Purpose
To investigate the effects of ranibizumab 0.5 mg on gray hyper-reflective subretinal lesions diagnosed by spectral-domain optical coherence tomography (SD OCT) in patients with exudative age-related macular degeneration (AMD).
Design
Retrospective interventional study.
Methods
Data from 28 consecutive patients affected with neovascular AMD that presented subretinal hyper-reflective lesions as visualized by SD OCT were collected. Gray hyper-reflective subretinal lesion characteristics were analyzed before and after intravitreal ranibizumab 0.5 mg injection.
Results
Thirty eyes of 28 patients (5 male, 23 female, aged 57–91 years) were included. At study entry, gray lesion was associated with exudative features in 24 of 30 eyes (80%), including subretinal fluid (SRF) in 20 of 30 eyes (67%) and retinal cystoid spaces in 11 of 30 eyes (37%). Twenty-four eyes with exudative features at study entry received prompt treatment; 6 eyes without exudative features at study entry received deferred treatment (after 1 month observation), when exudative signs emerged (SRF in 3/6 eyes and retinal cystoid spaces in 5/6 eyes). Ninety-three percent of the gray lesions responded to ranibizumab treatment at 2 months and 77% at 6 months. Gray hyper-reflective subretinal lesion thickness was significantly reduced after treatment at both 2 months (from 482 ± 116 μm to 367 ± 102 μm, P < .0001) and 6 months (from 482 ± 116 μm to 369 ± 71 μm, P < .0001).
Conclusion
Our findings suggest that gray hyper-reflective subretinal lesions might be considered as a qualitative criterion for retreatment of exudative AMD. They may represent an early sign of active choroidal neovascularization, and should prompt to early treatment.
Age-related macular degeneration (AMD) is the main cause of visual impairment in developed countries among individuals over 50 years of age. Recently, the prognosis for neovascular AMD has improved owing to the availability of intravitreal anti–vascular endothelial growth factor (VEGF) therapy.
Ranibizumab (Lucentis; Genentech Inc, San Francisco, California, USA [a subsidiary of Roche, Basel, Switzerland]) is a recombinant humanized monoclonal antibody fragment that neutralizes all biologically active forms of VEGF-A. Treatment with intravitreal ranibizumab for exudative AMD has demonstrated, in 2 phase III clinical studies, efficacy in prevention of vision loss and improvement in mean visual acuity in a large proportion of patients. These results were obtained using a fixed-dosing regimen of ranibizumab 0.5 mg or 0.3 mg monthly, injected over 24 and 12 months, respectively.
The treatment protocol used in the Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular AMD (MARINA) and Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in AMD (ANCHOR) studies is hardly reproducible in real-life clinical settings. Two pivotal clinical studies (The Prospective Optical Coherence Tomography Imaging of Patients with Neovascular AMD Treated with Intra-Ocular Ranibizumab [PrONTO] and Comparison of Age-Related Macular Degeneration Treatment Trials [CATT] ) demonstrated that a variable-dosing regimen was effective (even though not strictly equal to a fixed-dosing regimen) in improving visual acuity and optical coherence tomography (OCT) outcomes, needing a far lower number of injections over a period of 12 and even 24 months. The only 2 criteria for reinjection based on OCT for the PrONTO and CATT studies were an increase of at least 100 μm in retinal thickness or presence of any hyporeflective retinal fluid.
Nonetheless, OCT imaging of the retina allows us to analyze many other characteristics and signs of choroidal neovascularization (CNV) activity than the usual aforementioned OCT-based retreatment criteria. Many papers have described findings such as outer retinal tubulations that can contribute to an increase in retinal thickness that is unresponsive to therapy and intraretinal hyper-reflective dots that are signs of activity and that do not contribute to retinal thickening in AMD. In the same category of OCT lesions, subretinal hyper-reflective lesions are neither retinal fluid nor retinal thickening.
A statistically significant association was found between fluorescein angiography leakage and eyes that displayed low reflectivity or undefined boundaries from subretinal material in classic and occult CNV. The authors of these studies suggested that subretinal lesions that displayed low intrinsic reflectivity and undefined boundaries could correspond either to the neovascular structure itself or to an inflammatory reaction. Giani and associates found high optical reflectivity and well-defined subretinal lesions in minimally classic CNV on spectral-domain OCT (SD OCT). Keane and associates, using time-domain OCT, found a significant reduction of subretinal lesions after anti-VEGF treatment.
The presence of hyper-reflective subretinal lesions that appear gray (“dirty gray,” as described by Freund and associates ) on SD OCT, corresponding to the previously described lesions by Giani and associates, Liakopoulos and associates, and Keane and associates, was observed in our clinical setting in patients diagnosed with exudative AMD. These lesions presented certain characteristics that we suspected to be signs of CNV activity.
Given that the role of hyper-reflective subretinal lesions has been ignored in earlier publications, we decided to perform a study of these lesions with the purpose of observing and analyzing their response to anti-VEGF therapy. We hypothesized that these subretinal hyper-reflective lesions could regress after treatment with intravitreal anti-VEGF therapy.
Methods
We reviewed the charts of all consecutive eyes with exudative AMD that underwent intravitreal ranibizumab 0.5 mg injection at the University Eye Clinic of Creteil between January 2013 and October 2013. This retrospective interventional study was performed in agreement with the French bioethical legislation and in agreement with the Declaration of Helsinki for research involving human subjects. The French Society of Ophthalmology’s ethics committee approval was obtained for this retrospective study.
Criteria for inclusion were: age ≥50 years old, presence of subfoveal CNV secondary to AMD as evaluated by fundus biomicroscopy, fluorescein angiography (FA) and infracyanine green angiography (ICGA), and presence of hyper-reflective subretinal lesions detected on SD OCT. Patients with previous treatment for exudative AMD in the study eye in the last 3 months were excluded, as well as patients that had previous surgery in the study eye over the last 6 months, refractive error of more than -6 diopters, macular hemorrhage, adult-onset foveomacular vitelliform dystrophy and other pattern dystrophies, fibroglial scar, CNV attributable to causes other than AMD, or active intraocular inflammation in the study eye.
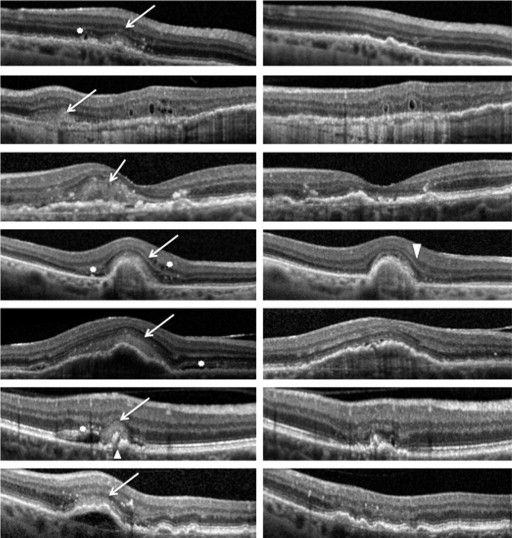
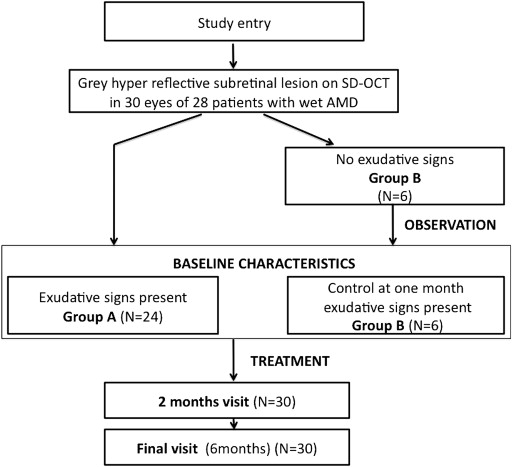
We performed a study baseline evaluation for all patients, which corresponded to the moment that gray hyper-reflective subretinal lesions were detected on SD OCT. Baseline characteristics for each eye were established at the visit when the gray lesion was noticed to be associated with exudative signs ( Figure 1 ).
At study baseline, each patient underwent assessment of best-corrected visual acuity (BCVA) using standard Early Treatment of Diabetic Retinopathy Study (ETDRS) charts and an ophthalmologic examination that included slit-lamp biomicroscopy, fundus examination, FA, ICGA, and SD OCT (Spectralis HRA-OCT; Heidelberg Engineering, Heidelberg, Germany). At each subsequent visit, we used the “follow-up” protocol of Spectralis Heidelberg Retina Angiograph OCT, which performs reacquisition of each OCT line actually referenced with the original and/or previous acquisition. All angiograms and corresponding SD OCT scans were independently evaluated at study baseline, at 2 months, and at final visit by 3 retinal specialists (E.H.S., G.Q., H.O.).
At study baseline, patients underwent treatment if neovascular activity was present (Group A), defined as either decrease of BCVA (at least 1 ETDRS line), or presence of fluid on OCT, or leakage from CNV on FA. If none of these criteria were found at study baseline, patients were observed for signs of evolution (control visit at 1 month, standard protocol in our clinic) (Group B). Outer retinal tubulations, as they are already known to be a degenerative retinal process, were not considered as a retreatment criterion. The number of injections was decided at the discretion of a senior ophthalmologist, according to intensity of neovascular activity, based on SD OCT features. Treatment consisted of intravitreal injection(s) of ranibizumab 0.5 mg performed monthly by a retinal specialist according to usual injection protocol.
At 2 and 6 months (final visit) from baseline, each patient underwent a detailed medical and ocular examination, assessment of BCVA using ETDRS charts, and ophthalmic examination, which included slit-lamp biomicroscopy, intraocular pressure assessment, fundus biomicroscopy, and SD OCT examination (standard assessment in our clinic). Patient demographic characteristics were recorded for the following data: sex, age, disease duration, and contralateral eye features.
We retrospectively assessed the morphologic effects of the treatment on gray hyper-reflective subretinal lesions, from baseline to 2 months and to final visit (6 months), by evaluating several gray lesion characteristics: (1) qualitative evolution of the gray hyper-reflective subretinal lesion: complete regression, partial regression, stabilization, increase; (2) gray lesion thickness, done by quantifying vertically the highest retinal thickness in the zone of the lesion; (3) gray lesion optical reflectivity: low (similar to inner/outer nuclear layer or plexiform reflectivity) or high (similar to retinal pigment epithelium [RPE] or nerve fiber layer reflectivity); (4) gray lesion boundaries: well-defined or undefined. The main outcome measure was the gray lesion characteristics at 2 months from baseline. Secondary outcome measures included BCVA changes and other OCT features, such as central macular thickness (CMT) and presence/absence of subretinal fluid (SRF), retinal cystoid spaces, and intraretinal hyper-reflective dots, at 2 and 6 months from baseline. We also analyzed the presence of recurrence of gray hyper-reflective subretinal lesions during follow-up.
Statistical analyses were performed using SAS (version 9.3; SAS Institute Inc, Cary, North Carolina, USA). Comparisons of mean BCVA, mean CMT, mean gray hyper-reflective subretinal lesion thickness, and mean number of injections between baseline visit, 2-month visit, and final visit were performed using the Student t test. The McNemar test was used to analyze morphologic qualitative changes (ie, gray lesion, fluid accumulation, and hyper-reflective dots) between baseline, 2-month visit, and final visit. The chosen level of statistical significance was P < .05.
Results
Clinical Characteristics of the Study Population at Study Baseline
Thirty consecutive eyes of 28 patients (5 male, 23 female, aged 57–91 years [mean 77.3 ± 7.2]) were included in the analysis. At baseline, BCVA was 61 ± 17.3 letters and CMT was 393 ± 125 μm. Demographic and clinical data are summarized in Table 1 . CNV was classified as classic in 8 eyes (27%), occult in 14 eyes (47%), minimally classic in 4 eyes (13%), and retinal angiomatous proliferation (RAP) in 4 eyes (13%). In our series, 9 of 30 eyes (30%) had undergone previous treatments for exudative AMD and 21 of 30 eyes (70%) were treatment naïve ( Table 1 ). One RPE tear was present in 1 eye at study baseline.
| Characteristics | No |
|---|---|
| Age, y (mean ± SD) | 77.3 ± 7.2 |
| No. of eyes | 30 |
| No. of patients | 28 |
| Sex (patients) | |
| Male | 5 |
| Female | 23 |
| Type of CNV (eyes) | |
| Classic | 8 |
| Occult | 14 |
| Minimally classic | 4 |
| Retinal angiomatous proliferation | 4 |
| Eyes with previous treatment | |
| Naïve eyes | 21 |
| Intravitreal ranibizumab | 9 |
| Photodynamic therapy | 2 |
| Laser photocoagulation | 1 |
Functional and Morphologic Changes After Treatment
At the 2-month visit (mean follow up 2.1 ± 0.8 months) the mean number of intravitreal injections was 1.90 ± 0.9. BCVA significantly changed (from 61 ± 17.3 letters to 65.8 ± 15.3 letters; P = .0071) compared to before treatment. CMT significantly decreased at the 2-month visit (from 393 ± 125 μm to 313 ± 72 μm; P < .0001) ( Table 2 ). After treatment, gray hyper-reflective subretinal lesions showed regression at 2 months in 28 of 30 eyes (93.3%) (either complete regression, in 18 of 30 eyes [60%], or partial regression, in 10 of 30 eyes [33.3%]) and stabilization in 2 of 30 eyes (6.7%). Mean gray lesion thickness decreased from 481 ± 115 μm before treatment to 366 ± 101 μm after treatment at 2 months ( P < .0001). At the 2-month visit, SRF, retinal cystoid spaces, and hyper-reflective dots were present in fewer eyes when compared to baseline (9 eyes vs 23 eyes, P = .0001; 12 eyes vs 16 eyes, P = .2188; and 18 eyes vs 26 eyes, P = .0078, respectively) ( Table 2 ).
| Before Treatment (Baseline) | After Treatment (2-Month Visit) | P | After Treatment (6-Month Visit) | P | |
|---|---|---|---|---|---|
| Presence of gray lesion | 30/30 (100%) | 12/30 (40%) | Not applicable | 12/30 (40%) | Not applicable |
| Gray lesion thickness (μm) (mean ± SD) | 481 ± 115 | 366 ± 101 | <.0001 | 369 ± 71 | <.0001 |
| CMT (μm) (mean ± SD) | 393 ± 125 | 313 ± 72 | <.0001 | 308 ± 75 | <.0001 |
| VA (ETDRS letters) (mean ± SD) | 61 ± 17.3 | 65.8 ± 15.3 | .0071 | 64.7 ± 16.3 | .0481 |
| Mean follow-up (mo) | 2.1 ± 0.8 | 6.2 ± 1.8 | |||
| No. of IVT | 1.90 ± 0.92 | 3.6 ± 1.8 | |||
| Other exudative features | |||||
| Subretinal fluid | 23/30 (77%) | 9/30 (30%) | .00012 | 8/30 (27%) | <.0001 |
| Retinal cystoid spaces | 16/30 (53%) | 12/30 (40%) | .2188 | 8/30 (27%) | .0078 |
| Hyper-reflective dots | 26/30 (87%) | 18/30 (60%) | .0078 | 20/30 (67%) | .0313 |
At the final visit, the mean follow-up period was 6.2 ± 1.8 months and the mean number of intravitreal injections was 3.6 ± 1.8. BCVA significantly changed at the final visit (from 61 ± 17.3 letters to 64.7 ± 16.3 letters; P = .0481) compared to before treatment. CMT also significantly decreased at the final visit (from 393 ± 125 μm to 308 ± 75 μm; P < .0001). Gray lesions showed complete regression at the final visit in 18 of 30 eyes (60%), partial regression in 5 of 30 eyes (16.6%), stabilization in 6 of 30 eyes (20%), and increase in 1 eye (3.3%). Mean gray lesion thickness decreased from 481 ± 115 μm before treatment to 369 ± 71 μm after treatment at the final visit ( P < .0001). At the final visit, SRF, retinal cystoid spaces, and hyper-reflective dots were present in fewer eyes when compared to baseline (8 eyes vs 23 eyes, P < .0001; 8 eyes vs 16 eyes, P = .0078; and 20 eyes vs 26 eyes, P = .0313, respectively).
A recurrence of exudative signs was recorded between months 2 and 6 for 12 of 30 eyes (40%). This recurrence happened at an average of 4.18 ± 1 months from baseline. Eyes that presented a recurrence of CNV activity between months 2 and 6 had had 2.5 ± 0.8 intravitreal injections on average. SD OCT at recurrence showed gray lesions in 9 of 12 eyes (75%), SRF in 6 of 12 eyes (50%), and retinal cystoid spaces in 7 of 12 eyes (58.3%).
Comparison Between Group A (Prompt Treatment) and Group B (Deferred Treatment)
In 24 of 30 eyes, the gray hyper-reflective subretinal lesion was present with exudative signs at study baseline (ie, hyporeflective fluid and cystoid spaces), and patients were treated with ranibizumab 0.5 mg intravitreal injections on a pro re nata (PRN) basis (Group A: prompt treatment) ( Figure 2 ). In 6 of 30 eyes, the gray hyper-reflective subretinal lesion was present but exudative signs (ie, hyporeflective fluid and cystoid spaces) were absent at study baseline (Group B: deferred treatment). Given the lack of consensus on gray lesion as a sign of active CNV, patients did not receive treatment, but were observed. All 6 of these eyes developed exudative signs after an observation period of 1 month and thus received treatment ( Figure 3 ). It is remarkable that for this deferred treatment group, the gray hyper-reflective subretinal lesion thickness increased (from 397 ± 76 μm to 450 ± 97 μm), and that occurrence of other exudative signs (SRF appeared in 3 of 6 eyes and retinal cystoid spaces in 5 of 6 eyes) was observed in all eyes. Subgroup analysis showed no difference at baseline in mean age, sex, mean BCVA, CMT, gray lesion thickness, and CNV subtype distribution between Group A and Group B ( Table 3 ).