Purpose
To evaluate the onset of posterior vitreous detachment (PVD) including early changes at the vitreoretinal interface after uneventful phacoemulsification in nonmyopic eyes using optical coherence tomography (OCT) and ultrasound.
Design
Prospective consecutive study.
Methods
Patients undergoing cataract surgery at our unit between January and October 2010 were recruited and examined with OCT and ultrasound preoperatively. Inclusion criteria were complete vitreoretinal attachment, no ocular pathology other than cataract, and no previous ocular surgery. All patients underwent phacoemulsification with intraocular lens implantation. Postoperatively, OCT and ultrasound were performed 1 month and 3 months after surgery. Exclusion criteria were axial length ≥25 mm, lattice degeneration, intraoperative complications, and incomplete follow-up.
Results
Forty-nine eyes of 49 patients could be enrolled in the study. Some degree of PVD was noted in 29 eyes (59.2%) 1 month after surgery and in 35 eyes (71.4%) 3 months after surgery. Moreover, a significant decrease in prevalence of initial PVD and a corresponding increase of more advanced PVD stages throughout the duration of the study was observed. In patients older than 70 years some degree of PVD was diagnosed in 92.3% compared to 47.8% in patients younger than 70 years ( P = .002).
Conclusion
OCT facilitates the detection of early vitreoretinal separation that indicates initial PVD. After phacoemulsification the prevalence of some degree of PVD is consequently more frequent when supplementary OCT is used. Furthermore, OCT discloses a significant progression of PVD in the postoperative course. Patients older than 70 years are more likely to develop pseudophakic PVD.
Posterior vitreous detachment (PVD) is characterized by dehiscence of the posterior vitreous cortex from the inner limiting membrane (ILM) in conjunction with vitreous liquefaction. Cataract surgery promotes PVD as a result of postoperative alterations of the vitreous structure. In particular, phacoemulsification was shown to induce PVD in 58.6% of otherwise healthy eyes within 1 year and in 75.9% within 5 years after the surgery. The majority of postoperative PVDs, however, occur within the first postoperative month. The impact of phacoemulsification on the development of postoperative PVD was proved by Hilford and associates, showing a significantly higher prevalence of PVD in eyes that had undergone phacoemulsification compared to fellow phakic eyes.
To date, PVD has been assessed biomicroscopically and with ultrasound. Optical coherence tomography (OCT), however, has been found to facilitate the detection of early vitreoretinal interface changes that indicate initial PVD. Consequently, using supplementary OCT might disclose a higher overall PVD rate following phacoemulsification than previously reported.
The aim of our study is to evaluate the onset of PVD in nonmyopic eyes without lattice degeneration including the assessment of early vitreoretinal interface changes after uneventful phacoemulsification using OCT and ultrasound. Therefore, only eyes with complete vitreoretinal attachment were enrolled. Eyes with axial length ≥25 mm and lattice degeneration were excluded. Finally, complications associated with PVD were assessed.
Methods
Patients undergoing cataract surgery at the Department of Ophthalmology at the Medical University of Graz between January 10, 2010 and October 28, 2010 were recruited after their informed consent was obtained for this research protocol. Preoperatively, the patients underwent routine assessment for cataract surgery including best-corrected visual acuity (BCVA) measurement; applanation tonometry; laser interferometry (IOL Master; Carl Zeiss Meditec, Jena, Germany); slit-lamp examination including indirect ophthalmoscopy; and examination with high-definition OCT (Cirrus; Carl Zeiss Meditec) and ultrasound (Cine Scan S; Quantel Medical, Clermont-Ferrand, France) using a 10-MHz probe. All preoperative examinations were performed by 2 experienced examiners (A.B., C.S.). Inclusion criteria for the study were assumed complete vitreoretinal adherence; no other ocular diseases including age-related macular degeneration, diabetic retinopathy, myopic fundus, presence or history of uveitis, and venous or arterial occlusion; and no previous vitrectomy, buckle, or glaucoma surgery.
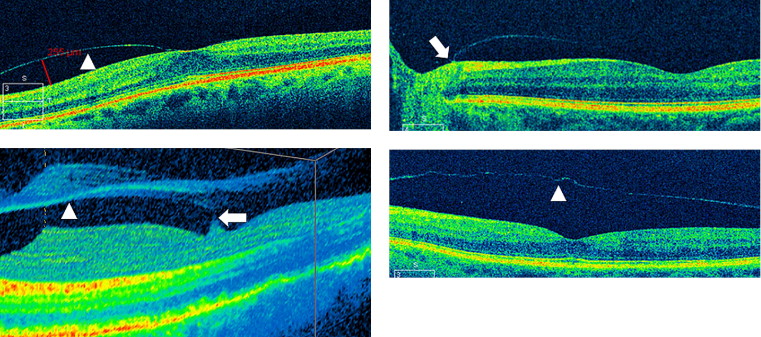
PVD was defined as the detachment of the posterior vitreous cortex from the inner retina. On ultrasound, the detached posterior vitreous cortex presented as a low-reflective membrane floating in the vitreous cavity. In OCT assessment, a discrete linear signal in the vicinity of the retina indicated the detached posterior vitreous cortex. The vitreoretinal separation was classified in stages. Accordingly, stage 0 indicated no PVD; stage 1 was characterized by focal perifoveal detachment of the posterior vitreous cortex involving 1 to 3 quadrants and a remaining attachment of the posterior vitreous cortex to the optic nerve head ( Figure 1 , Upper left and right); stage 2 was defined as perifoveal detachment of the posterior vitreous cortex in all quadrants with persistent vitreofoveal and vitreopapillary adhesion ( Figure 1 , Lower left); and stage 3 indicated complete detachment of the posterior vitreous cortex from the fovea but remaining attachment of the posterior vitreous cortex to the optic nerve head and midperiphery of the retina ( Figure 1 , Lower right). Stage 4 was diagnosed if the posterior vitreous cortex was completely detached from both the inner retinal surface and the optic nerve head. To verify stage 0, OCT and ultrasound were required. Stages 1 and 2 were only assessable with OCT. Stage 3 could be detected with OCT and ultrasound, whereas stage 4 was only assessable with ultrasound. In addition, the largest distance between the posterior vitreous cortex and the inner retina was measured when possible ( Figure 1 , Upper left). All patients underwent cataract surgery under local anesthesia using the phacoemulsification technique and implantation of a foldable intraocular lens in the capsular bag. Postoperatively, the patients were examined with OCT and ultrasound 1 month and 3 months after the surgery by 4 experienced examiners (A.B., C.S., D.I., E.M.L.). Exclusion criteria were any stage of PVD on the preoperative assessment, axial length ≥25 mm, lattice degeneration, intraoperative complications, and incomplete follow-up.
Statistical analysis was performed using SPSS 17.0 (SPSS, Inc, Chicago, Illinois, USA). Descriptive statistics were presented as mean ± standard deviation. Decimal BCVA was converted into logMAR. Associations between categorical variables were evaluated with Fisher exact test. The Kolmogorov-Smirnov test revealed normal distribution of continuous data. Accordingly, the difference in age was evaluated with the independent t test. The threshold for statistical significance was defined as P < .05.
Results
Overall, 263 eyes of 263 patients were recruited for the study. On the preoperative OCT assessment, some stage of PVD was observed in 188 eyes; stage 1 was detected in 129 eyes (68.6%), stage 2 in 54 eyes (28.7%), and stage 3 in 5 eyes (2.7%). Further, 12 eyes were excluded because of axial length ≥25 mm and 2 eyes because of lattice degeneration. In 3 patients, intraoperative complications including posterior capsule tear with vitreous loss (n = 2) and zonulysis (n = 1) occurred. Postoperatively, 9 patients were lost to follow-up. Consequently, only 49 eyes (49 patients) could be considered for analysis. The age of enrolled patients averaged 70.5 ± 9.5 years (range 41-87); 24 participants (49.0%) were female and 25 (51.0%) were male. The mean BCVA was 0.48 ± 0.23 logMAR, the intraocular pressure averaged 15.6 ± 1.9 mm Hg, and the mean axial length was 23.08 ± 1.09 mm.
Postoperatively, some degree of PVD was observed in 29 eyes (59.2%) at 1 month and in 35 eyes (71.4%) at 3 months after the surgery. PVD stage 1 was noted in 13 eyes (26.5%) at month 1 and in 6 eyes (12.2%) at month 3 ( P = .04); stage 2 in 4 eyes (8.1%) at month 1 and in 10 eyes (20.4%) at month 3 ( P = .008); stage 3 in 1 eye (2.0%) at month 1 and in 3 eyes (6.1%) at month 3 ( P < .001); and stage 4 in 11 eyes (22.4%) at month 1 and in 16 eyes (32.6%) at month 3 ( P = 0.42). Among eyes with stage 1 PVD, the posterior vitreous cortex was detached in 1 quadrant in 5 eyes, in 2 quadrants in 3 eyes, and in 3 quadrants in 5 eyes at 1 month. The superior quadrant was most frequently affected, followed by the nasal, inferior, and temporal quadrant. At month 3, only 1 quadrant was involved in 4 eyes, 2 quadrants in 1 eye, and 3 quadrants in 1 eye. Again, the superior quadrant was most frequently affected, followed by the nasal, inferior, and temporal quadrant. The distance between the posterior vitreous cortex and the inner retina averaged 181.7 ± 78 μm in stage 1, 198.2 ± 77.4 μm in stage 2, and 548.7 ± 113.9 μm in stage 3.
In patients older than 70 years (n = 26), some degree of PVD occurred in 24 eyes (92.3%) throughout the duration of the study, whereas in patients younger than 70 years (n = 23), PVD was detected in only 11 eyes (47.8%). This difference was significant ( P = .002). Additionally, the age of patients with a detectable degree of PVD at the end of follow-up averaged 73 ± 8.1 years, in contrast to 64.3 ± 10.1 years in those with no PVD ( P = .003). Twenty of 24 female patients (83.3%) experienced PVD, compared to 15 of 20 male patients (60.0%). This difference was not statistically significant ( P = 0.19). The age of female patients averaged 72.6 ± 8.4 years and male patients 68.5 ± 10.2 years ( P = .13).
Symptoms of complications associated with PVD were observed in 2 patients. One patient complained about photopsia during the first postoperative month; no retinal detachment or retinal break could be detected. At month 1, he had PVD stage 4. In the other patient, vitreofoveal traction syndrome was noted at the last follow-up visit ( Figure 1 , Lower left); no cystoid macular edema was associated.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


