Purpose
To describe the histologic development of the human central retina from fetal week (Fwk) 22 to 13 years.
Design
Retrospective observational case series.
Methods
Retinal layers and neuronal substructures were delineated on foveal sections of fixed tissue stained in azure II–methylene blue and on frozen sections immunolabeled for cone, rod, or glial proteins. Postmortem tissue was from 11 eyes at Fwk 20–27; 8 eyes at Fwk 28–37; 6 eyes at postnatal 1 day to 6 weeks; 3 eyes at 9 to 15 months; and 5 eyes at 28 months to 13 years.
Results
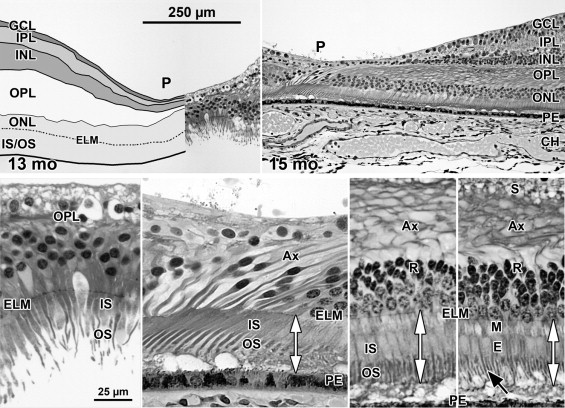
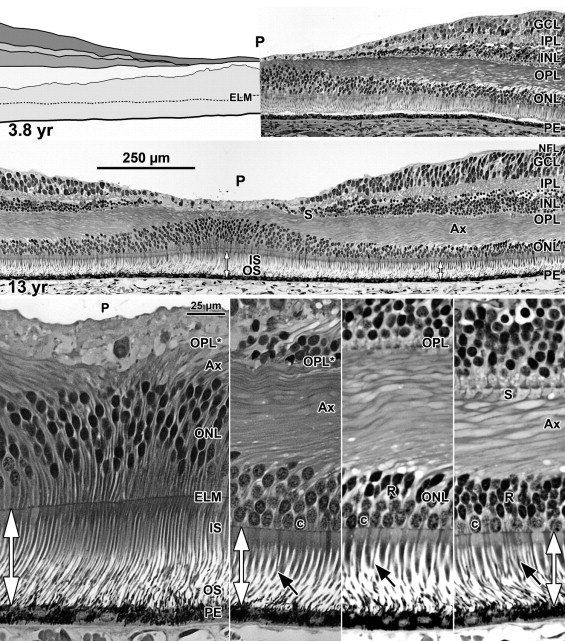
At Fwk 20–22 the fovea could be identified by the presence of a single layer of cones in the outer nuclear layer. Immunolabeling detected synaptic proteins, cone and rod opsins, and Müller glial processes separating the photoreceptors. The foveal pit appeared at Fwk 25, involving progressive peripheral displacement of ganglion cell, inner plexiform, and inner nuclear layers. The pit became wider and shallower after birth, and appeared mature by 15 months. Between Fwk 25 and Fwk 38, all photoreceptors developed more distinct inner and outer segments, but these were longer on peripheral than foveal cones. After birth the foveal outer nuclear layer became much thicker as cone packing occurred. Cone packing and neuronal migration during pit formation combined to form long central photoreceptor axons, which changed the outer plexiform layer from a thin sheet of synaptic pedicles into the thickest layer in the central retina by 15 months. Foveal inner and outer segment length matched peripheral cones by 15 months and was 4 times longer by 13 years.
Conclusions
These data are necessary to understand the marked changes in human retina from late gestation to early adulthood. They provide qualitative and quantitative morphologic information required to interpret the changes in hyper- and hyporeflexive bands in pediatric spectral-domain optical coherence tomography images at the same ages.
The fovea is the most critical portion of the primate retina for high visual acuity and color vision. Its early development begins at fetal week (Fwk) 12 with a complex expression sequence of cell-specific molecules. After midgestation an outward displacement of inner retinal layers occurs to form a foveal pit, and after birth an inward displacement of the photoreceptors occurs to raise foveal cone density 10×. Spectral-domain optical coherence tomography (SDOCT) has become a valuable tool for clinical diagnosis of normal and diseased adult human retina. Recently, Maldonado and associates used a portable research hand-held SDOCT unit to image human retinas from premature infants through adults. They documented the immaturity of the neonatal human fovea and the marked difference in SDOCT layers from the standard adult SDOCT image. However, there is still little detailed description of histologic development and almost no quantitative information about human central retina around birth. This paper provides the first detailed histologic description of human central retinal development from midgestation to early adulthood. In particular, it shows the marked change in foveal photoreceptors from a single layer of short undeveloped cones up to birth to tightly packed and highly elongated cones in the adult. Understanding these changes is essential to make the correct interpretation of neonatal SDOCT images.
Methods
Tissue sections for this study were from eyes obtained under Human Subjects Approval #010447. Eyes younger than Fwk 22 were from elective abortions; only normal-appearing fetuses were used. Older prenatal and neonatal eyes were from infants who survived days to weeks in the neonatal intensive care unit; babies with diagnosed conditions that might affect retinal development were not included in this report. Infant eyes were obtained from children who died of accidental causes, or of diseases that did not affect the retina.
Eyes were enucleated 2–8 hours after death, the cornea and lens removed, and the globe immersed in a mixture of 4% paraformaldehyde and 0.5% glutaraldehyde in pH.7.4 phosphate buffer (PB) for 1 day to months. In 1 of the pair of eyes, the horizontal meridian was embedded in glycol methacrylate and cut serially at 2–3 μm. This method gives excellent preservation and imaging of tissue. In a few eyes the entire globe was embedded in paraffin and sectioned serially at 12 μm. This method was less time-consuming, but the retina was usually detached from the pigment epithelium. In some eyes the horizontal meridian was frozen-sectioned serially at 12 μm, allowing immunolabeling at a known locus with a wide variety of antibodies. In each method, every tenth slide was stained with 1% azure II–methylene blue to locate the fovea.
Immunolabeling
Frozen sections were labeled as previously reported. The rod marker was mouse anti-rod opsin (4D2, 1/300; gift of R. Molday). Cone markers included rabbit anti-long-and-medium (L&M)-wavelength-selective cone opsin (1/2000, gift of J. Saari), rabbit anti-short (S)-wavelength-selective cone opsin (1/10 000, gift of J. Nathans), mouse anti-cone arrestin (7G6, 1/250–500; gift of P. MacLeish), and mouse anti-cone alpha transducin (A1.1, 1/250–500; gift of J. Hurley). Mouse anti-synaptophysin (1/1000; Sigma-Aldrich S5768) labels cone synaptic pedicles heavily and rod synaptic spherules lightly. Rabbit anti–cellular retinaldehyde-binding protein (CRALBP; 1/1000; gift of J. Saari) was used to label Müller glial cytoplasm.
Imaging
All photographs were from sections in which the foveal center could be reliably identified. Plastic or paraffin sections stained with azure II–methylene blue were imaged digitally using a Nikon E1000 wide-field digital microscope (Nikon Instruments, Inc, Melville, New York, USA). Immunolabeled sections were imaged using a high-sensitivity Hamamatsu camera (Hamamatsu Corp, Bridgewater, New Jersey, USA) or in an Olympus Fluoview 1000 confocal microscope (Olympus America, Center Valley, Pennsylvania, USA). All images were processed in Adobe Photoshop CS5 for size, color balance, sharpness, and contrast.
Segmentation of Light Micrographs
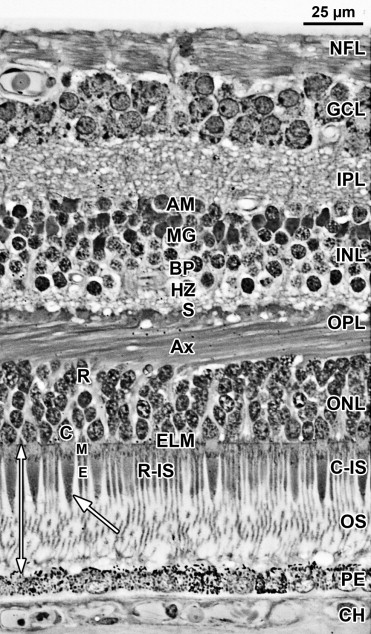
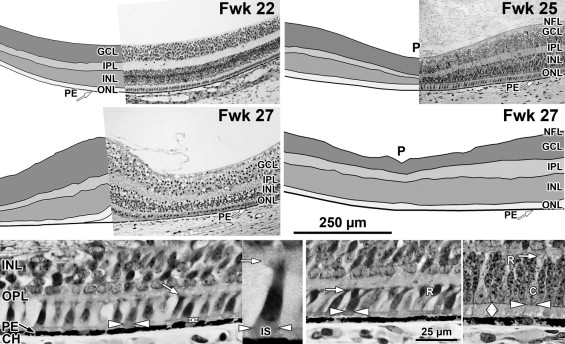
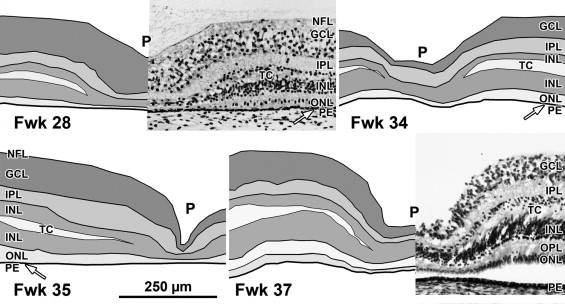
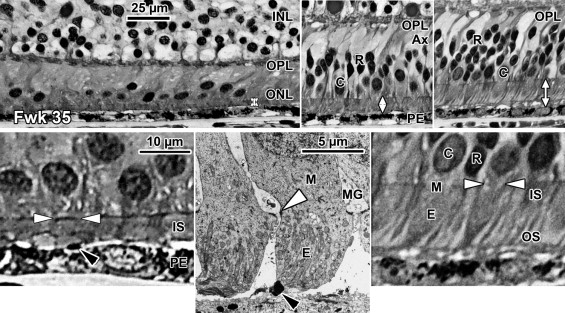
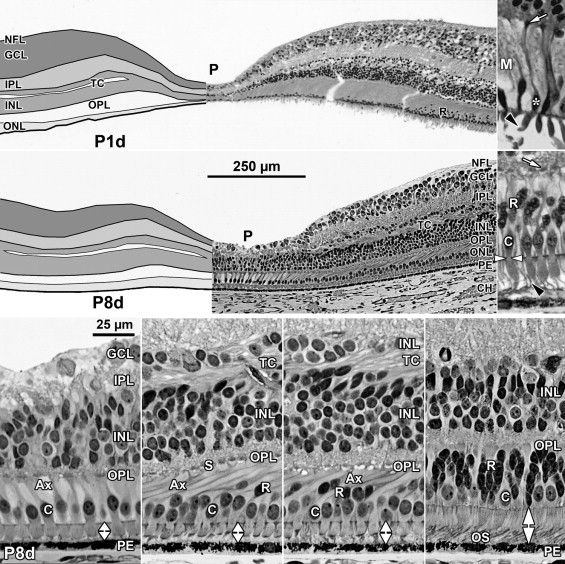
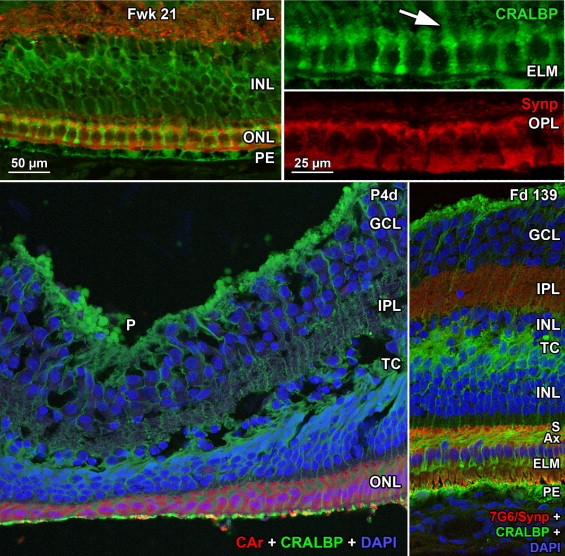
A low-power foveal montage was created at selected ages. One half was imaged in Photoshop and paths drawn along the inner surface of the retina and the borders between the ganglion cell (GCL) and inner plexiform layer (IPL); the IPL and inner nuclear layer (INL); the INL and outer plexiform layer (OPL); the OPL and outer nuclear layer (ONL); and the inner border of the retinal pigment epithelium (RPE). Layers were differentially shaded (( Figure 1 provides an orientation to the layers; Figures 2 through 7 show shaded layers).). In young retinas, the OPL was thin and included in ONL, but in older retinas the OPL was distinguished as a separate layer. A dotted path was drawn along the external limiting membrane (ELM), separating the proximal ONL from the distal inner segment/outer segment (IS/OS) layer.
Results
Normal Adult Retina
A section from adult human retina ∼2 mm nasal to the fovea is shown in Figure 1 for orientation to the layers and abbreviations used in the developing retina. A young adult fovea is shown in Figure 7 (Bottom row).
Development of the Human Fovea
Fetal week 20–27 (11 retinas examined)
At midgestation, the future fovea had 3 distinctive differences from peripheral retina. First, the GCL is much thicker than in the periphery. Second, up to birth the ONL is formed by a single layer of cone photoreceptors ( Figure 2 , Bottom left; Figure 8 , Top left). This is in marked contrast to the thicker peripheral ONL containing both cones and rods ( Figure 2 , Bottom right; Figure 8 , Top middle-left and middle-right). Third, the OPL is a narrow band of cone synaptic pedicles ( Figure 2 , Bottom left and insert, Bottom middle; Figure 8 , Top right upper panel, and Bottom). These layers will undergo developmental changes that are critical to interpreting late prenatal and neonatal SDOCT images. In the following narrative, numbers like 800 μm or 2 mm indicate distance from the fovea center.
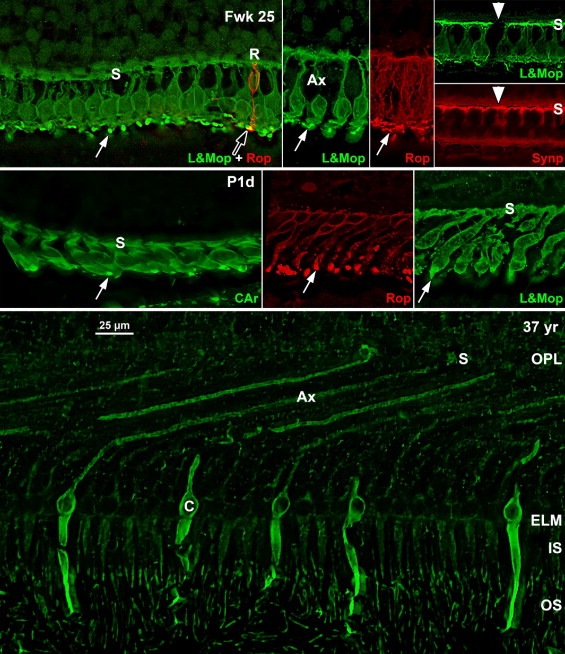
Rods are absent across the central 1500–1800 μm, forming a region called the rod-free zone underlying the thickened GCL. Central cones are 6–8 μm wide, with a large prominent nucleus ( Figure 2 , Bottom left insert; Figure 8 , Top left; Figure 9 , Top right lower panel). Each cone has a short axon (Ax), which ends below the cell body in a synaptic pedicle ( Figure 2 , Bottom left insert, arrow; Figure 8 , Top middle-left); these form the thin OPL ( Figure 8 , Top row). Foveal cones are separated by Müller cell processes filling the space between cones. These end in small bulbs forming junctions with the cone cell bodies, creating the ELM ( Figure 2 , Bottom left, white arrowhead; Figure 9 , Top row).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


