Purpose
To assess tear cytokine levels and clinical outcomes in moderate and severe meibomian gland dysfunction (MGD) after 2 months of treatment with topical loteprednol etabonate and eyelid scrubs with warm compresses vs eyelid scrubs with warm compresses alone.
Design
Randomized controlled trial.
Methods
Patients with moderate and severe MGD were randomized into 2 groups: topical loteprednol etabonate and eyelid scrubs with warm compresses (Group I, 34 eyes) or eyelid scrubs with warm compresses (Group II, 36 eyes). We evaluated cytokine levels, tear film break-up time (TBUT), corneal and conjunctival fluorescein staining, biomicroscopic examination of lid margins and meibomian glands, and the Ocular Surface Disease Index before initiating treatment and 1 month and 2 months after treatment.
Results
There were significant decreases in the levels of interleukin (IL)-6, IL-8, and IL-1β in Group I, and IL-6 and IL-8 in Group II. Moreover, the observed decreases of these cytokines in Group I were attributed to a remarkable decrease between treatment and 1 month after treatment. In Group I, there were improvements in all of the clinical outcomes, with prominent improvement in TBUT, corneal and conjunctival fluorescein staining, and meibum quality after 1 month of treatment, compared with Group II. An improvement in meibomian gland expressibility and MGD stage reduction were more remarkable in Group I.
Conclusions
Topical loteprednol etabonate and eyelid scrubs with warm compresses were tolerated and efficacious for the treatment of moderate and severe MGD. We suggest that such beneficial effects could manifest after 1 month.
Normal meibum lipids act as a barrier by preventing the evaporation and contamination of the tear film and by maintaining lubrication across the ocular surface. Meibomian gland dysfunction (MGD) is a prevalent condition and one of the major causes of dry eye syndrome. MGD is a chronic, diffuse abnormality of the meibomian glands and is commonly characterized by terminal duct obstruction and changes in glandular secretion. Modified and deficient meibum lipids result in tear instability, evaporative dry eye, and eyelid inflammation, which are all commonly detectable signs of MGD.
Many investigators have reported that the chronic inflammatory status in patients with MGD is associated with high concentrations of tear cytokines. One study compared inflammatory tear cytokine levels between MGD patients and normal controls and found that concentrations of interleukin (IL)-6 and pro-matrix metalloproteinase (MMP)-9 were significantly higher in the MGD patients. Higher concentrations of IL-6, IL-8, IL-12, interferon-γ (IFN-γ), and tumor necrosis factor-α (TNF-α) were reported in dysfunctional tear syndrome with MGD. Both IL-1β and IL-17 levels were elevated in tears from MGD patients. Based on cross-sectional analysis of tear cytokine levels, a longitudinal evaluation of changes in tear cytokine levels before, during, and after treatment would be helpful in demonstrating the efficacy of treatment and in determining an effective time point for achieving treatment response.
Eyelid management, including warm compresses and lid scrubs, has been known to be a conservative and traditional treatment modality for MGD. It is thought to improve meibomian gland function and ocular comfort by melting and releasing the abnormally modified meibum. However, eyelid scrubs with warm compresses alone are insufficient to modulate the inflammatory process in moderate and severe MGD. Thus, eyelid management needs to be supported by additional treatment to achieve satisfactory and quick responses. Systemic tetracycline, doxycycline, and minocycline have been effective in treating moderate to severe MGD through their anti-inflammatory, antimetalloproteinase, and antiapoptotic properties. Topical azithromycin, a macrolide antibiotic with presumed anti-inflammatory effects, has been reported to be effective for MGD. Both topical cyclosporine and diquafosol have also shown promising results in treating MGD.
Loteprednol etabonate, a novel C 20 ester-based corticosteroid, was retrometabolically designed to provide potent anti-inflammatory efficacy, but with decreased impact on intraocular pressure (IOP). After exerting its therapeutic effects at the site of action, it is rapidly converted to inactive metabolites, thereby resulting in fewer adverse effects. Several clinical studies evaluating the efficacy of 0.5% loteprednol etabonate ophthalmic suspension (Lotemax; Bausch and Lomb Inc, Rochester, New York, USA) in patients with acute anterior uveitis, giant papillary conjunctivitis, seasonal allergic conjunctivitis, postoperative inflammation, and ocular pain showed efficacy of this medication. In addition, many investigators reported that topical loteprednol etabonate treatment is associated with a relatively lower chance of clinically significant increases in IOP (10 mm Hg or higher).
Based on the known efficacy and safety of topical loteprednol etabonate, we evaluated the effect of this medication in combination with eyelid scrubs with warm compresses on changes of IL-6, IL-7, IL-8, IL-1β, IL-17α, IL-12p70, monocyte chemotactic protein-1 (MCP-1), TNF-α, and IFN-γ levels and clinical outcomes in patients with moderate and severe MGD, comparing before treatment, 1 month after treatment, and 2 months after treatment. Furthermore, we compared those results with a control group treated with eyelid scrubs with warm compresses alone.
Methods
This randomized controlled trial was approved prospectively by the Institutional Review Board of Severance Hospital, Yonsei University College of Medicine (Seoul, South Korea) and conducted according to the Declaration of Helsinki and Good Clinical Practices. Informed consent was obtained from all patients after explanation of the purpose and possible consequences of the study.
Inclusion criteria included patients with moderate and severe MGD. MGD was diagnosed by evidence of lid margin or tarsal conjunctival erythema, bulbar conjunctival hyperemia, telangiectasia, thickening, irregularity of the eyelid margins, or meibomian gland orifice inclusions. The stage of MGD was assessed by evaluating conjunctival inflammation, clinical symptoms, fluorescein corneal and conjunctival staining, and clinical signs, including lid margin abnormality, expressibility, and altered secretion. Exclusion criteria included a history of previous ocular or intraocular surgery, glaucoma or ocular hypertension, ocular infection, non–dry eye ocular inflammation, ocular allergy, autoimmune disease, history of intolerance or hypersensitivity to any component of the study medications and to other corticosteroids, wearing contact lenses during the study period, presence of current punctal occlusion, pregnancy, lactating women, and children. Additionally, patients were excluded if they were using any topical ocular or systemic medication for treatment of MGD or dry eye syndrome, including topical or oral antibiotics, topical cyclosporine A, topical or oral steroids, topical nonsteroidal anti-inflammatory drugs, topical ocular allergy medications, or artificial tears. Seventy patients who met the inclusion criteria were enrolled consecutively from the Severance Eye and ENT Hospital, Yonsei University College of Medicine, Seoul, South Korea, between August 1, 2012 and March 31, 2013. After a wash-out period of 2 weeks for patients using any other topical or systemic medication, enrolled patients were allocated randomly into 2 groups ( Figure 1 ). A randomization sequence was created using EXCEL 2007 (Microsoft, Redmond, Washington, USA) with a 1:1 allocation using random block sizes of 2, 4, and 6, by an independent doctor. The allocation sequence was concealed from the physician enrolling and assessing patients in sequentially numbered, opaque, and sealed envelopes. After the content of the envelope was revealed, the physician and patients were aware of the allocation and the corresponding treatment. However, outcome assessors and data analysts were kept masked to the allocation. In Group I, 34 eyes of 34 patients topically received loteprednol etabonate 4 times a day following eyelid scrubs with warm compresses 2 times a day for 2 months. In Group II, 36 eyes of 36 patients received eyelid scrubs with warm compresses 2 times a day for 2 months. Four patients (4 eyes) in Group I and 6 patients (6 eyes) in Group II were lost to follow-up. Measurements of the remaining 60 eyes of 60 patients were used for statistical analysis.
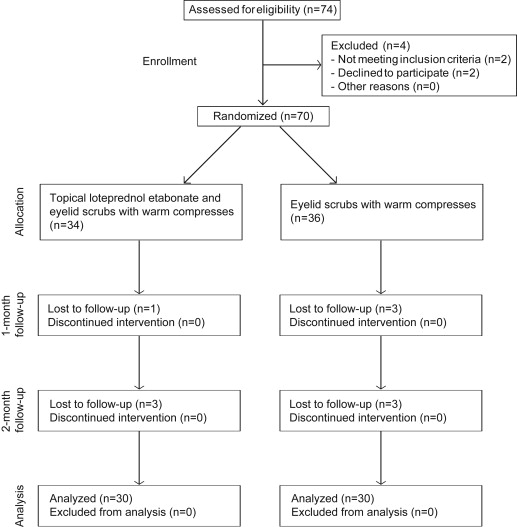
The outcome assessments were performed before treatment, after 1 month of treatment, and after 2 months of treatment by 1 physician masked to group assignments. The study eye was chosen to be the eye having a higher stage of MGD. If the MGD stage for each eye was equal, the right eye was enrolled as the test eye. To minimize the extent to which 1 test influenced the results of the tests that followed, each test was performed in the same order. Tear collection was performed first, followed by the biomicroscopic examination of TBUT, corneal and conjunctival fluorescein staining, IOP measurement, examination of lid margins and meibomian glands, and the Ocular Surface Disease Index (OSDI) questionnaire. Patients were instructed to perform standard eyelid management via face-to-face education at every follow-up time and were instructed not to wipe or scrub their eyelid margins on the day of tear sampling.
For tear cytokine analysis, 30 μL of phosphate-buffered saline was injected into the inferior conjunctival sac using a micropipette. Approximately 20 μL of tear fluid in buffer was collected with a micropipette. In order to minimize irritation of the ocular surface or lid margin, unstimulated tear fluid was collected from the marginal tear strip of the lower lid near the lateral canthus. Anesthetic drops were not administered. Tear samples were immediately transferred to 0.5 mL Eppendorf tubes (Eppendorf, Fremont, California, USA) and placed on dry ice. The tubes were kept in a −70°C freezer until they were used for the immunoassay. Cytokines were measured using the BD Cytometric Bead Array (BD Bioscience, San Jose, California, USA). The cytokines analyzed were IL-6, IL-7, IL-8, IL-1β, IL-17α, IL-12p70, MCP-1, TNF-α, and IFN-γ. The measurements were performed essentially as previously described. Briefly, 20 μL of tear fluid was thawed and added to a 50 μL mixture containing capture antibody–bead reagent and 50 μL detector antibody–phycoerythrin reagent. The mixture was subsequently incubated for 3 hours at room temperature and washed to remove unbound detector antibody–phycoerythrin reagent before flow cytometry. Data were acquired and analyzed using BD Cytometric Bead Array software that calculated the cytokine concentration based on standard curves and a 4 parameter logistic curve–fitting model. Flow cytometry was performed using the BD LSRII system (BD Bioscience). The lower limits of detection were the following: IL-1β, 2.3 pg/mL; IL-6, 1.6 pg/mL; IL-7, 0.5 pg/mL; IL-8, 1.2 pg/mL; IL-12p70, 0.6 pg/mL; IL-17α, 0.3 pg/mL; IFN-γ, 1.8 pg/mL; TNF-α, 0.7 pg/mL; and MCP-1, 1.3 pg/mL. The lowest cytokine concentration in the linear portion of the standard curve was used for statistical comparison between tear samples with concentrations below this level.
To measure TBUT, a drop of nonpreserved saline solution was added to a fluorescein strip (Haag-Streit, Koeniz, Switzerland) that was applied to the inferior palpebral conjunctiva. The patients were instructed to blink 3 or 4 times for a few seconds to ensure adequate mixing of the dye, then the eye was examined using a slit lamp with maximum cobalt blue light. The patients were asked to open their eye widely and look straight ahead, and the physician used a stop watch to measure the time it took for a single black dot or line to appear on the cornea. The test was performed 3 times and the results were averaged. Staining scores were obtained by adding together the following scores of the exposed cornea and conjunctiva: Oxford staining score (range: 1–15) and Dry Eye Workshop staining (DEWS) score (range: 0–33). IOP measurement was performed using a noncontact tonometer (NT-530; NCT Nidek Co, Ltd, Aichi, Japan). The test was performed 3 times and the results were averaged. Patients showing clinically significant increases (10 mm Hg or more) of IOP over baseline were not allowed to use topical loteprednol etabonate, during which careful follow-up of IOP was performed.
Microscopic examination of the lid margins and meibomian glands was performed last, because this examination can affect TBUT, ocular surface staining, and IOP measurements. The physician scored for the presence or absence of the following lid margin abnormalities: lid margin irregularity, plugging of the meibomian orifices, lid margin vascular engorgement, and anterior or posterior replacement of mucocutaneous junction. The degree of expressibility, using firm digital pressure applied over 5 lower lid glands, was based on the following: grade 0, all 5 glands expressible; grade 1, 3–4 glands expressible; grade 2, 1–2 glands expressible; and grade 3, zero glands expressible. The degree of meibum quality, using firm digital pressure applied over 8 lower lid glands, was based on the following: grade 0, clear; grade 1, cloudy; grade 2, cloudy with granular debris; and grade 3, thick like toothpaste. Each of the 8 glands of the lower eyelid was graded on a scale from 0 to 3. The scores of the 8 glands were summed to obtain a total score (the maximum score for each eye was 24).
All patients completed the validated 12-item OSDI questionnaire that is assessed on a scale from 0 to 100, with higher scores representing greater disability. The total OSDI score was calculated using the following formula: OSDI score = (sum of scores for all questions answered × 100)/(total number of questions answered × 4). The patients were also asked to rate subjective symptoms (ocular irritation symptom score: ocular discomfort, itching, and photophobia with limitations of activities) on a scale of 0 (no symptoms) to 3 (severe symptoms). Safety was assessed by monitoring any adverse events during the entire course of study.
Statistical Analysis
The primary outcome measure of this study was inflammatory tear cytokine levels at 2 months after treatment. The secondary outcome measures included data on both clinical tests and patient-reported outcomes. Based on the analysis of repeated IL-6 levels using a compound symmetry covariance pattern model, when given parameters were adopted (α error 0.05, power 80%, and number of repeated measurements 3), 25 eyes for each group were required. Adjusting for a drop-out rate of 20%, a minimum of 32 patients was to be enrolled in the study.
A linear mixed model with post hoc analysis was used to evaluate possible differences between the 2 groups and 3 time courses in the measurement of cytokine levels and clinical outcomes, all with the unstructured covariance matrix. In the presentation of noncontinuous scale values, such as expressibility, the ocular irritation symptom score, and the MGD stage, we performed the generalized estimating equations model. To compare the age and IOP between Group I and Group II before treatment, an independent t test analysis was used. The χ 2 test was used to compare sex and laterality between groups. The data were analyzed according to intention-to-treat principle, and data analysts were masked to the allocated treatment.
Statistical analysis was performed using SAS software (version 9.2; SAS Institute, Inc, Cary, North Carolina, USA). Differences were considered statistically significant when the P values were less than .05.
Results
The patients’ characteristics are summarized in Table 1 . No significant differences in any parameters were found between groups before treatment. The flow of patients through the study is shown in Figure 1 . Table 2 shows comparisons of tear cytokine concentrations before and after treatment in both groups. In the linear mixed model, regarding the interaction effect between the 2 groups and 3 time courses, there were statistically significant differences in the measurement of IL-6, IL-8, and MCP-1 ( P = .030 for IL-6, P = .037 for IL-8, and P = .017 for MCP-1, Table 2 ). The mean IL-6 level in Group I and Group II showed a significant decrease over time (adjusted P = .014 for Group I and adjusted P = .004 for Group II). When comparing before treatment with 1 month after treatment in both groups, the mean IL-6 level in Group I showed a significant decrease, whereas that in Group II did not show a significant decrease (adjusted P = .007 for Group I and adjusted P = .525 for Group II). At 1 month after treatment, there was a lower level of IL-6 in Group I compared with that in Group II, although not statistically significant (adjusted P = .088, raw P = .029, Figure 2 ). The mean IL-8 and IL-1β levels in Group I decreased significantly over time (adjusted P = .001 for IL-8 and adjusted P = .030 for IL-1β). Both cytokine levels in Group I, when comparing before treatment with 1 month after treatment, showed a significant decrease, whereas those in Group II did not show any changes (adjusted P = .004 for IL-8 in Group I and adjusted P = .022 for IL-1β in Group I). At 1 month, the level of IL-8 was significantly lower in Group I than in Group II (adjusted P = .034, Figure 2 ). The mean IL-8 level in Group II also significantly decreased after 2 months of treatment (adjusted P < .001). In Group I, there was a tendency toward a lower level of IL-7 over time (adjusted P = .068). At 2 months after treatment, the levels of IL-7 were significantly lower in Group I than in Group II (adjusted P = .030, Figure 2 ). For IL-17α, MCP-1, TNF-α, IL-12p70, and IFN-γ, slight but not statistically significant decreases of tear cytokine levels were found in Group I, but no definite changes were detected in Group II.
| Group I a | Group II b | |
|---|---|---|
| Patients (eyes) | 30 (30) | 30 (30) |
| Sex | ||
| Women | 19 | 20 |
| Men | 11 | 10 |
| Laterality | ||
| Right | 19 | 18 |
| Left | 11 | 12 |
| Age (y) | ||
| Mean (SD) | 66.77 (10.13) | 67.10 (11.67) |
| Range | 46–81 | 44–81 |
| IOP (mm Hg) | ||
| Mean (SD) | 11.90 (3.11) | 12.60 (2.28) |
| Range | 5–18 | 7–16 |
a Topical loteprednol etabonate and eyelid scrubs with warm compresses.
| Group I a | Group II b | P Value c | |||||
|---|---|---|---|---|---|---|---|
| Before | 1 Month | 2 Months | Before | 1 Month | 2 Months | ||
| IL-6 (pg/m) | 76.45 (21.3) | 21.84 (8.1) | 16.71 (2.8) | 76.27 (26.5) | 50.97 (10.1) | 16.31 (3.8) | .030 |
| IL-7 (pg/m) | 12.82 (2.2) | 11.87 (2.2) | 9.52 (1.8) | 17.49 (2.7) | 19.98 (2.7) | 16.99 (2.2) | .396 |
| IL-8 (pg/m) | 1315.6 (266.1) | 436.5 (165.8) | 270.2 (66.8) | 1310.2 (327.3) | 1109.9 (195.5) | 373.2 (79.1) | .037 |
| IL-1β (pg/m) | 14.01 (3.2) | 7.38 (1.3) | 8.95 (2.0) | 14.84 (4.1) | 8.40 (1.6) | 7.08 (2.5) | .419 |
| IL-17α (pg/m) | 5.35 (1.4) | 5.04 (1.1) | 4.82 (0.9) | 6.96 (1.6) | 8.07 (1.3) | 7.85 (1.1) | .440 |
| MCP-1 (pg/m) | 212.9 (50.8) | 198.1 (38.9) | 157.8 (47.6) | 236.3 (66.6) | 256.0 (51.0) | 334.1 (62.9) | .017 |
| TNF-α (pg/m) | 6.36 (2.3) | 4.07 (1.1) | 3.94 (0.9) | 7.46 (3.0) | 4.05 (1.4) | 3.92 (1.1) | .909 |
| IL-12p70 (pg/m) | 4.08 (1.3) | 4.77 (0.9) | 5.60 (0.8) | 6.19 (1.7) | 5.93 (1.2) | 6.61 (1.0) | .837 |
| IFN-γ (pg/m) | 4.57 (1.6) | 4.42 (0.8) | 4.59 (0.9) | 7.49 (2.0) | 6.36 (1.1) | 7.19 (1.1) | .536 |
a Topical loteprednol etabonate and eyelid scrubs with warm compresses.
b Eyelid scrubs with warm compresses.
c P values from linear mixed model with post hoc analysis considering the interaction effect between the 2 groups and the 3 time courses.
The results of clinical signs and symptoms before and after treatment in both groups are shown in Table 3 . In the linear mixed model, regarding the interaction effect between groups and time courses, there were statistically significant differences in the measurement of TBUT, corneal fluorescein staining, conjunctival fluorescein staining, DEWS, the Oxford staining score, lid margin abnormality, and meibum quality ( P = .014 for TBUT, P = .012 for corneal fluorescein staining, P < .001 for conjunctival fluorescein staining, P < .001 for DEWS, P = .001 for the Oxford staining score, P = .032 for lid margin abnormality, and P < .001 for meibum quality, Table 3 ). The mean TBUT in Group I and Group II showed significant improvement over time (adjusted P < .001). Although no significant difference was found between groups at each time point, changes in TBUT in Group I were significantly larger than those in Group II when comparing before treatment with 1 month after treatment (adjusted P = .012, Figure 3 ).
| Group I a | Group II b | P Value | |||||
|---|---|---|---|---|---|---|---|
| Before | 1 Month | 2 Months | Before | 1 Month | 2 Months | ||
| TBUT (s) c | 3.33 (0.31) | 7.15 (0.41) | 9.85 (0.60) | 3.48 (0.31) | 6.10 (0.41) | 8.15 (0.60) | .014 |
| Staining score c | |||||||
| Cornea | 2.77 (0.33) | 1.30 (0.27) | 0.73 (0.26) | 2.20 (0.33) | 1.80 (0.27) | 1.40 (0.26) | .012 |
| Conjunctiva | 4.67 (0.30) | 2.63 (0.24) | 1.77 (0.24) | 3.90 (0.30) | 2.90 (0.24) | 2.33 (0.24) | <.001 |
| DEWS | 4.67 (0.30) | 2.63 (0.24) | 1.77 (0.24) | 3.90 (0.30) | 2.90 (0.24) | 2.33 (0.24) | <.001 |
| Oxford | 6.13 (0.38) | 3.43 (0.30) | 2.33 (0.33) | 5.20 (0.38) | 3.97 (0.30) | 3.10 (0.33) | .001 |
| Eyelid | |||||||
| Margin abnormality c | 2.83 (0.13) | 1.37 (0.11) | 0.37 (0.10) | 2.97 (0.13) | 1.70 (0.11) | 0.93 (0.10) | .032 |
| Meibum quality c | 17.73 (0.39) | 10.73 (0.50) | 5.07 (0.59) | 17.17 (0.39) | 13.20 (0.50) | 8.83 (0.59) | <.001 |
| Expressibility d | 30 (100) | 11 (36.67) | 1 (3.33) | 29 (96.67) | 17 (56.67) | 7 (23.33) | .019 |
| Subjective score | |||||||
| OSDI c | 47.22 (3.58) | 24.44 (3.07) | 6.94 (1.91) | 55.49 (3.58) | 28.61 (3.07) | 13.54 (1.91) | .510 |
| OIS d | 30 (100) | 11 (36.67) | 0 (0) | 30 (100) | 19 (63.33) | 6 (20) | <.001 |
| MGD stage d | 30 (100) | 5 (16.67) | 0 (0) | 30 (100) | 20 (66.67) | 6 (20) | <.001 |
| IOP | 11.90 (0.50) | 13.13 (0.42) | 13.20 (0.52) | 12.60 (0.50) | 11.80 (0.42) | 12.53 (0.52) | .002 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


