Purpose
To analyze graft survival of endothelial keratoplasty (EK) under a previous failed penetrating keratoplasty (PK) compared to repeat PK.
Design
Retrospective, comparative case series.
Methods
Analysis involved consecutive patients who underwent either a repeat PK or EK under PK, after failed PK, whose primary surgical indication was pseudophakic bullous keratopathy at a single tertiary center. Clinical data and donor and recipient characteristics were recorded from our prospective cohort from the Singapore Corneal Transplant Study. Main outcome measure was graft survival up to 5 years follow-up.
Results
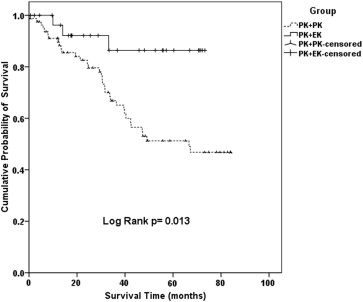
We included a total of 113 eyes that underwent a repeat PK (n = 81) or EK under a failed PK (n = 32). Cumulative graft survival probabilities comparing repeat PK with EK under PK were at 91.9% vs 96.2% (1 year), 82.6% vs 91.6% (2 years), 66.8% vs 86.4% (3 years), and 51.3% vs 86.4% up to 5 years follow-up, respectively (log-rank P value = .013). Multivariate Cox regression analysis was performed, which adjusted for: age, sex, risk factors for graft failure (corneal neovascularization, ocular surface disease, glaucoma, active corneal inflammation, anterior synechiae), donor endothelial cell count, and repeat donor size. Repeat PK was a significant risk factor for graft failure compared to performing an EK under PK (hazard ratio: 10.17; 95% CI 1.10–93.63; P = .041).
Conclusion
In our study of eyes with bullous keratopathy, endothelial keratoplasty under a previously failed PK provided better graft survival outcomes than repeat PK, adjusting for potential confounders and risk factors for graft failure.
Endothelial keratoplasty (EK) is steadily replacing penetrating keratoplasty (PK) for the treatment of endothelial disease leading to corneal decompensation. The main advantages of EK over PK include rapid postoperative visual rehabilitation and less allograft rejection. Despite these advantages, most studies have suggested that graft survival between PK and EK are actually similar (though few have been direct comparative studies). Nonetheless, after a failed primary PK, a surgeon may now choose to selectively replace the endothelium by performing an EK under the existing PK, instead of the traditional repeat PK, which has been shown to have poor long-term survival rates of 21%–70%. As EK has been shown to potentially have a lower rejection risk compared to PK, one advantage of performing EK over PK would be the potential for a reduced risk of rejection, as compared to a repeat PK.
Initial studies have found graft survival rates of EK under a failed PK to be around 55%–100% at 1 year. However, many of these studies were noncomparative case series, with a limited follow-up period. Of the few studies that examined long-term graft survival, Anshu and associates found a graft survival rate of 74% at 4 years (up to 96% in a subgroup of patients without previous glaucoma shunt surgery). Kitzmann and associates found a trend toward better 3-year graft survival in 7 eyes that underwent EK compared to 17 eyes that had a repeat PK (57.9% vs 68.6%, P = .507), although this result was not statistically significant (likely owing to the small sample size). In addition, the authors found no statistically significant differences in postoperative best-corrected visual acuity (BCVA) between the 2 groups at 1, 2, or 3 years, although others have reported significant improvements in postoperative BCVA in EK after failed PK. To date, there are no studies that have conclusively shown better outcomes for EK after failed PK, compared to repeat PK.
Without a direct comparative study (with an adequate sample size) from the same cohort, we are unable to support or refute claims that EK after a failed PK is indeed a useful alternative to performing a repeat PK. Therefore, we conducted this current study to better clarify the graft survival rates after repeat PK compared to EK after the first failed PK.
Patients and Methods
The Singapore Corneal Transplant Study is a prospective cohort study that tracks and records all clinical data, surgical complications, and outcomes in patients who undergo corneal transplants at the Singapore National Eye Center (SNEC). For this study, we included consecutive patients who first underwent PK for pseudophakic bullous keratopathy (PBK) at the SNEC from January 1, 1991 to December 31, 2008, and subsequently underwent either EK or a PK, after the first PK suffered from graft failure (January 1, 1993 to December 31, 2010). All surgeries were performed by the 5 corneal surgeons at our center, which included cases performed or partially performed by corneal fellows in training under direct supervision. Our study followed the principles of the Declaration of Helsinki, with ethics approval obtained from the Singapore Health Services Centralized Institutional Review Board. We reviewed patients’ demographics, clinical features, and surgical techniques. Visual acuity (VA) was measured using the Snellen VA chart and we analyzed results using logarithm of the minimal angle of resolution (logMAR) equivalent units. The Singapore Eye Bank provides all donor corneas with standard internal guidelines for PK and EK grafts and we obtained all donor information from their database, including donor endothelial cell counts. Our main outcome measure was percent graft survival up to 5 years postoperatively. Graft failure was defined as irreversible loss of optical clarity, sufficient to compromise vision for a minimum of 3 consecutive months.
Surgical Technique: Penetrating Keratoplasty
Penetrating keratoplasties were performed using a standard technique with a Hanna vacuum trephine system (Moria Inc, Antony, France), as previously described. The original failed PK was either surgically removed or excised using the Hanna trephine. A 0.25- to 0.50-mm oversized donor cornea then was punched out, endothelial side up, and sutured onto the recipient with 10-0 nylon, using either an 8-bite, 10-0 nylon double continuous running suture or a combination of a single 8-bite 10-0 nylon continuous and 8 interrupted sutures. Intraoperative astigmatic control was achieved using a microscope-mounted keratoscope. A bandage contact lens was placed at the end of the surgery, and subconjunctival dexamethasone (0.1%) (Decadron; Merck & Co Inc, Rahway, New Jersey, USA), gentamicin (14 mg/mL) (Garamycin; Schering AG, Berlin-Wedding, Germany), and cefazolin (50 mg/mL) (Ancef; GlaxoSmithKline, Research Triangle Park, North Carolina, USA) was injected (0.1 mL of each).
Surgical Technique: Endothelial Keratoplasty Under Failed Penetrating Keratoplasty
All EK surgeries were performed using a technique previously described, with slight modifications and no stripping of Descemet membrane. Donors were prepared by the surgeon using an automated lamellar therapeutic keratoplasty system (ALTK; Moria SA, Antony, France) and generally oversized by 0.5–1.0 mm over the original PK in order to cover the previous graft-host junction. The Tan EK forceps (ASICO, Westmont, Illinois, USA) or EndoGlide donor insertion forceps (Angiotech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK) was then used to pull the donor cornea through the scleral incision using nonfolding techniques: over either a Sheets glide (n = 16, BD Visitec, New Jersey, USA) or the EndoGlide donor inserter device (n = 16; Angiotech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK). An inferior peripheral iridectomy was performed through a limbal stab incision in all cases. The scleral wound and anterior maintainer paracentesis incisions were secured with 10/0 nylon interrupted sutures, and a full air tamponade (8 minutes) under slight compression was achieved with a large bubble in the anterior chamber, with removal of interface fluid using venting incisions. Similarly, subconjunctival corticosteroid and antibiotic injections, as described above, completed the procedure.
Postoperative Care
All patients received a standard postoperative topical antibiotic (levofloxacin 0.5%) and corticosteroid regime: 1 drop of topical prednisolone acetate 1% every 3 hours for a month, 4 times daily for 2 months, and tapered by 1 drop per 3 months down to 1 drop per day dosing by 1 year, and thereafter continued indefinitely. Bandage contact lenses were generally removed after re-epithelialization, usually 1–2 weeks after surgery. In selected cases, patients were also started on oral prednisolone 1 mg/kg, slowly tapered postoperatively over 2 months; and other systemic immunosuppression (oral mycophenolate mofetil [MMF] 2 g daily, or oral cyclosporin A [CsA]) was tapered to achieve therapeutic levels, at the discretion of the surgeons. These cases tended to be younger patients or patients deemed at relatively higher risk of allograft rejection, for example those with significant corneal vascularization, detailed in previous studies.
Statistical Analysis
Statistical analysis included descriptive statistics, where the mean and standard deviation (SD) were calculated for the continuous variables; while frequency distribution and percentages were used for categorical variables. Comparisons between categorical variables were conducted by the Pearson χ 2 test/Fisher exact tests and for continuous data Mann-Whitney U test to compare between repeat PK and EK under PK groups. Visual acuity (logMAR) was compared using the paired-sample t test/Wilcoxon signed rank test comparing baseline to 12 months follow-up; Kaplan-Meier survival analysis using the log-rank test was conducted to compare and determine the survival probabilities of repeat PK and EK under PK groups. The survival period of failed grafts was defined as the time between the date of surgery and recorded date of graft failure. Statistical Package for the Social Sciences version 17.0 (SPSS Inc, Chicago, Illinois, USA) was used to analyze the data. A P value <.05 has been considered statistically significant.
Results
During the study period, a total of 113 eyes met our inclusion criteria. The baseline characteristics of those eyes that subsequently underwent a repeat PK (n = 81) or EK under a failed PK (n = 32) are summarized in Table 1 . The EK grafts were generally oversized by at least 0.50–1.0 mm in size, so that the edges of the EK would pass over the graft-host interface (79% of EK under PK cases); while in some of the larger PK cases, we made the EK diameter the same (21% of cases) in the attempt to reduce the amount of peripheral folding or EK graft edge lift ( Table 1 ). There were no significant differences in immunosuppression regime in either group, either in the number of patients who were treated with MMF (repeat PK: 5/81 vs EK under PK: 1/32, P = .678) or CsA (repeat PK: 8/81 vs EK under PK: 5/32, P = .526) or in the duration of systemic immunosuppressant therapy (mean duration, MMF: repeat PK = 16.8 ± 15.6 months vs EK under PK = 13.0 ± 0.0 months, P = 1.0; CsA: repeat PK = 24.3 ± 25.6 months vs EK under PK = 10 ± 6.4 months, P = .269). The mean donor thickness for endothelial keratoplasty grafts was 160 ± 44 μm. Time between grafts was 50 ± 33 months in the repeat PK group, compared to 90 ± 48 months in the EK under PK group ( P < .001). Visual acuity was poor in both groups (baseline VA in eyes with failed PK) before the repeat PK (logMAR VA 1.8 ± 0.3) or EK (logMAR VA 1.4 ± 0.6) was performed. Visual acuity significantly improved in both groups (gain in logMAR VA, repeat PK: 0.87 ± 0.6, P < .001; EK under PK: 0.65 ± 0.7, P < .001).
| Characteristics | Corneal Grafts | P Value | ||
|---|---|---|---|---|
| Total | PK-PK | PK-EK | ||
| (N = 113) | (N = 81) | (N = 32) | ||
| Mean age, y (±SD) | 69.1 (12.7) | 70.9 (11.7) | 64.4 (14.1) | .014 |
| Sex, n (%) | ||||
| Male | 61 (54) | 47 (58) | 14 (43.8) | .170 |
| Female | 52 (46) | 34 (42) | 18 (56.3) | |
| Race, n (%) | ||||
| Chinese | 90 (79.6) | 65 (80.2) | 25 (78.1) | .533 |
| Malay | 8 (7.1) | 7 (8.6) | 1 (3.1) | |
| Indian | 6 (5.3) | 4 (4.9) | 2 (6.3) | |
| Other | 9 (8.0) | 5 (6.1) | 4 (12.5) | |
| Donor characteristics | ||||
| Donor age (y), median (range) | 59 (16–76) | 62 (17–76) | 57 (16–76) | .137 |
| Donor ECD, mean (±SD) | 2792 (380) | 2756 (392) | 2922 (367) | .039 |
| Donor diameter (mm), mean (±SD) | 8.2 (0.5) | 8.0 (0.4) | 8.6 (0.4) | <.001 |
| Second graft oversize, n (%) | ||||
| 0 mm (same size) | 40 (38.1) | 34 (44.2) | 6 (21.4) | |
| 0.25 mm | 15 (14.3) | 14 (18.2) | 1 (3.6) | |
| 0.5 mm | 23 (21.9) | 18 (23.4) | 5 (17.9) | <.001 |
| ≥0.75 mm | 27 (24.5) | 11 (14.3) | 16 (57.2) | |
| Reason for original PK failure | ||||
| Endothelial failure | 70 (61.9) | 45 (55.6) | 25 (78.1) | |
| Graft rejection | 27 (23.9) | 22 (27.2) | 5 (15.6) | |
| Glaucoma | 5 (4.4) | 4 (4.9) | 1 (3.1) | .208 |
| Infection | 6 (5.3) | 6 (7.4) | 0 | |
| Other | 5 (4.4) | 4 (4.9) | 1 (3.1) | |
Graft survival rates were significantly better in eyes that underwent EK under a failed PK, compared to the repeat PK group (cumulative survival probability 86.4% vs 51.3%, respectively, log-rank P value = .013) ( Figure ). Cumulative graft survival probabilities comparing repeat PK with EK under PK were at 91.9% vs 96.2% (1 year), 82.6% vs 91.6% (2 years), 66.8% vs 86.4% (3 years), and 51.3% vs 86.4% up to 5 years follow-up, respectively. Among the EK under PK cases only, the cumulative graft survival probability comparing those with and without prior glaucoma surgery was 77.9% vs 93.3% over 5 years (log-rank P value = .369). We then performed multivariate Cox regression analysis correcting for potential confounders and factors that were significantly different between our groups: age, sex, risk factors for failure (corneal neovascularization, ocular surface disease, glaucoma, active corneal inflammation, and peripheral anterior synechiae), donor endothelial cell count, and the repeat donor size. We found that a repeat PK was a significant risk factor for graft failure compared to performing an endothelial keratoplasty under PK (hazard ratio: 10.17; 95% CI 1.10–93.63; P = .041) on multivariate analysis ( Table 2 ).
| Hazard Ratio | 95.0% Confidence Interval | P Value b | ||
|---|---|---|---|---|
| Recipient age | 1.01 | 0.98 | 1.05 | .508 |
| Recipient sex (male) | 2.23 | 0.93 | 5.33 | .072 |
| Risk factors a | ||||
| Corneal vascularization | 0.11 | 0.02 | 0.59 | .011 |
| Glaucoma | 1.44 | 0.62 | 3.33 | .392 |
| Active inflammation | 1.00 | 0.00 | 1.00 | .979 |
| Severe ocular surface disease | 1.63 | 0.18 | 14.56 | .660 |
| Peripheral anterior synechiae | 0.49 | 0.16 | 1.56 | .229 |
| Donor endothelial cell count | 1.00 | 0.99 | 1.01 | .381 |
| Time between grafts (mo) | 1.00 | 0.99 | 1.01 | .745 |
| Donor size (mm) | 0.43 | 0.13 | 1.40 | .161 |
| Repeat PK (compared to PK-EK) | 10.17 | 1.10 | 93.63 | .041 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


