Purpose
To evaluate the effectiveness of an orbital tissue expander designed to stimulate orbital bone growth in an anophthalmic socket.
Design
Retrospective, noncomparative, interventional case series.
Methods
settings: Institutional. study population: Nine consecutive patients with unilateral congenital anophthalmos. intervention: The orbital tissue expander is made of an inflatable silicone globe sliding on a titanium T-plate secured to the lateral orbital rim with screws. The globe is inflated by a transconjunctival injection of normal saline through a 30-gauge needle to a final volume of approximately 5 cm 3 . Computed tomography scans were used to determine the orbital volume. The data studied were: demographics, prior orbital expansion procedures, secondary interventions, orbital symmetry, and implant-related complications. main outcome measures: The primary outcome measure was the orbital volume change, and the secondary outcome measures were changes in forehead, brow, and zygomatic eminence contour and adverse events.
Results
The average patient age at implantation was 41.89 ± 39.42 months (range, 9 to 108 months). The initial average volume of inflation was 3.00 ± 0.87 cm 3 (range, 2.0 to 4.0 cm 3 ), and the average final volume of 4.33 ± 0.50 cm 3 (range, 4.0 to 5.0 cm 3 ) was achieved. The duration of expansion was 18.89 ± 8.80 months (range, 4 to 26 months). All patients demonstrated an average increase in the orbital tissue expander implanted orbital volume of 5.112 ± 2.173 cm 3 (range, 2.81 to 10.38 cm 3 ). The average difference between the volume of the implanted and the initial contralateral orbit was 5.68 ± 2.34 cm 3 , which decreased to 2.53 ± 1.80 cm 3 at the final measurement ( P < .001, paired t test). All implants remained inflated except for 2 iatrogenic punctures at the second inflation and 1 that was the result of implant failure. All were replaced.
Conclusions
The integrated orbital tissue expander is safe and effective in stimulating anophthalmic socket bone growth.
This article describes the first clinical series of patients in whom an integrated orbital tissue expander designed to treat children with congenital anophthalmos was implanted. The incidence of anophthalmos is 0.2 to 0.6 per 10 000 births. The principal manifestations are minified eyelids with abbreviated palpebral and bulbar conjunctivae and ipsilateral hypoplasia of the bony orbit. It is well recognized that orbital volume growth parallels ocular growth and that the absence of an eye will result in noticeable hemifacial deformity. Studies have demonstrated that socket enlargement in an anophthalmic orbit is proportional to the volume implanted and that expandable orbital implants achieve better bony stimulation compared with conventional solid silicone spheres. Pressure was found to be effective in stimulating expansion of the craniofacial skeleton.
Management of the hypoplastic juvenile orbit requires simultaneous treatment of both the soft tissue hypoplasia and asymmetric bone growth. Many investigators have advocated prompt treatment of minified eyelids and the severely contracted conjunctival fornices with progressively enlarging acrylic forniceal conformers in the first few months of life. However, the process of enlarging the dimensions of the phimotic palpebral fissure and shallow fornices does not address the underlying hypoplastic bony orbit. Coordinated expansion of the hypoplastic orbital skeleton through volume replacement is an integral component of the rehabilitation.
Three types of orbital expanders are available to clinicians to treat the congenital orbital volume deficit problem: hard spherical implants, inflatable soft tissue expanders, and hydrogel expander implants. The hard implants require serial surgical exchanges of discrete and increasing size of spheres, thus incurring multiple anesthesias and repeated trauma to the conjunctiva. Despite the arduous process of serial replacements, volume disparity remains apparent, and additional expansion may be required. The inability to keep pace with the bony growth of the contralateral normal orbit will result in retardation of the ipsilateral hemiface.
Inflatable soft tissue expanders are another option, but adaptation for orbital expansion is time-consuming, invasive, and technically difficult in an infant. The principal challenges lie in controlling the direction of expansion and maintaining expander fixation within the orbit to exert sustained omnidirectional expansion pressure. Frequently, uncontrolled forward protrusion of the expander during inflation will displace the conformer or extrude early.
Hydrogel expander implants are the third option. These implantable spheres, made from either poly (2-hydroxyethyl methacrylate) or copolymers of polymethyl methacrylate and polyvinyl pyrrolidone, can absorb up to 2000% of their weight in water and can increase up to 30 times their original volume. However, as the hydrogel implant reaches its equilibrium water content, the expansion forces are reduced substantially and bony stimulation ceases. Although the hydrogel expander is an appealing alternative to the conventional expansion options, the enthusiasm is tempered by the concern of the biomaterial’s long-term fragility and friability to maintain sustained pressure in the orbit. Further, the requisite periodic exchanges to a larger-diameter hydrogel sphere to maintain pressure holds little advantage over the conventional method of serial replacement with hard spheres of known geometry.
We have designed an integrated orbital tissue expander to address the shortcomings of the conventional orbital expansion options. We conducted a proof-of-concept study in an anophthalmic feline model that convincingly demonstrated that sustained application of biomechanical force to the craniofacial skeleton by serial orbital tissue expander inflations can lead to orbital socket enlargement. The orbital tissue expander was cleared for use by the United States Food and Drug Administration in 2006 after establishing its safety and efficacy.
The primary purpose of this study was to present the methodology of orbital tissue expander implantation and to evaluate the efficacy, stability, and safety of the device in stimulating orbital bony growth in a series of patients with congenital anophthalmos. The results of a preliminary clinical study of 9 consecutive patients are presented and discussed.
Methods
Orbital Tissue Expander
The expander (Innovia LLC, Miami, Florida, USA) consists of a 9.5-mm diameter silicone rubber expandable sphere (0.5 to > 5.0 cm 3 ) that is attached to a 9.3 × 4.8-mm cylindrical titanium injection chamber. The slotted tunnel formed perpendicular to the injection chamber is coupled to the 3.0-mm wide vertical arm of a titanium T-plate. The vertical arm permits the orbital tissue expander to slide along the shaft to achieve optimal central positioning and is slotted along its length to provide needle access to the injection chamber through the T-plate ( Figure 1 , Left). The horizontal arm of the T-plate is secured to the lateral orbital rim with titanium screws, preventing the expander from displacing forward during inflation.
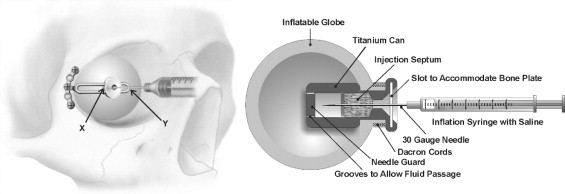
A 30-gauge needle connected to a syringe filled with sterile normal saline is inserted into the orbital tissue expander through a self-sealing injection septum housed in the titanium chamber. A deep central depression on the surface of the injection port permits detection by palpation through the overlying conjunctiva and provides a secured entry point for needle insertion. The needle guard at the back of the titanium chamber prevents inadvertent puncturing of the posterior balloon wall. Grooves along the needle guard allow fluid to pass from the injection chamber into the balloon for inflation ( Figure 1 , Right).
Clinical Study
The clinical records of all patients who had undergone orbital tissue expander implantation for correction of a hypoplastic orbit from December 2007 through May 2010 were retrieved from the Medical Records Department of the Magrabi Eye Specialist Hospital in Cairo, Egypt. Orbital computed tomography (CT) scans were obtained before the expander insertion and during each follow-up examination, when possible. The information analyzed included gender, patient age at orbital tissue expander implantation, prior orbital or forniceal surgeries, pre-expander and postexpander orbital CT volume measurements, and external appearance of the zygomatic eminence and frontal bone contour.
Orbital Tissue Expander Insertion Technique and Inflation
Orbital tissue expander insertions and postoperative assessments were performed by 2 orbital surgeons (D.T.T., M.A.). All expander placement procedures were performed with the patient under general anesthesia. A sharp dissection directed toward the lateral orbital rim was made through a 1-cm horizontal skin incision over the lateral canthus ( Figure 2 , Top left). A curvilinear periosteal incision over the lateral orbital rim was dissected off to expose a broad area of the bone to accommodate the crossbar of the T-plate. The periorbita was dissected off the lateral wall, and a 2.0-cm transverse incision was made at the mid orbit to provide a portal of entry for implant insertion. A Stevens scissor was introduced into the center of the orbit, and the blades were widened to spread the intraconal orbital tissues to create a pocket to accommodate the expander ( Figure 2 , Top right). An intact layer of conjunctiva was preserved to serve as a tissue barrier over the expander.
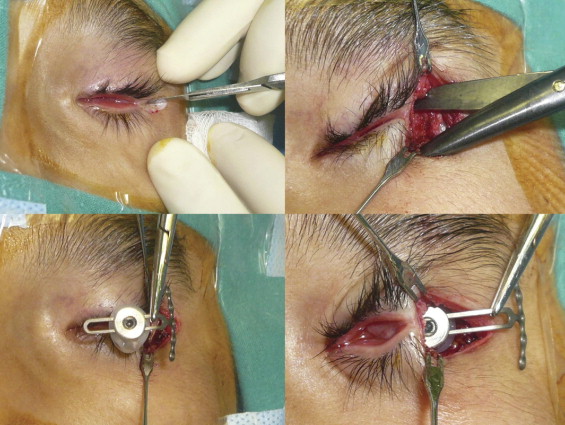
A bone plate was selected and bent 90 degrees at the junction between the crossbar and the shaft using a pair of bending pliers. The orbital tissue expander was coupled to the T-plate in the slotted tunnel. Before insertion into the orbit, the expander was inflated with sterile saline to confirm its integrity. Fluid and air were aspirated from the expander to collapse the balloon to its minimal size ( Figure 2 , Bottom left) to facilitate its insertion through the periorbital opening ( Figure 2 , Bottom right). After the implant was positioned in the center of the orbit, the crossbar of the T-plate then was maneuvered to rest over the lateral bony rim. A high-speed drill with a drill bit length of 5 mm and a diameter of 0.76 mm was used to create the drill holes. The T-plate was secured into position with 3 1-mm diameter, 5-mm long screws ( Figure 3 , Top left).
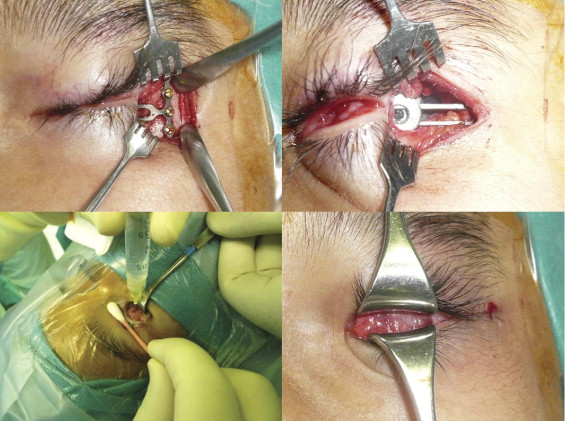
After securing the T-plate in position, a 30-gauge needle attached to a 1- to 3-mL syringe was inserted into the injection chamber under direct view of the injection port ( Figure 3 , Top right). Sterile normal saline was injected slowly to inflate the expander until the T-plate began to bend outward ( Figure 3 , Bottom left). In most cases, the expander was inflated with 3.0 cm 3 fluid, which is equivalent to the insertion of an 18-mm static acrylic implant ( Table 1 ). To verify that the injected volume was retained, the thumb over the plunger was released, allowing the backpressure in the expander to raise the plunger and refill the syringe with approximately the same amount of fluid volume injected. The periosteum was closed over the cross bar of the T-plate with 5-0 polyglactin sutures. The skin incision was closed with absorbable sutures ( Figure 3 , Bottom right).
| Injected Volume (mL) | Anteroposterior Diameter (mm) | Equatorial Diameter (mm) |
|---|---|---|
| 0.0 | 11.8 ± 0.1 | 9.5 ± 0.1 |
| 0.5 | 14.8 ± 0.4 | 11.3 ± 0.1 |
| 1.0 | 16.7 ± 0.4 | 13.5 ± 0.4 |
| 1.5 | 17.9 ± 0.4 | 15.1 ± 0.3 |
| 2.0 | 19.0 ± 0.4 | 16.6 ± 0.4 |
| 2.5 | 20.0 ± 0.5 | 17.7 ± 0.3 |
| 3.0 | 20.8 ± 0.4 | 18.7 ± 0.2 |
| 3.5 | 21.6 ± 0.6 | 19.6 ± 0.4 |
| 4.0 | 22.4 ± 0.5 | 20.4 ± 0.3 |
| 4.5 | 23.1 ± 0.6 | 21.2 ± 0.3 |
| 5.0 | 23.7 ± 0.4 | 21.9 ± 0.4 |
Implant Inflation Protocol
The inflation volumes and intervals were under the discretion of the surgeon. The second inflation was given 4 to 6 months after implantation and performed under strict sterile protocol. The inflations continued until a final volume of 4 to 5 cm 3 was reached or until the T-plate began to bend forward. Care was taken to avoid overinflation, causing pressure spikes, as well as underinflation and thereby requiring unnecessary follow-up visits.
Orbital Volume Measurement
Orbital CT scans with axial, coronal, and sagittal views were obtained. To minimize ionizing radiation risk in this pediatric cohort, all orbital CT imaging studies were performed while adhering to the as-low-as-reasonably-achievable principle by implementing the following safety measures: dose modulated by reducing the milliamps (mA) and peak kilovoltage (kvp) settings to the minimum, limiting the number of examinations for each case to the minimum, targeting only the orbital region, and adjusting the slice thickness and proper tube speed. Highest-quality images were not required, and lower-resolution scans usually were adequate in assessing the bone induction effect from orbital expansion. All CT volume measurements were obtained independently by the same radiologist (A.O.A.), who traced the orbital boundaries in consecutive transverse CT slices. Calculation of orbital volumes was performed by adding the cross-sectioned areas according to the summation of area method using standard workstation software (Philips Brilliance 64 version 4.0.2 [Philips Healthcare, Amseterdam, Netherlands], and Toshiba Aquilion 64 MDCT version 3.20 ER005-TSX 101 [Toshiba America Medical Systems, Inc. Tustin, California, USA]). The duration of expansion was designated as the date of orbital tissue expander implantation to the latest follow-up examination.
Statistical Analyses
Paired t tests were used to compare volumes of the orbits with the orbital tissue expander with their contralateral orbits and to compare the baseline volume with the final volume of each orbit. Paired t tests also were used to compare the increases in volume between the implanted orbits and the corresponding contralateral orbits during the follow-up period. A P value < .05 was considered statistically significant.
Results
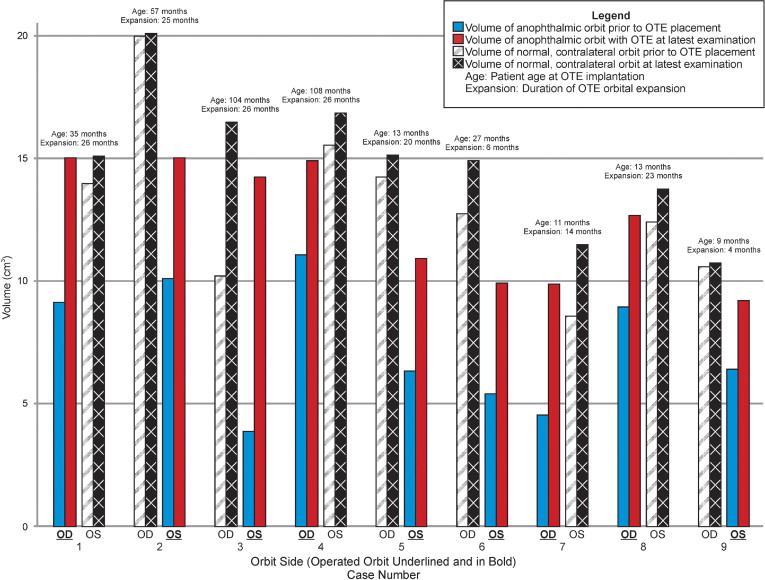
Nine patients with unilateral congenital anophthalmos underwent orbital tissue expander implantation ( Table 2 ). Of these, 4 were female and 5 were male, indicating no statistical differences in gender in the frequency of the disease during the study ( P = 1.00, McNemar test). Similarly, there were 4 right orbits and 5 left orbits affected, implying no statistical favoring of one orbit over the other ( P = 1.00, McNemar test). The average age at the time of expander implantation was 42 months (range, 9 to 108 months). Four patients had 3 prior acrylic implants and 1 patient had 8 implants or fornix and eyelid reconstructive procedures. Only 4 patients were implanted with the expander as the first socket expansion procedure.
| Case No. | Gender | Affected Orbit | Age (mos) | Previous Orbit or Lid Procedures |
|---|---|---|---|---|
| 1 | F | Right | 35 | 3 |
| 2 | M | Left | 57 | 3 |
| 3 | M | Left | 104 | 0 |
| 4 | F | Right | 108 | 8 |
| 5 | F | Left | 13 | 3 |
| 6 | M | Left | 27 | 0 |
| 7 | F | Right | 11 | 0 |
| 8 | M | Right | 13 | 3 |
| 9 | M | Left | 9 | 0 |
| Average | 41.89 | 2.22 | ||
| Standard deviation | 39.42 | 2.64 |
Initial inflation was stopped at the closest whole number, and the number of subsequent injections and total volume injected are shown in Table 3 . At the time of this writing, 6 of the 9 patients received 1 subsequent inflation within 6 months of the initial implantation, and none had more than 1 subsequent inflation. The mean initial fluid volume was 3.0 ± 0.9 cm 3 , and the final fluid inflation volume was 4.3 ± 0.5 cm 3 . The duration of expansion ranged from 4 months to 26 months, with an average follow-up period of 18.9 ± 8.8 months.
| Case No. | Initial Fluid Volume (cm 3 ) | Subsequent Inflations | Final Fluid Volume (cm 3 ) | Duration of Expansion (mos) | Orbital Volume before Surgery (cm 3 ) | Orbital Volume after Surgery (cm 3 ) | Anophthalmic Orbit-to-Control Orbital Volume Ratio c | Complications | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Anophthalmic Orbit | Control a | Difference b | Anophthalmic Orbit | Control a | Difference b | Before Surgery | After Surgery | ||||||
| 1 | 3.0 | 1 | 5.0 | 26 | 9.16 | 14.01 | 4.85 | 15.03 | 15.12 | 0.09 | 0.65 | 0.99 | Iatrogenic puncture; OTE replaced |
| 2 | 3.0 | 1 | 5.0 | 25 | 10.10 | 20.01 | 9.91 | 15.03 | 20.12 | 5.09 | 0.50 | 0.75 | Iatrogenic puncture; OTE replaced |
| 3 | 2.0 | 1 | 4.0 | 26 | 3.88 | 10.24 | 6.36 | 14.25 | 16.50 | 2.25 | 0.38 | 0.86 | T-plate repositioned |
| 4 | 2.0 | 1 | 5.0 | 26 | 11.07 | 15.54 | 4.47 | 14.91 | 16.85 | 1.94 | 0.71 | 0.88 | None |
| 5 | 2.0 | 1 | 4.0 | 20 | 6.35 | 14.27 | 7.92 | 10.93 | 15.14 | 4.21 | 0.44 | 0.72 | None |
| 6 | 4.0 | 0 | 4.0 | 6 | 5.39 | 12.76 | 7.37 | 9.92 | 14.91 | 4.99 | 0.42 | 0.67 | None |
| 7 | 3.0 | 1 | 4.0 | 14 | 4.54 | 8.57 | 4.03 | 9.90 | 11.50 | 1.60 | 0.53 | 0.86 | None |
| 8 | 4.0 | 0 | 4.0 | 23 | 8.95 | 12.43 | 3.48 | 12.67 | 13.77 | 1.10 | 0.72 | 0.92 | Implant leak; OTE replaced |
| 9 | 4.0 | 0 | 4.0 | 4 | 6.42 | 10.58 | 4.16 | 9.23 | 10.73 | 1.50 | 0.61 | 0.86 | None |
| Average | 3.00 | 0.67 | 4.33 | 18.89 | 7.32 | 13.16 | 5.84 | 12.43 | 14.96 | 2.53 | 0.55 | 0.84 | |
| Standard Deviation | 0.87 | 0.50 | 0.50 | 8.80 | 2.57 | 3.38 | 2.18 | 2.46 | 2.83 | 1.80 | 0.13 | 0.10 | |
b Volume difference between the anophthalmic orbit and the contralateral control orbit.
c A ratio of 1.0 implies the volume of the anophthalmic and control orbits are identical.
At the most recent examination, all implants remain inflated. Additional inflations still may be required in 6 of the 9 patients in which the optimal expander target volume of 5.0 cm 3 had not been reached and orbital volume disparity by CT evaluation remains apparent. The final orbital volume measurements were collected at a median duration of 25.0 months with a range of 13.7 to 25.5 months after initial implant insertion.
For 8 of the 9 patients, the initial CT measurement was acquired before surgery. For these patients, the median interval from initial measurement to surgery was 2.5 months, with a range of 0 to 8.3 months. One patient underwent the initial measurement 16 months after the initial surgery, and the final measurements were obtained 22.4 months after the surgery.
At the most recent follow-up examination, all patients had an increase in the orbital volume after expander implantation ( Figure 4 ). The average volume of the anophthalmic orbit before surgery was 7.32 ± 2.57 cm 3 , which was 5.84 cm 3 less than that of the contralateral orbit ( P < .001, paired t test). The placement and inflation of the orbital tissue expander until final examination resulted in an average growth of the affected orbit of 5.11 ± 2.17 cm 3 , compared with 1.80 ± 1.89 cm 3 in the contralateral normal orbit ( P < .001), reducing the difference between orbits to 2.53 cm 3 ( P = .003; Table 4 ). The average expander-implanted orbital volume as a percent of contralateral orbital volume increased from an initial 55% to 84% at latest follow-up examination ( P < .001; Table 4 ).