Purpose
To compare Icare ONE tonometry by clinic examiner and parent/guardian to Goldmann applanation in children with known/suspected glaucoma; to evaluate the trend in intraocular pressure (IOP) with 4 repeated measurements using Icare ONE; and to evaluate the feasibility of instructing parents on the use of the Icare ONE device in the clinic setting.
Design
Nonrandomized, prospective clinical study.
Methods
Patients with known or suspected glaucoma were recruited from the Duke pediatric glaucoma clinic. Parent(s) of all subjects gave informed consent (and children gave assent) for participation in this research study. IOP was measured using Icare ONE by clinic examiner and parent/guardian, then using Goldmann applanation (masked physician). Each parent/guardian completed an ease-of-use survey.
Results
Sixty eyes (60 children) were included. Absolute value of mean IOP difference (ICare ONE clinic examiner vs Goldmann applanation) was 3.3 ± 4.0 mm Hg ( P = .001). Icare ONE IOP by clinic examiner was within 3 mm Hg of Goldmann applanation in 68% (n = 41 eyes). In eyes with >3 mm Hg difference, Icare ONE was higher than Goldmann applanation in 84%. IOP demonstrated a statistically significant downward trend with repeated sequential measurements with Icare ONE ( P = .0053, r 2 = 0.9894). All parents accomplished Icare ONE tonometry on at least 1 eye; 98% reported it was “easy to learn to use.”
Conclusion
Icare ONE tonometry appears accurate and well-tolerated compared to Goldmann applanation, and holds promise for clinic and home tonometry in children. IOP trends downward with successive measurements using Icare ONE, demonstrating a possible effect from presumed patient relaxation.
Accurate measurement of intraocular pressure (IOP) poses challenges in the diagnosis and management of children with known and suspected glaucoma. Measuring IOP with conventional tonometers using anesthetic drops often engenders an anxiety response in children; this response, when associated with breath-holding, eyelid squeezing, and Valsalva, can result in elevated venous pressure and falsely elevated IOP measurements.
The Icare rebound tonometer (Icare Finland, Helsinki, Finland), a portable device that does not require an anesthetic, is a particularly attractive option for the pediatric population. The original Icare device is commercially available in the United States and has been shown to be well tolerated and to give reproducible results in the pediatric population. Flemmons and associates demonstrated good correlation between Icare and Goldmann applanation tonometry in pediatric patients with known and suspected glaucoma, and showed that parents are able to use the device for home tonometry in children.
The Icare ONE, a newer device intended for home tonometry in adults, is in commercial use in Europe, but is not yet FDA approved or commercially available in the United States. The Icare ONE, with its computer link, small size, and relatively low proposed cost, may have advantages over the original Icare for tonometry use in children. A recent study demonstrated good correlation of Icare ONE to Goldmann applanation tonometry in the adult population. To date, we are aware of no published studies assessing the feasibility or accuracy of Icare ONE compared to Goldmann applanation tonometry in the pediatric population (PubMed search terms: rebound tonometry and pediatric glaucoma or childhood glaucoma, Icare and pediatric glaucoma or childhood glaucoma).
The present study compares the Icare ONE vs Goldmann applanation tonometry in children with known or suspected glaucoma. In addition, the present study evaluates the trend in IOP recorded with repeated, sequential measurements using Icare ONE in a given child’s eye. Finally, the study evaluates the feasibility of instructing parents on the use of the Icare ONE device in the clinic setting.
Methods
This prospective clinical study was IRB-approved, and written informed consent was obtained from parent(s) for all enrolled subjects. Written assent was also obtained from subjects 12 years or older. Children were recruited from routine clinical visits at the Duke pediatric glaucoma clinic between August 1, 2010 and April 15, 2011. Recruited subjects had known or suspected glaucoma in at least 1 eye. All subjects were able to cooperate with Goldmann applanation tonometry and all had a parent who was willing and able to attempt Icare ONE tonometry at the clinic visit.
Demographic information collected from the clinic chart included patient age, sex, visual acuity, glaucoma diagnosis, secondary ocular diagnoses, number of prior glaucoma surgeries, number of glaucoma medicines, central corneal thickness (CCT) (if measured within the last year), and presence of nystagmus, strabismus, or central corneal opacities.
Icare ONE was used on all subjects: Icare ONE is a handheld device measuring 11 cm × 8 cm and weighing 150 g ( Figure 1 ). A magnetic solenoid probe rebounds off the cornea; the change in velocity is used to determine the IOP in mm Hg. The device acquires 6 IOP readings in rapid succession and computes an average value. If the standard deviation on this mean is considered low enough, a green light appears, signifying an acceptable IOP reading. Conversely, a red light indicates an unacceptably high standard deviation, requiring a repeat measurement. While the mean measured IOP is not shown, the panel indicates the interval into which the mean reading falls. For example, if the mean measured IOP were 16 mm Hg, the IOP would be displayed in the interval 14–18 ( Figure 2 ). The readings are saved by date and time, and may be downloaded to a computer using special software.
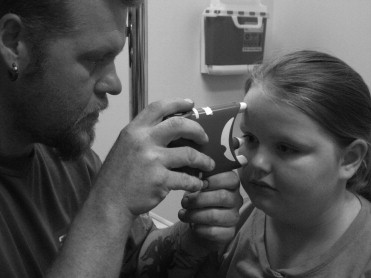

For each subject, 4 IOP measurements by Icare ONE were attempted on each eye: 2 were performed by a trained clinic examiner and 2 by the parent. Only reliable IOP measurements were recorded. The parents were instructed on the use of Icare ONE at the time of the clinic visit and were supervised by a clinic examiner at all times. In 1 group of subjects (Group 1, n = 33), the clinic examiner performed the first 2 IOP measurements and the parent performed the second 2 IOP measurements. In the second group of subjects (Group 2, n = 20), the clinic examiner and parent alternated (clinic examiner obtained the first and third measurements, and the parent obtained the second and fourth measurements).
After all Icare ONE measurements were obtained, Goldmann applanation was performed by a single clinician (S.F.F.) who was masked to the Icare ONE readings. The clinician obtained 2 Goldmann applanation readings for each eye, alternating between right and left eyes in children with 2 eyes. An average Goldmann applanation IOP was calculated for each eye. All IOP measurements were obtained prior to pupil dilation. At the end of the clinic visit, each parent completed a survey of his or her experience and ease of use of the Icare ONE device.
The Icare ONE measurements were downloaded from the study device at the end of each clinic session. For comparison of IOP by Icare ONE to IOP by Goldmann applanation tonometry, the average of the 2 Icare ONE readings taken by the clinic examiner was used for each analyzed eye.
For analysis of data, 1 eye per child was included, either the right eye or the eye with glaucoma (in unilaterally affected or monocular subjects). Continuous normally distributed variables between groups were compared using a paired, 2-tailed t test in which the threshold for significance was P < .05. Mean ± standard deviation is documented unless otherwise specified. Linear regression analysis was carried out on the 4 Icare ONE measurements from each study eye. A Bland-Altman plot of the average vs the difference between Icare ONE measurements by the clinic examiner and Goldmann applanation measurements (in mm Hg) was carried out. Finally, an area under the receiver operator curve (ROC) was performed to determine the sensitivity of the measurements taken by the clinic examiner using Icare ONE in detecting a Goldmann applanation measurement higher than 21 mm Hg. A Fisher exact test was used to assess the effect of vision, strabismus, nystagmus, corneal abnormalities, CCT, and previous surgery/surgeries on difference between Icare ONE and Goldmann applanation. Statistical analysis was performed using JMP Pro 9 (SAS, Cary, North Carolina, USA), and Graphpad prism version 5.01 for Windows (GraphPad Software, San Diego, California, USA; www.graphpad.com ) was used to generate Bland-Altman plots.
Results
Included were 60 eyes of 60 subjects. Demographic and clinical data are recorded in Table 1 . The mean subject age was 11.7 years (range 5–17 years). Forty-nine of 60 children (83%) had an established glaucoma diagnosis in the study eye; the rest were glaucoma suspects. Most children (39/60, 65%) had undergone 1 or more glaucoma surgeries, and 41 of 60 (68%) were taking at least 1 glaucoma medication.
| N | % | |
|---|---|---|
| Sex | ||
| Male | 29 | 48 |
| Female | 31 | 52 |
| Glaucoma diagnosis | ||
| Aphakic/pseudophakic | 19 | 32 |
| Anterior segment dysgenesis | 3 | 5 |
| Congenital | 13 | 22 |
| Juvenile open-angle | 4 | 7 |
| Steroid-induced | 1 | 2 |
| Suspect | 11 | 18 |
| Uveitis-associated | 7 | 12 |
| Other | 2 | 3 |
| Strabismus a | ||
| Yes | 22 | 37 |
| No | 37 | 62 |
| Nystagmus | ||
| Yes | 7 | 12 |
| No | 53 | 88 |
| Corneal abnormalities b | ||
| Yes | 5 | 8 |
| No | 55 | 92 |
| Glaucoma surgeries (#) | ||
| 0 | 21 | 35 |
| 1 | 15 | 25 |
| 2 | 15 | 25 |
| ≥3 | 9 | 15 |
| Glaucoma medications (#) | ||
| 0 | 19 | 32 |
| 1 | 14 | 23 |
| 2 | 19 | 32 |
| ≥3 | 8 | 13 |
| Visual acuity (Snellen) | ||
| ≥20/40 | 39 | 65 |
| 20/50–20/100 | 9 | 15 |
| 20/125–0/200 | 4 | 7 |
| 20/200 | 8 | 13 |
a Assessment of strabismus was not applicable to the one monocular subject enrolled in the study.
b Three subjects had clear corneal transplants and 2 had Haab striae through the visual axis. Measurement technique was not altered for these patients.
Clinic examiners (N.G.G. or S.G.P.) successfully obtained IOP by Icare ONE in each eye of all enrolled subjects. All parents were able to obtain IOP by Icare ONE in at least 1 eye. In 1 subject, the parent was unable to obtain a reliable IOP by Icare ONE in the monocular subject’s glaucomatous eye after several repeated attempts. In this case, the IOP obtained by the parent was excluded from the analysis.
Icare ONE IOP readings by the clinic examiner were statistically different from Goldmann applanation IOP readings (mean difference [absolute value] 3.3 ± 4.0 mm Hg, P = .001). The corresponding mean difference (absolute value) between Icare ONE IOP readings by clinic examiner vs parent (pooling both Group 1 and 2) was 1.9 ± 1.9 mm Hg, P = .008 ( Table 2 ). There was a good linear correlation between Icare ONE IOP measured by the parent and by the examiner (R 2 = 0.95) and between Icare ONE and Goldmann applanation tonometry (R 2 = 0.83 for examiner and R 2 = 0.83 for parent).
| Method of Tonometry | N (# Eyes) | IOP, mm Hg Mean ± SD | IOP, mm Hg Median |
|---|---|---|---|
| Icare ONE by clinic examiner | 60 | 19.0 ± 8.4 | 18.3 |
| Icare ONE by parent | 59 | 17.8 ± 8.3 | 17.0 |
| GAT | 60 | 16.9 ± 5.7 | 16.5 |
| Icare ONE by clinic examiner – GAT a | 60 | 3.3 ± 4.0 | 2.0 |
| Icare ONE by clinic examiner – ICare ONE by parent b | 59 | 1.9 ± 1.9 | 1.5 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


