Purpose
To study longitudinal changes in the posterior pole in eyes with dome-shaped macular configuration within the staphyloma.
Design
Prospective, longitudinal study.
Methods
We prospectively examined the macular area in 35 eyes (26 patients) with dome-shaped macular configuration and high myopia (mean spherical equivalent, −14.83 ± 4.50 diopters) using swept-source optical coherence tomography. Scleral and choroidal thicknesses were measured at the fovea and at 4 parafoveal locations 2000 μm from the foveal center. Height of the macular bulge was measured as well.
Results
During the mean follow-up of 24.8 ± 2.5 months, the scleral thickness significantly decreased at the fovea from 496.1 ± 95.7 μm to 484.7 ± 96.2 μm ( P < .001) and at all 4 parafoveal locations ( P < .001, respectively). The scleral thinning was asymmetric, with an estimated decrease per year of 5.6 μm at the foveal center, 11.1 μm superiorly, 12.1 μm inferiorly, 10.4 μm temporally, and 5.8 μm nasally. The ocular concavities deepened over time, and mean macular bulge height increased from 136.5 ± 60.9 μm to 157.6 ± 67.0 μm ( P < .001). The choroid within the staphyloma showed generalized thinning during follow-up. Mean choroidal thickness decreased significantly at the fovea from 28.3 ± 17.2 μm at baseline to 22.9 ± 17.2 μm ( P < .001).
Conclusions
Progressive asymmetric scleral thinning occurred in the macular region of eyes with dome-shaped macular configuration. The scleral thinning was more pronounced in the parafoveal area than at the foveal center, resulting in an increase of the macular bulge height.
Dome-shaped macular configuration is a distinctive entity in eyes with high myopia where macular curvature shows an inward convexity within the posterior staphyloma, as opposed to the physiologic curvature of the posterior pole. It is now recognized that dome-shaped macular configuration is not a rare finding in highly myopic eyes, with estimated prevalence of 10.7% in a European study and 9.3% in a Japanese study. Although it was first described as dome-shaped macula, later, 3-dimensional (3D) image reconstruction of the posterior pole topography by optical coherence tomography (OCT) showed that dome-shaped macula may involve several patterns: typical dome-shaped convexity, the more common horizontal band-shaped ridge traversing the posterior staphyloma, and the rare vertical oval-shaped dome.
The mechanisms that underlie this unusual tomographic feature remain unknown. Several hypotheses have been assumed, such as localized choroidal or scleral thickening, ocular hypotony, tangential vitreomacular traction, and resistance to staphylomatous deformation. Based on 3D reconstructed images, we previously hypothesized that dome-shaped macular configuration occurs as a part of the process of asymmetric expansion of the globe, secondary to regional differences in the structural strength of the sclera at the macula. This may lead to formation of a horizontal ridge by uneven scleral thinning. Because the sclera is the primary determinant of eyeball shape and the main changes in ocular elongation in high myopia take place at the scleral coat, tracking changes in the sclera may help to elucidate the mechanism underlying the formation of a dome-shaped macular configuration.
With the advances in OCT, the third-generation swept-source OCT at 1 μm wavelength allows deep penetration imaging of the choroid and sclera in highly myopic eyes. The higher imaging speed allows for dense scanning over large areas and subsequent 3D image reconstruction of posterior pole topography. With the use of this noninvasive OCT technology, it is possible to track the longitudinal changes in the posterior segment topography in vivo. We previously reported marked regional scleral thinning in the superior and inferior parafoveal areas in eyes with dome-shaped macular configuration within the posterior staphyloma. The current prospective study aims to analyze changes to the choroid and sclera and morphologic changes in the shape of the posterior pole over the course of 2 years in eyes with a dome-shaped macular configuration.
Patients and Methods
This prospective study was approved by the Ethics Committee at Kyoto University Graduate School of Medicine and conducted in accordance with the tenets of the Declaration of Helsinki. The nature of the current study and the implications of participating in this research were explained to all study candidates, after which a written informed consent was obtained from each participant before any study procedures or examinations were performed.
In the current study, we prospectively followed 35 eyes (26 patients) with dome-shaped macular configuration using swept-source OCT at Kyoto University Hospital from the beginning of October 2010 to the end of January 2014. The diagnosis of dome-shaped macular configuration was based on the presence of an inward bulge of the retinal pigment epithelium (RPE) line, greater than 50 μm on the most convex vertical or horizontal OCT sections above a presumed line tangent to the outer surface of the RPE at the bottom of the posterior staphyloma in highly myopic eyes. We refer to the latter measurement as “macular bulge height.” The results of 3D OCT imaging performed at baseline were presented in our previous report.
High myopia was defined as refractive error of −6.0 diopters or more and/or an axial length of 26.5 mm or greater. The exclusion criteria were poor image quality owing to media opacity (eg, cataracts or corneal opacity) and/or a history of ocular surgery, other than cataract surgery. The original refractive error was used for eyes that underwent cataract surgery. Curtin’s classification was used to classify posterior staphylomas based on location and size. We further excluded eyes with inferior posterior staphyloma (type V). In the current study, all eyes had a posterior staphyloma of type I, II, III, or IX.
All subjects underwent a comprehensive ocular examination, including autorefractometry, best-corrected visual acuity measurement with a 5 m Landolt chart, corneal curvature, axial length measurement using partial coherence interferometry (IOL-Master; Carl Zeiss Meditec, Jena, Germany), slit-lamp examination, intraocular pressure measurement, dilated color fundus photography (TRC-NW8F or TRC50LX; Topcon Corp, Tokyo, Japan), and swept-source OCT examination. All eyes with macular complications underwent simultaneous fluorescein angiography and indocyanine green angiography, using the Spectralis HRA+OCT (Heidelberg Engineering, Heidelberg, Germany).
Swept-Source Optical Coherence Tomography and Scan Protocols
The prototype swept-source OCT used in the current study has been previously described and has recently become commercially available (DRI OCT-1; Topcon Corp, Tokyo, Japan). All swept-source OCT examinations were performed by a trained examiner after pupillary dilation. For all patients, horizontal and vertical line scans (12 mm) through the fovea were obtained. For each scan, ∼50 B-scan images were obtained and averaged to reduce speckle noise. The vertical scan was centered on the fovea; the horizontal scan was centered on the midpoint between the fovea and optic disc. 3D imaging datasets were acquired using a raster scan protocol of 512 (horizontal) × 128 (vertical) A-scans per dataset. Each raster scan consisted of 128 B-scans and covered an area of 12 × 8 mm 2 or 12 × 9 mm 2 , centered on the fovea. In each of the 128 B-scans used for 3D analysis, the RPE line was segmented automatically with manual corrections if necessary to construct 3D images of posterior pole curvature.
Scleral and Choroidal Thickness Measurement Protocol
We measured the choroidal and scleral thicknesses at the center of the fovea and at surrounding parafoveal regions at points 2000 μm superiorly, inferiorly, temporally, and nasally in the multi-averaged OCT scans ( Figure 1 ), with a built-in caliber tool within the software. Retinal thickness was defined as the distance between the vitreoretinal interface and the outer border of the RPE; choroidal thickness was defined as the distance between the outer border of the RPE line and the chorioscleral interface; scleral thickness was defined as the distance between the chorioscleral interface and the outer scleral border. Retinal, choroidal, and scleral thicknesses at the foveal center were calculated as the average of the measurements in the vertical and horizontal OCT images.
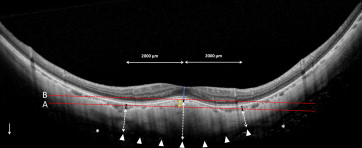
We measured the macular bulge height as the height of the inward bulge of the RPE above a presumed line tangent to the RPE (dashed red line A) at the fovea within the bottom of the posterior staphyloma in the most convex vertical or horizontal scans ( Figure 1 ). Macular bulge height depends mainly on 2 parameters. One is the difference between scleral and choroidal thicknesses at the fovea and parafoveal regions, and the second is the change in depth of ocular concavities within the staphyloma. To highlight these dynamic changes at the bottom of the staphyloma, a second line (B) was drawn parallel to the line (A) tangent to the outer surface of the RPE at the fovea (dashed red line B, Figures 1–4 and Supplemental Figure , available at AJO.com ). The distance between lines A and B is equivalent to the macular bulge height.
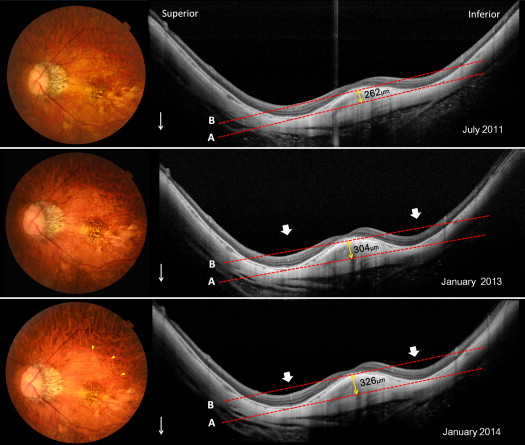
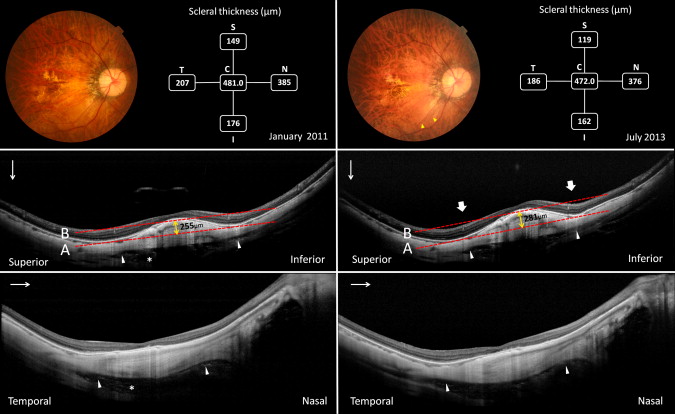
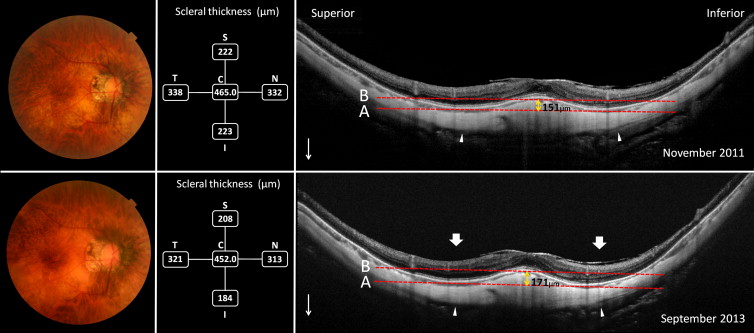
The outer scleral border was carefully identified in the OCT scans from retro-ocular structures such as the Tenon capsule, episcleral tissue, inferior oblique aponeurosis, and orbital fat. Scleral tissues were identified by their lamellar morphology, the continuity of the outer surface of the sclera within the raster lines in the vertical and horizontal scans, and high reflectivity values.
In the follow-up examinations, the choroidal and scleral thicknesses were measured again with careful attention to measure at the exact location from the initial visit. In 6 eyes, the outer border of the sclera was not clearly visible at the fovea, either at the first visit or at follow-up, which precluded comparison. These eyes were excluded from the study. In all recruited subjects, the outer border of the sclera at the fovea and at the 4 parafoveal locations was identified clearly at the initial and follow-up visits.
Statistical Analysis
Statistical analysis was performed using SPSS statistical software (version 20; SPSS Inc, Chicago, Illinois, USA). All values are expressed as mean ± standard deviation. The measured visual acuity was converted to the logarithm of the minimal angle of resolution (logMAR) for statistical analysis. The paired t test was used to compare numerical variable means for baseline and follow-up measurements. Choroidal and scleral thicknesses at various parafoveal regions were compared to that of the foveal center using 1-way analysis of variance with least significant difference post hoc analysis. A P value of less than .05 was considered to be statistically significant.
Results
In the current study, 35 eyes (26 subjects; 6 men and 20 women) with dome-shaped macular configuration were examined by swept-source OCT. The mean follow-up period was 24.8 ± 2.5 months (range: 20–30 months). All patients were Japanese. Table 1 shows the baseline characteristics for this patient population. At the initial visit, mean age was 65.9 ± 12.7 years (range: 41–86 years); mean refractive error was −14.83 ± 4.50 diopters (range: −7.5 to −27.0 diopters); mean axial length was 29.86 ± 2.16 mm (range: 26.68–34.65 mm). Three-dimensional images revealed a band-shaped ridge traversing the bottom of the posterior staphyloma in 30 eyes (85.7%) and a dome-shaped bulge in 5 eyes (14.3%). In all eyes, macular bulge height was greater in the vertical scan as compared to the horizontal scan. None of the eyes examined had a vertically oriented oval dome. Among our patients, choroidal neovascularization (CNV) was seen in 17 eyes (48.6%), serous retinal detachment in 2 eyes (5.7%), lamellar hole in 3 eyes (8.6%), full-thickness macular hole in 1 eye (2.9%), extrafoveal schisis in 9 eyes (25.7%), and foveal schisis in 1 eye (2.9%). Five eyes with CNV received anti–vascular endothelial growth factor treatments during the follow-up period.
| Number of eyes/patients | 35/26 |
| Mean follow-up (mo) | 24.8 ± 2.5 (20 to 30) |
| Sex, male/female (%) | 6/20 (23.1 %/76.9 %) |
| Age (y) a | 65.9 ± 12.7 (41 to 86) |
| Axial length (mm) a | 29.86 ± 2.16 (26.68 to 34.65) |
| Refractive error (diopters) a | −14.83 ± 4.50 (−7.50 to −27.0) |
| Visual acuity (logMAR) a | 0.37 ± 0.49 (−0.18 to 1.70) |
| Intraocular pressure (mm Hg) a | 14.5 ± 3.0 (9 to 22) |
| Average corneal curvature (mm) a | 7.72 ± 0.24 (7.30 to 8.30) |
| Macular bulge configuration | |
| Band-shaped ridge | 30 (85.7%) |
| Dome-shaped bulge | 5 (14.3%) |
| Type of posterior staphyloma, eyes (%) | |
| I | 12 (34.3%) |
| II | 20 (57.1%) |
| III | 1 (2.9%) |
| IX | 2 (5.7%) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


