Purpose
To evaluate the effectiveness of artificial tears and corticosteroids on mitigating the acute ocular surface response to low-humidity environments.
Design
Single-group, crossover clinical trial.
Methods
Twenty subjects with aqueous deficient dry eye were enrolled. Subjects meeting inclusion criteria at visit 1 were exposed to a baseline 90-minute low-humidity environment at visit 2. They then used artificial tears for 2 weeks prior to low-humidity exposure at visit 3, followed by 0.1% dexamethasone for 2 weeks prior to the final low-humidity exposure at visit 4. Outcome measures included corneal and conjunctival staining, blink rate, and irritation symptoms before and after each low-humidity exposure. Digital polymerase chain reaction (PCR) was performed to measure HLA-DR RNA transcripts in conjunctival cells taken by impression cytology at each visit.
Results
There was significantly less corneal and conjunctival epitheliopathy after the low-humidity exposure at visit 4 compared to after the low-humidity exposure at visit 3 ( P = .003). Subjects reported significantly less eye irritation during the low-humidity exposure after using the dexamethasone (visit 4) compared to artificial tears (visit 3) ( P = .01). HLA-DR transcripts significantly decreased after the stress at visit 4 (post dexamethasone) compared to visit 2.
Conclusion
Our study demonstrates that corticosteroid eye drops mitigate the acute adverse effects of an experimental low-humidity challenge, likely owing to suppression of stress-activated inflammatory pathways. While extended use of corticosteroids is not indicated, other anti-inflammatory therapies with activity against stress-activated pathways may prove as effective.
Dry eye is a multifactorial disease of the tears and ocular surface. The tear-secreting glands and the ocular surface function as a complex integrated unit, interconnected by sensory and autonomic nerves. Dysfunction of any part of this unit results in an unstable and hyperosmolar tear film that no longer adequately supports the ocular surface. Inflammation is a consequence of this dysfunction and contributes to tear instability, corneal epithelial disease, and ocular discomfort. Thus, dry eye disease is usually accompanied by symptoms of eye irritation, visual disturbances, tear film instability, and damage to the ocular surface, which can impair quality of life.
Certain environmental factors, such as low humidity, extended computer usage, and air drafts have been shown to contribute to the development of dry eye symptoms in normal individuals, as well as worsen symptoms and signs in those with dry eye disease. This may be a result of the desiccating stress that air drafts, low humidity, and decreased blinking have on the ocular surface. This stress leads to increased tear instability, evaporation, and osmolarity, which contributes to a downstream cascade of events that promote production of inflammatory mediators (cytokines and matrix metalloproteinases), epithelial apoptosis, and loss of conjunctival goblet cells. Moreover, this desiccating stress has been shown to decrease tear production and the protective function of the tear film.
We previously demonstrated, using an environmentally controlled goggles system, that certain factors, such as low tear volume, render individuals with dry eye disease more susceptible to an ocular surface response to desiccating environmental stimuli. Thus, reduced tear production from aging, use of systemic medications with anticholinergic effects, or autoimmune conditions such as Sjögren syndrome and rheumatoid arthritis may contribute to development of environmentally induced dry eye.
Despite the adverse effects of low-humidity environments on the ocular surface, many people spend the majority of their waking hours in air-conditioned, low-humidity, and drafty environments. Low-humidity environments are ubiquitous. The 2009 Residential Energy Consumption Survey performed by the United States Energy Information Administration found that 87% of households and almost all office buildings and commercial buildings in the United States are air-conditioned. Additionally, an ambient relative low humidity of 40%–60% is recommended for office buildings. This is problematic because desiccation of the ocular surface owing to low humidity is a potent inflammatory stress. Normal individuals have the ability to recover from the adverse environmental exposure, via reflex tearing from the lacrimal glands and goblet cells. Aqueous-deficient dry eye patients, on the other hand, have decreased goblet cell number and mucus production, have reduced tear volume, and may have loss of reflex tearing owing to decreased corneal sensitivity or lacrimal gland inflammation.
Because exposure to low-humidity environments is difficult to avoid, the use of therapies to mitigate the adverse effects of desiccating environmental stimuli is critical in preventing the development and/or progression of dry eye disease. Topically applied corticosteroid, but not saline, was reported to prevent corneal barrier disruption in response to experimental low-humidity environmental challenge in mice. A significant decrease in superior corneal dye staining was noted after exposure to a controlled adverse environment in subjects treated with dexamethasone delivered by iontopheresis. The efficacy of topical corticosteroid in preventing worsening ocular surface epithelial disease in dry eye patients subjected to a low-humidity environment has not been previously evaluated. The purpose of this study was to evaluate the effectiveness of 2 therapies, artificial tears and corticosteroid eye drops, on mitigating the acute ocular surface response to low-humidity environments. The study was designed to sequentially evaluate the effects of artificial tears followed by a corticosteroid because most dry eye patients initially treat environmentally induced irritation with over-the-counter (OTC) artificial tears.
Methods
Study Design
We performed a prospective, single-group, crossover clinical trial to determine the effectiveness of artificial tears, which hydrate and lubricate the ocular surface, and corticosteroid eye drops, which inhibit inflammation, on mitigating the adverse effects of a short-term low-humidity desiccating environment on the severity of irritation symptoms and ocular surface epithelial disease. The study was conducted in accordance with the Declaration of Helsinki, and the Baylor College of Medicine Institutional Review Board approved the protocol and informed consent form prior to study initiation. The trial was also registered with clinicaltrials.org (#NCT01797822).
Twenty subjects were enrolled in the study after providing written informed consent. Subjects were selected based on the following inclusion criteria: inferior tear meniscus height of ≤230 μm as a measure of low tear volume; corneal and conjunctival dye staining score of ≥3 in at least 1 eye; normal corneal sensitivity (score ≥5 measured with the Cochet-Bonnet esthesiometer); absence of other ocular surface disease, including anterior blepharitis, meibomian gland disease, conjunctivochalasis, and conjunctival scarring; tear break-up time (TBUT) <7 seconds; and Ocular Surface Disease Index (OSDI) symptom score >20. Patients were excluded if they had a corneal transplant, retinal detachment, or glaucoma filtering surgery in the past; cataract or LASIK surgery within 12 months of study enrollment; diabetes mellitus; or uncontrolled systemic infection or inflammation, or used prescription eye drops within 4 weeks of study enrollment.
Experimental Low-Humidity Environment
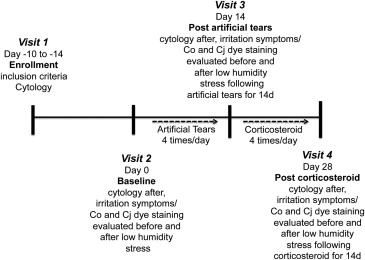
The study design is summarized in Figure 1 . Subjects who met inclusion criteria were enrolled at visit 1 and returned 10–14 days later to be subjected to a baseline controlled low-humidity environment using a previously reported goggles system (visit 2), and repeat low-humidity exposures after using artificial tears for 14 days (visit 3) followed by dexamethasone drops for 14 days (visit 4), for a total of 3 low-humidity challenges during the study period. At each visit, subjects wore the controlled environmental goggles for 90 minutes while watching a movie on a video display placed on a stand, elevated slightly above eye level, 4.5 feet away. Ambient air was pumped continuously at a flow rate of 2–5 L/min through a conditioning system into the goggles such that the air surrounding the eyes and eyelids maintained a relative humidity of 18%–25% throughout the test period. Relative humidity, temperature, and air flow was measured every 10 seconds by sensors in the goggles and was recorded in an electronic database.
Blink Rate Measurement
Blink rate was measured using electromyography signals detected by the NeuroSky MindBand Bluetooth device (NeuroSky, Silicon Valley, California, USA). The MindBand was placed on the forehead, directly above the goggles, for the duration of the 90-minute test period. The dry electrodes on the MindBand measured the changing electrical potential of muscles during blinks. The signal was sent by Bluetooth to a computer, translated into real-time waveforms by NeuroSky software, and displayed in the ClimaTears Test Suite (Biocentric Developments LLC, Austin, Texas, USA). To determine the blink threshold specific to each subject, the size of the waveform was observed and the threshold was set to the point at which the waveform consistently crossed the threshold during a blink. The blink count readings were verified with manual blink counting for 1 minute by the investigator at 5 minutes, 45 minutes, and 75 minutes into the study.
Assessments
The same examiner performed all of the objective clinical tests. An ocular surface evaluation was performed before and after the 90-minute low-humidity exposure at visits 2, 3, and 4 ( Figure 1 ). Tear and ocular surface evaluations included fluorescein TBUT as a measure of tear stability, and corneal fluorescein and conjunctival lissamine green staining as measures of epithelial cell health. TBUT was performed by a previously reported method. Corneal fluorescein staining viewed through a yellow filter was graded in 5 zones in the cornea and 6 zones in the exposed bulbar conjunctiva (3 nasal and 3 temporal) using a previously reported grading scheme that was corrected for the percent area of staining in each zone. In addition to biomicroscopic grading of the fluorescein staining at the time of each visit, the corneal fluorescein staining was also imaged using the camera of an iPod Touch (Apple, Cupertino, California, USA) with the ProCamera application (Cocologics, Mannheim, Germany) at each visit before and after the low-humidity challenge. The same examiner viewed the videos and graded the staining, while masked to subjects’ identity, at the conclusion of the study.
Severity of eye irritation symptoms was measured using a validated questionnaire adapted from the Ocular Surface Disease Index. The questionnaire contained 5 questions regarding the frequency of photophobia, gritty/sandy sensation, burning/stinging, blurred vision, and fluctuating vision. We also assessed severity of ocular symptoms using a visual analog scale (VAS) measuring the severity and frequency of dry eye symptoms, severity of blurred vision related to dry eye, and frequency of blinking.
Treatment
After the baseline low-humidity exposure (visit 2), subjects used artificial tears (preservative-free Refresh Optive Sensitive; Allergan, Inc, Irvine, California, USA) 4 times daily for 2 weeks and returned to our clinic for a second 90-minute exposure to the low-humidity environment (visit 3). After the third visit, subjects used preservative-free 0.1% dexamethasone solution (Greenpark Pharmacy, Houston, Texas, USA) 4 times daily for 2 weeks and returned to our clinic for a third and final 90-minute exposure to the low-humidity environment at visit 4.
Conjunctival Gene Expression
Cells obtained by impression cytology of the nasal bulbar conjunctiva of each eye using the EyePrim device (OpiaTech, Paris, France), which applies a Supor 450 membrane (Pall, Port Washington, New York, USA) with uniform pressure to the conjunctival surface, were placed in 0.5 mL of RNA lysis buffer (Qiagen, Valencia, California, USA) and stored at −80 C. Total RNA was isolated from the membranes using an RNeasy Mini Plus Kit (Qiagen, Valencia, California, USA) following the manufacturer’s protocol. Cytology was performed at the enrollment visit and following the low-humidity exposure at visits 2–4. The RNA concentration was measured by its absorption at 260 nm using a spectrophotometer (NanoDrop 2000; Thermo Scientific, Wilmington, Delaware, USA), first-strand cDNA was synthesized using random hexamers and M-MuLV reverse transcriptase (Ready-To-Go You-Prime First-Strand Beads; Amersham Pharmacia Biotech, Inc, Piscataway, New Jersey, USA), and the DNA concentration was measured with a Qubit spectrophotometer (Life Technologies, Grand Island, New York, USA). Digital polymerase chain reaction (PCR) to detect copy number of HLA-DR transcripts was performed with a QuantStudio 3D Digital PCR system (Life Technologies) according to the manufacturer’s instructions and normalized by concentration of cDNA. The fold changes in HLA-DR expression relative to the baseline visit were calculated for each patient and the fold changes among all patients per visit were averaged.
Statistical Analyses
We used Statmate (GraphPad Software Inc, La Jolla, California, USA) to perform sample size calculations using the results from a prior study of 25 subjects exposed to a low-humidity environmental challenge. A sample size of 20 subjects was calculated to have 90% power of detecting a statistical difference ( P = .05) in total corneal and conjunctival staining before and after the environmental challenge. Data were compared using paired t test or repeated-measures analysis of variance (1-way ANOVA) with GraphPad Prism.
Results
Twenty subjects meeting the inclusion criteria were enrolled in the study. All subjects had aqueous tear deficiency and were classified as either non–Sjögren syndrome aqueous tear deficiency (non-SS ATD) (n = 14) or Sjögren syndrome aqueous tear deficiency (SS ATD) (n = 6) using the proposed American College of Rheumatology criteria. We chose subjects with aqueous tear–deficient dry eye based on prior findings of a significant ocular surface response to a controlled low-humidity environment in this patient population. One subject had LASIK surgery 13 years ago and none had systemic diseases except for Sjögren syndrome.
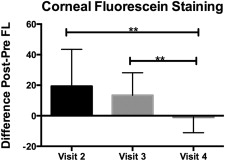
Corneal Staining and Tear Break-up Time
Fluorescein staining is an indicator of corneal epithelial barrier disruption from epithelial cell damage that is attributed to dry eye–induced stress and inflammation on the ocular surface. We calculated the difference (deltas, n = 20 eyes) in fluorescein corneal staining before (pre) and after (post) the low-humidity challenge at visits 2–4. There was no statistical difference in the deltas between visits 2 (baseline with no treatment) and 3 (post artificial tears). However, there was a statistically significant difference ( P = .002) between visits 2 and 4 (post steroid), and between visits 3 and 4 ( P = 0.003) ( Figure 2 ). Subjects with SS ATD had a magnitude of response similar to those with non-SS ATD. Heterogeneity in subject responses consisted of varying magnitudes of worsening in corneal fluorescein staining after low-humidity exposure. There was no statistical difference in tear break-up time before and after the low-humidity challenge at any of the visits.
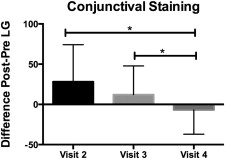
Conjunctival Staining
Lissamine green staining is routinely used to evaluate conjunctival cell membrane abnormalities in dry eye. We calculated the difference (delta, n = 20 eyes) in conjunctival lissamine green staining before (pre) and after (post) the low-humidity challenge at visits 2–4. There was no statistical difference in the deltas between visits 2 (baseline with no treatment) and 3 (post artificial tears). There was a statistically significant difference ( P = .01) between visit 2 and visit 4 (post steroid), and between visits 3 and 4 ( P = .03) ( Figure 3 ).