month, 3 months, 6 months, and 1 year as per institutional standard protocol. Postoperative EUAs included measurement of IOP, corneal diameters, corneal clarity, lens clarity, optic disc status, and refractive error. If a patient’s IOP was found to be high, appropriate treatment adjustments were made by adding anti-glaucoma medications and closer monitoring for IOP was done.
Outcome Parameters
The primary outcome measure was IOP control. Absolute success was defined as an IOP ≤15 mm Hg without the need for topical anti-glaucoma medications and qualified success was defined as an IOP ≤15 mm Hg with the use of topical anti-glaucoma medications at the end of 1 year follow-up post surgery.
Secondary outcome measures were change in corneal diameter, corneal clarity, optic disc status, refraction, and the need for topical anti-glaucoma medication to control intraocular pressure. The mean refractive error was measured in spherical equivalent (SEQ). Intraoperative and immediate postoperative complications such as hyphema, iris manipulation, iris/lenticular damage, and microcatheter-related problems such as obstruction and misdirection were evaluated. Late postoperative complications such as hypotony (IOP <6 mm Hg), bleb complications (leak, infections, fibrosis), and failure (ie, need for re-surgery) were also evaluated.
Statistical Analysis
Statistical analysis was done using SPSS version 15 software (SPSS Inc, Chicago, Illinois, USA). Quantitative data were expressed as mean ± standard deviation and qualitative data were expressed as frequency distribution and their percentage. The statistical significance of quantitative variables was determined by independent t test/paired t test for parametric variables and Wilcoxon signed rank test/Mann-Whitney U test for nonparametric variables. For comparing the statistical significance of categorical variables between 2 groups, χ 2 test/Fisher exact test was applied. The level of statistical significance was taken as P < .05.
Results
Demography
Forty-seven patients with congenital glaucoma were initially screened for inclusion in the study. Of these, parents of 2 patients declined participation and 12 patients did not meet the inclusion criteria. Of the 33 patients who underwent surgery, 3 patients were lost to follow-up after the initial 1 week. Hence, data analysis was done for 60 eyes of 30 patients with bilateral primary congenital glaucoma who underwent illuminated microcatheter–assisted trabeculotomy in 1 eye and combined trabeculotomy-trabeculectomy with mitomycin C in the other eye as per the randomization.
The mean age of the patients was 6.63 ± 5.74 months (range: 8 days – 23 months). Twenty-six patients (86.7%) were infants (<1 year), and 20 patients (67%) were less than 6 months old. Twenty-two patients (73%) were male and 8 patients (27%) were female. Family history of glaucoma in siblings was elicited in 2 patients (6.7%). Corneal cloudiness was the main presenting complaint in 43.3% (13/30) of patients, followed by enlarged eyeball in 33.3% (10/30), excessive tearing and sensitivity to light in 16.7% (5/30), and eye rubbing in 6.7% (2/30). The presenting IOP of subjects was 21.80 ± 9.76 mm Hg and 21.73 ± 8.89 mm Hg in Groups I and II, respectively ( P = 0.84). Baseline parameters (IOP, corneal clarity, corneal diameters, vertical cup-to-disc ratio, refractive error) of both the groups were statistically comparable. All patients were being treated with ocular hypotensive medications preoperatively. A summary of the demographic data and baseline parameters is given in Table 1 .
| Parameters | Group I a Circumferential Trabeculotomy With Illuminated Microcatheter (N = 30 Eyes) | Group II a Combined Trabeculotomy-Trabeculectomy (N = 30 Eyes) | P Value |
|---|---|---|---|
| Age | 6.63 ± 5.74 months (range: 8 days to 23 months) | – | |
| Sex | Male – 22 patients (73%) Female – 8 patients (27%) Male: Female = 3:1 | – | |
| Family history of glaucoma in siblings | 2 patients (6.67%) | – | |
| IOP (mm Hg) | 21.80 ± 9.76 | 21.73 ± 8.89 | .84 |
| Corneal clarity | |||
| Grade 1 | 10 eyes | 12 eyes | .63 |
| Grade 2 | 12 eyes | 13 eyes | |
| Grade 3 | 8 eyes | 5 eyes | |
| Horizontal corneal diameter (in mm) | 13.22 ± 1.28 | 13.18 ± 1.24 | .92 |
| Vertical cup-to-disc ratio | 0.74 ± 0.15 (n = 10) | 0.78 ± 0.13 (n = 12) | .50 |
| Refractive error in SEQ | −3.60 ± 1.99 D (n = 10) | −3.50 ± 1.63 D (n = 12) | .90 |
a Group I: eyes that underwent illuminated microcatheter–assisted circumferential trabeculotomy; Group II: eyes that underwent combined trabeculotomy-trabeculectomy with mitomycin C.
Intraoperative and Immediate Postoperative Course
In Group I, complete catheterization was achieved in 24 eyes (80%) and partial catheterization in 6 eyes (20%) owing to catheter obstruction or misdirection requiring conversion into conventional trabeculotomy. Iris prolapse occurred in 1 eye in Group I and was managed by gentle repositioning of the iris, whereas iris manipulation was required in all cases of Group II during peripheral iridectomy. In Group I, mild and transient hyphema was noted in 28 eyes and total hyphema in 2 eyes, of which 1 eye required hyphema drainage. Transient hyphema was noted in 16 eyes of Group II and resolved spontaneously within 3-5 days postoperatively. The incidence of hyphema was significantly greater in Group I compared to Group II ( P = .0001). Shallow anterior chamber was not encountered in any of the eyes in Group I, whereas 2 eyes in Group II developed shallowing of the anterior chamber postoperatively and were managed conservatively. In 1 eye of Group I, an illuminated microcatheter encircled the Schlemm canal for the second time without exiting through the superior canal cut-down. A small cut-down of the Schlemm canal at the advancing tip helped retrieve the catheter.
Success Rate
There was a significant reduction of intraocular pressure at all follow-ups in both the groups ( P < .005 at 1 month, P < .001 at 3, 6, and 12 months), with no significant difference in the achieved IOP between the 2 groups ( Figure ). At the end of 1 year, IOP reduction was 46.92% and 46.61% in Group I and Group II, respectively ( P < .001) ( Table 2 ). Of the eyes that underwent complete catheterization (n = 24), IOP fell by 49.25% (from 22.25 ± 10.88 mm Hg preoperatively to 11.29 ± 2.38 mm Hg at 12 months; P < .001). At follow-up, 4 eyes in Group I (1 among those that underwent complete catheterization and 3 eyes among those with partial catheterization) and 3 eyes of Group II (2 eyes at 1 month and 1 eye at 3 months) required medications to control intraocular pressure ( P > .99). Three of 6 eyes among those that had undergone partial catheterization required medications at 1 month, which was significantly more than those that had undergone complete catheterization (1/24) ( P = .018; Fisher exact test). None of these patients required repeat filtering surgery at 1 year. Patients requiring medications were started initially on topical timolol drops and topical dorzolamide was added, if required to control the intraocular pressure.
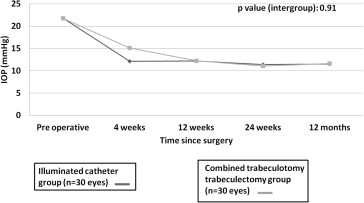
| IOP (in mm Hg) | Group I a Circumferential Microcatheter Trabeculotomy (N = 30 Eyes) | Group II a Combined Trabeculotomy-Trabeculectomy (N = 30 Eyes) | P Value |
|---|---|---|---|
| Baseline | 21.80 ± 9.76 | 21.73 ± 8.89 | .84 |
| 1 month | 12.13 ± 4.10 | 15.13 ± 7.86 | .07 |
| 3 months | 12.27 ± 0.85 | 12.26 ± 4.68 | .99 |
| 6 months | 11.43 ± 2.24 | 11.06 ± 2.27 | .53 |
| 12 months | 11.56 ± 3.33 | 11.60 ± 3.03 | .96 |
a Group I: eyes that underwent illuminated microcatheter–assisted circumferential trabeculotomy; Group II: eyes that underwent combined trabeculotomy-trabeculectomy with mitomycin C.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


