Purpose
To determine relationships between vision, forward scatter, and total corneal and graft thicknesses after Descemet stripping endothelial keratoplasty (DSEK).
Design
Prospective, cohort study.
Methods
Forty-four eyes with Fuchs endothelial dystrophy were examined before and at 1, 3, 6, and 12 months after DSEK; all eyes were pseudophakic after surgery. Central total corneal and graft thicknesses were measured using confocal microscopy. Best-corrected high-contrast visual acuity (BCVA) was measured using the electronic Early Treatment Diabetic Retinopathy Study protocol, and forward light scatter was measured using a straylight meter.
Results
Total corneal thickness was 610 ± 50 μm (mean ± standard deviation) before DSEK, increased to 680 ± 74 μm by 1 month after DSEK ( P < .001), and stabilized at 660 ± 68 μm by 3 months after DSEK ( P = .03 vs 1 month). Graft thickness was 170 ± 57 μm at 1 month, decreased to 157 ± 49 μm by 3 months ( P = .004), and then remained stable through 12 months (156 ± 51 μm; P = .99 vs 3 months). BCVA was 0.44 ± 0.21 logarithm of the minimal angle of resolution (logMAR) units (Snellen equivalent, 20/55) before DSEK, improved to 0.26 ± 0.20 logMAR units (Snellen equivalent, 20/36) by 3 months ( P < .001), and improved to 0.16 ± 0.16 logMAR units (Snellen equivalent, 20/29) at 12 months ( P < .001 vs 3 months). BCVA and forward light scatter did not correlate with corneal or graft thickness after DSEK.
Conclusions
Stromal edema resolves by 3 months after DSEK for Fuchs dystrophy, whereas visual acuity continues to improve through 12 months. Thicker corneas and grafts are not associated with worse visual acuity or increased forward scatter.
Posterior lamellar keratoplasty has replaced penetrating keratoplasty as the treatment of choice for corneal endothelial dysfunction, and presently, the most common technique is Descemet stripping endothelial keratoplasty (DSEK) with the graft prepared by using a microkeratome. During DSEK, the Descemet membrane and endothelium are removed from the host cornea and are replaced with donor tissue that consists of the Descemet membrane, endothelium, and a posterior stroma of variable thickness. As a result, the cornea after DSEK is thicker than a normal cornea, which may affect the visual outcome.
The goal of this prospective, observational study was to determine if worse visual acuity and higher intraocular forward light scatter were associated with thicker corneas or grafts after DSEK, because such relationships could provide guidance for optimizing the thickness of grafts during preparation. We examined changes in total corneal, host, and graft thicknesses after DSEK and determined if postoperative vision and light scatter were correlated with the thickness of the cornea or graft. Early postoperative visual acuity and light scatter data have been reported previously for the initial patients in this series.
Methods
Subjects
Patients requiring DSEK because of decreased vision caused by Fuchs endothelial dystrophy were recruited from the cornea service at Mayo Clinic, Rochester, Minnesota. Subjects were excluded if they had a filtering bleb or uncontrolled glaucoma; central corneal scarring unrelated to Fuchs dystrophy, as determined by slit-lamp biomicroscopy; or a history of herpetic keratitis, maculopathy, or optic neuropathy. All eyes were either pseudophakic or had lenticular changes requiring cataract extraction.
Descemet Stripping Endothelial Keratoplasty Procedure
DSEK was performed under general or local anesthesia by using a method similar to that described by Price and Price, and as described previously. In all cases, the donor tissue was prepared with a mechanical microkeratome (ALTK; Moria, Antony, France) by either the surgeon (n = 11) or by the local eye bank (n = 33). The intended thickness of the anterior corneal lamella was 300 or 350 μm. After stripping Descemet membrane from the central cornea, the posterior lamellar donor tissue was folded and inserted by forceps or over a glide into the anterior chamber through a 5- to 6-mm scleral tunnel incision, and the anterior chamber was filled with air to aid attachment of the graft to the host cornea. Phacoemulsification and intraocular lens insertion were performed through the same incision before stripping the Descemet membrane in cases that were combined with cataract surgery. After surgery, eyes were treated with a topical antibiotic for 1 week and with topical prednisolone acetate 1%, 4 times daily, which was tapered to once daily over 3 to 6 months after surgery.
Confocal Microscopy
Central corneas were examined by using a confocal microscope (ConfoScan 4 with z-ring adapter; Nidek Technologies, Freemont, California, USA) before and after DSEK. The technique has been described in detail previously. Briefly, the confocal objective was aligned with the anesthetized cornea to provide an en face image of the endothelium, and central full-thickness scans of the cornea were obtained with the z-ring adapter in place. Digital images were recorded at 25 frames per second, and the step distance between frames was 4 μm; the instrument repeated scans through the cornea until 350 frames were acquired.
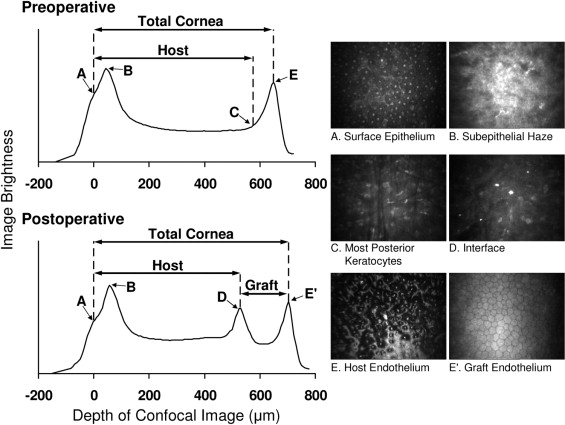
The best full-thickness scan through the cornea was selected for analysis. Thickness was calculated by using the graph of intensity of the confocal images through the cornea ( Figure 1 ). Before and after DSEK, total corneal thickness was the distance between the brightest image of the surface epithelium and the endothelium. Because Descemet membrane is not visible in confocal microscopy images, we estimated host thickness before DSEK as the distance between the surface epithelium and the most posterior image containing keratocytes, but not endothelial cells. After DSEK, host thickness was the distance between the surface epithelium and the graft–host interface, which was identified by either a peak in the intensity graph or by the presence of brightly reflective particles (similar to that after laser in situ keratomileusis ), and graft thickness was the distance between the interface and the endothelium.
Outcome Measures
Patients were examined before surgery and at 1, 3, 6, and 12 months after DSEK. At each examination, high-contrast visual acuity, low-contrast visual acuity, and intraocular forward light scatter were measured, in addition to thickness measurements by confocal microscopy. Before surgery, central corneal thickness also was measured by ultrasonic pachometry (DGH 1000; DGH Technologies, Inc, Frazer, Pennsylvania, USA).
Best-corrected high-contrast visual acuity (BCVA) and uncorrected high-contrast visual acuity were measured by using the electronic Early Treatment Diabetic Retinopathy Study testing protocol. Low-contrast visual acuity was measured by using a backlit 10% Sloan Translucent Low Contrast Chart (Precision Vision, LaSalle, Illinois, USA) under photopic (screen brightness, 139 cd/m 2 ) and mesopic (screen brightness, 1.1 cd/m 2 ) conditions. Visual acuity was recorded as letter scores and then converted to logarithm of the minimal angle of resolution (logMAR) units and Snellen equivalent. Contrast sensitivity was measured by using the Functional Acuity Contrast Test (Vision Sciences Research Corporation, San Ramon, California, USA), as described previously.
Intraocular forward light scatter was measured with a straylight meter (C-Quant; Oculus, Lynnwood, Washington, USA) by using the compensation comparison method to measure retinal stray light. Forward light scatter was proportional to the straylight parameter and was expressed as the logarithm of the straylight parameter.
Statistical Analysis
Outcome measures were compared from before and after surgery by using generalized estimating equation models to allow for possible correlation between fellow eyes of the same patient. P values were adjusted for multiple comparisons by using the Bonferroni technique, and P ≤ .05 was considered statistically significant. Correlations between thickness, visual acuity, and forward scatter were assessed by the Pearson correlation coefficient if the data were distributed normally and by the Spearman test if they were not. P values for correlations were calculated by using generalized estimating equation models. To detect a correlation of at least 0.45, which would indicate a predictive relationship (from r 2 ) of 20%, we required a sample size of 36 eyes when thickness was stable at 6 months after DSEK (α = 0.05, β = 0.20). Post hoc analyses determined the minimum detectable correlation at 12 months (α = 0.05, β = 0.20).
For precut donors, graft thickness reported by the eye bank was compared with graft thickness measured by confocal microscopy at 3 months by using a paired t test, and the correlation was assessed by the Pearson correlation coefficient. Preoperative total corneal thickness measured by confocal microscopy was compared with that measured by ultrasonic pachometry by using the Wilcoxon signed-rank test. All statistical analyses were performed with SAS statistical software (SAS Institute, Inc, Cary, North Carolina, USA).
Results
Subjects
Forty-four eyes of 38 patients were enrolled in the study, of which 40 eyes (34 patients) were examined through 6 months. At 12 months, data were available only for 31 eyes (27 patients) because 2 patients (2 eyes) dropped out of the study, 1 patient (1 eye) died, 1 eye (1 patient) had graft failure, and 9 eyes (9 patients) had not reached the 12-month examination yet. Mean age ± standard deviation at the time of surgery was 67 ± 10 years (range, 41 to 87 years).
Corneal Thickness
Before surgery, total corneal thickness measured by confocal microscopy (610 ± 50 μm) was lower than that measured by ultrasonic pachometry (659 ± 50 μm; P < .001). By using confocal microscopy, total corneal thickness increased by 1 month after DSEK compared with that before surgery ( P < .001) and remained stable between 3 and 12 months ( P = .99; Table 1 ; Figure 2 ). Host thickness decreased by 1 month after DSEK ( P < .001), and thereafter remained stable through 12 months ( P = .37 vs 1 month; Table 1 ; Figure 2 ). Graft thickness decreased between 1 and 3 months after DSEK ( P = .004), and then remained stable through 12 months ( P = .99 vs 3 months; Table 1 ; Figure 2 ). Central graft thickness ranged from 82 to 340 μm at 3 months and from 77 to 327 μm at 12 months.
| Mean Thickness ± Standard Deviation (μm) | |||||
|---|---|---|---|---|---|
| Preoperative | 1 Month | 3 Months | 6 Months | 12 Months | |
| All eyes a | |||||
| N | 41 | 40 | 41 | 37 | 29 |
| Total cornea b | 610 ± 50 | 680 ± 74 | 660 ± 68 | 667 ± 64 | 659 ± 66 |
| Host c | 571 ± 49 | 510 ± 42 | 502 ± 52 | 508 ± 38 | 503 ± 35 |
| Graft d | — | 170 ± 57 | 157 ± 49 | 159 ± 47 | 156 ± 51 |
a Data for just the eyes that were examined through 12 months were similar to those for all eyes (see Supplemental Table 1 available at AJO.com ).
b Preoperative vs 1 month, P < .001; 1 month vs 3 months, P = .03; 3 vs 12 months, P = .99; preoperative vs 12 months, P = .006.
c Preoperative vs 1 month, P < .001; 1 month vs 12 months, P = .37.
Visual Acuity and Contrast Sensitivity
BCVA improved between before and 1 month after DSEK ( P = .002), between 1 and 3 months after DSEK ( P = .006), and between 3 and 12 months after DSEK ( P < .001, Figure 2 ). Low-contrast visual acuity also improved by 1 month compared with that before surgery, with continued improvement between 1 month and 12 months ( Table 2 ; Figure 3 ).
| Mean logMAR Visual Acuity ± Standard Deviation (Snellen Equivalent) | |||||
|---|---|---|---|---|---|
| Preoperative | 1 Month | 3 Months | 6 Months | 12 Months | |
| All eyes a | n = 44 | n = 43 | n = 43 | n = 40 | n = 31 |
| High contrast (best corrected) b | 0.44 ± 0.21 (20/55) | 0.34 ± 0.21 (20/44) | 0.26 ± 0.20 (20/36) | 0.22 ± 0.17 (20/33) | 0.16 ± 0.16 (20/29) |
| High contrast (uncorrected) c | 0.68 ± 0.25 (20/96) | 0.53 ± 0.24 (20/68) | 0.49 ± 0.21 (20/62) | 0.47 ± 0.18 (20/59) | 0.39 ± 0.21 (20/49) |
| Low contrast (photopic, best corrected) d | 0.90 ± 0.22 (20/159) | 0.72 ± 0.22 (20/105) | 0.64 ± 0.20 (20/87) | 0.62 ± 0.20 (20/83) | 0.55 ± 0.21 (20/71) |
| Low contrast (mesopic, best corrected) e | 1.05 ± 0.08 (20/224) | 0.99 ± 0.12 (20/195) | 0.95 ± 0.12 (20/178) | 0.94 ± 0.14 (20/174) | 0.88 ± 0.13 (20/152) |
a Data for just the eyes that were examined through 12 months were similar to those for all eyes (see Supplemental Table 2 available at AJO.com ).
b Preoperative vs 1 month, P = .002; 1 month vs 3 months, P = .006; 3 vs 12 months, P < .001.
c Preoperative vs 1 month, P = .005; 1 month vs 3 months, P = .43; 3 vs 12 months, P = .02.
d Preoperative vs 1 month, P < .001; 1 month vs 3 months, P = .01; 3 vs 12 months, P = .04.
e Preoperative vs 1 month, P = .006; 1 month vs 3 months, P = .06; 1 month vs. 12 months, P = .01.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


