Purpose
To characterize the phenomenon of dissociation of the 2 components of the sustained-release fluocinolone acetonide implant (Retisert; Bausch & Lomb) during removal or exchange procedures, or both, and to evaluate outcomes after these events.
Design
Retrospective, observational case series.
Methods
Retrospective review of 27 consecutive sustained-release fluocinolone acetonide implant (Retisert) exchanges or removals between 2001 and 2010 at the Cole Eye Institute. All patients had received the implant as treatment for noninfectious uveitis. Preoperative and postoperative characteristics were analyzed, and operative reports were reviewed to characterize the effects of intraoperative implant dissociation.
Results
Twenty-seven sustained-release fluocinolone acetonide implant (Retisert) exchange or removal surgeries were performed by 3 surgeons in 20 eyes of 19 patients. Of these 27 procedures, dissociation of the implant strut from the drug-containing cup occurred in 11 eyes (40.7%). Retrieval of the dislocated cup led to intraoperative complications, including posterior retinal tear (n = 1) and limited suprachoroidal hemorrhage (n = 1). The length of time that the implant resided in the eye correlated significantly with cup dissociation. Dissociated implants resided a mean of 47.4 months, whereas intact implants resided a mean of 32.5 months ( P = .0032). There was no long-term or short-term vision loss attributed to intraoperative implant dissociation.
Conclusions
Sustained-release fluocinolone acetonide implant (Retisert) dissociation is a common occurrence in exchange or removal procedures, or both. The longer an implant resided in the eye, the greater the tendency toward dissociation. Preparation for this complication should be contemplated in any implant removal or exchange procedure.
The fluocinolone acetonide intravitreal implant (Retisert; Bausch & Lomb, Rochester, New York, USA) is a sustained-release intravitreal implant approved by the Food and Drug Administration for the treatment of chronic noninfectious uveitis. It delivers a low dose of fluocinolone steroid (0.3 to 0.4 μg/day) for a period of approximately 3 years. The pivotal phase 3 clinical trial that established the efficacy and safety profile of the implant was conducted over a 3-year period. At 34 weeks after device implantation, the uveitis recurrence rate was just 6.4%, a significant improvement over the 54.6% recurrence rate in the 34 weeks before device implantation. The major side effects reported in the trial included cataract (93% over 3 years) and elevated intraocular pressure (67% over 3 years). The sustained-release fluocinolone acetonide intravitreal implant received Food and Drug Administration approval in the United States and became widely commercially available in 2005. Subsequently, many of the early recipients of the device, including the clinical trial patients, have had recurrence of their disease and have required reimplantation of a new device. The success of second implants in this patient population has been documented in 2 case series. Some surgeons implant a second implant at a new incision site, whereas others remove the old implant and place a new one at the same site with the idea that this lifelong disease may require numerous implants during a patient’s life and the wounds near these devices heal slowly because of the presence of a steroid.
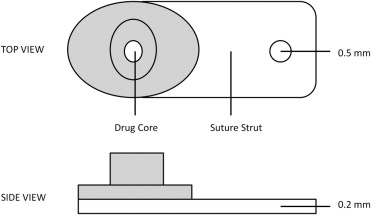
The fluocinolone acetonide intravitreal implant comprises 2 parts that are held together with a silicone adhesive ( Figure 1 ). There have been reports of dissociation during implant exchange procedures. Herein we report a series of Retisert exchange and removal procedures that shows that the implants have a tendency to dissociate into their 2 constituent parts when they are removed from an eye (herein referred to as implant dissociation). We undertook the current study to document the frequency of this occurrence in our practice and to characterize patient outcomes after these events.
Methods
After obtaining Cleveland Clinic Institutional Review Board approval, a retrospective, consecutive case series of all patients who underwent fluocinolone acetonide intravitreal implant exchange or removal procedures at the Cole Eye Institute between 2001 and 2010 was conducted. All surgeries were performed by 3 surgeons (P.K.K., R.P.S., J.E.S.). All completed surgical procedures were included for analysis. Indications for reimplantation included recurrence of uveitis 2.5 years or more after initial implantation, prophylaxis of recurrence 2.5 years or more after initial implantation, or suspicion that the implant was no longer working before 2.5 years after initial implantation. Fluocinolone acetonide intravitreal sustained-release implants with the brand names Envision (product name used during initial clinical trials; Bausch and Lomb, Rochester, New York, USA) and Retisert (Bausch and Lomb) were included in the analysis.
Surgical Procedure for Implant Exchange or Removal
The procedure for primary implantation of a fluocinolone acetonide intravitreal sustained-release implant has been described previously. Written informed consent was obtained from each patient before surgery. After placement of a 25-gauge infusion cannula 3.5 mm posterior to the limbus in the inferonasal quadrant, the old implant was accessed through the previous pars plana sclerotomy. All of the old scleral wound sutures were removed except for the anchoring suture around the fluocinolone acetonide intravitreal sustained-release implant. The old surgical wound was reopened with a 15-degree Supersharp blade (Accutome Inc, Malvern, Pennsylvania, USA) around the anchoring suture larger than the original incision and the implant was grasped with a forceps on the strut before the anchoring suture was cut. The fluocinolone acetonide intravitreal sustained-release implant then was removed from the eye as the wound was held open with forceps to avoid having the wound sheer the device. The old implant was always removed before placement of a new one. Some eyes underwent additional procedures as part of a combined surgery, including complete pars plana vitrectomy with or without membrane peel. After removal of the old implant, the new implant was placed through the same incision as described previously.
Statistical Analysis
The following clinical data were collected for analysis: presence or absence of intraoperative implant dissociation; residence time of the old implant; surgeon experience (number of cases performed); intraocular pressure; previous eye surgeries; surgeries or procedures, or both, required after fluocinolone acetonide intravitreal sustained-release device implantation; intraocular pressure; Snellen visual acuity; and other surgical complications. Snellen visual acuity was converted to logarithm of the minimal angle of resolution (logMAR) units for statistical analysis. Change in visual acuity was assessed further as 3-line acuity losses or gains. A 2-tailed t test was used in the analysis of implant time in the eye and surgeon experience. The Fisher exact test was used to compare the need for additional surgery in the dissociation and nondissociation groups, to compare the presence of pre-existing glaucoma surgery in the 2 groups, and to compare dissociation rates between Retisert and Envision implants.
Results
Three surgeons performed 27 fluocinolone acetonide intravitreal sustained-release device exchange or removal surgeries in 20 eyes of 19 patients. One surgeon performed 23 of 27 cases. The mean follow-up was 6.8 years (range, 6 to 118 months). Of the 27 surgeries, 25 (93%) were implant exchanges and 2 (7%) were implant removals. The mean patient age was 53.2 years (range, 29 to 77 years). The mean pre-exchange or removal visual acuity was 0.52 logMAR (approximately 20/66 Snellen; range, 20/20 to 3/200 Snellen). Preoperative visual acuity in the dissociation group was a mean 0.59 logMAR (approximately 20/78 Snellen) and 0.46 logMAR (approximately 20/58 Snellen) in the no-dissociation group ( P = .44). Eleven Envision implants from the early clinical trials were exchanged (n = 9, 33%) or removed (n = 2, 7%) among the 27 cases. At the time of implant exchange or removal, 17 eyes (85%) had undergone a glaucoma procedure before surgery, with 9 (33%) eyes undergoing a trabeculectomy and 8 (30%) undergoing a tube shunt. The indications for surgery were as follows: idiopathic panuveitis (8 eyes, 30%), sarcoidosis-associated uveitis (5 eyes, 19%), idiopathic posterior uveitis (3 eyes, 11%), multiple sclerosis-associated intermediate uveitis (1 eye, 4%), pars planitis (1 eye, 4%), Behçet syndrome (1 eye, 4%), and multifocal choroiditis and panuveitis (1 eye, 4%).
Of the 27 surgeries, 11 cases had dissociation of the implant strut from the drug-containing cup during surgery (40.7%). The length of time that the implant had been resident in the eye correlated significantly with cup dissociation. Dissociated implants had been resident in the eye a mean of 47.4 months (95% confidence interval, 28.0 to 66.9), whereas intact implants had been resident in the eye a mean of 32.5 months (95% confidence interval, 4.1 to 60.9; P = .0032; Figure 2 ). One implant removal in the intact group was performed at just 1 month after initial implantation because of endophthalmitis. Even when this patient was excluded, the difference between the 2 groups remained significant ( P = .0040). Implant dissociation did not correlate with surgeon experience ( P = .15; Figure 3 ). Envision implants were not significantly more likely to dislocate (6/11 [55%] Envision implants vs 5/16 [31%] Retisert implants; P = .26), and previous glaucoma surgery did not correlate with implant dissociation ( P = .69).




