Purpose
To evaluate a series of orbital diffuse large B-cell lymphomas (DLBCL) for prognostic features and therapeutic outcomes.
Design
Retrospective multicenter case study of clinical and immunohistochemical features of 20 patients.
Methods
Clinical, histopathologic, and immunohistochemical features were correlated with outcomes. Immunohistochemistry for biomarkers including Bcl-6, CD5, CD10, CD20, FOXP1, GCET1, and MUM1 was performed to differentiate between 2 major genetic subtypes of DLBCL: activated B-cell-like (ABC) and germinal center B-cell-like (GCB).
Results
Sixteen patients presented with unilateral and 4 with bilateral tumors. Three had bony erosion of the orbit on imaging studies. Of 14 patients with detailed follow-ups, 3 had a prior or concurrent lymphomatous disease; 8 had stage I disease (limited to the orbit) at presentation; and 3 were newly diagnosed with systemic (stage IV) DLBCL. Localized disease was treated with combined systemic chemotherapy, including rituximab and radiation with no deaths to date; there was 1 death related to systemic DLBCL. Clinical staging was the best predictive method and no immunohistochemical feature or subcategory (ABC vs GCB) correlated with outcome.
Conclusions
Primary orbital DLBCL has a more favorable prognosis than systemic DLBCL and may arise from a preexistent hematolymphomatous neoplasm (4 out of 20 cases). In our series, orbital DLBCL had a 57% likelihood of being restricted to the ocular adnexa. Clinical staging was more helpful in predicting outcome than any single immunohistopathologic feature or combination of biomarkers. Orbital radiation of 30 gray in conjunction with systemic chemotherapy with rituximab can achieve disease-specific survival approaching 100% in purely localized cases.
Diffuse large B-cell lymphoma (DLBCL) is the most common nonocular lymphoma, accounting for 30% of systemic non-Hodgkin lymphomas. It is an aggressive disease that can arise de novo or, less commonly, from the transformation of a preexisting low-grade B-cell lymphoma. DLBCL affects men slightly more frequently than women, with a median age of 64 years at presentation. Approximately half of DLBCL patients present with stage I or II disease, and 71% will have extranodal involvement during the course of their disease. Treated DLBCL has an overall 46% 5-year survival. Two subcategories of DLBCL have been characterized by means of gene expression profiling techniques. The first is termed germinal center B-cell-like (GCB) and has a more favorable prognosis and response to chemotherapy than the second, which is called activated B-cell-like (ABC). The 2 subtypes may respond differently to various chemotherapeutic regimens. As gene profiling is impractical for routine histopathologic diagnosis, immunohistochemical algorithms such as those proposed in the “Choi” and “Hans” methods use a stepwise analysis of specific markers to develop a more efficient way to separate the 2 phenotypic subtypes.
Despite being the third most common primary ocular adnexal lymphoma, primary orbital DLBCL is nonetheless relatively rare in the orbit (8% of ocular adnexal lymphomas) when compared to extranodal marginal zone and follicular lymphomas (about 50% and 25%, respectively). It should be noted, however, that in 15 cases of lymphoma of the lacrimal sac, 10 displayed features of DLBCL. DLBCL is more common when considered as a secondary manifestation of an already systematized lymphoma. Consequently, reports that focus on this disease within the orbit and that provide both reliable clinical and immunopathologic data are sparse. It was first discovered in 1989 that orbital DLBCL, even with bone destruction, was compatible with a 2.5-year survival rate of 56%. The authenticity of this finding was supported by several studies of many different ocular adnexal lymphomas that included small numbers of DLBCLs, which similarly indicated a prognosis that seemed more favorable than expected given the worrisome cytology of the lesions.
In the largest series to date of 353 orbital lymphomas that included 27 patients with DLBCL, only 5 patients had prior lymphoma elsewhere at the time of diagnosis, and 2 of these had evidence of a preexisting lower-grade lymphoma that transformed into DLBCL. Unfortunately, neither the clinical features nor staging data with outcomes were supplied in this report. A recent multicenter collection of 37 DLBCL cases disclosed a 54% incidence of stage 1 presentation with a median follow-up of 10 months, but an exploration of correlative or prognosticating data was not undertaken. In a separate case series of 109 lymphoproliferative lesions, orbital DLBCL was identified in 9 patients, all of whom manifested disease beyond stage I.
To provide more supplemental information on the clinical, radiographic, pathologic, and immunophenotypic findings of ocular adnexal DLBCLs, we analyzed 20 new cases pooled from 3 institutions. We evaluated the cytomorphologic and immunohistochemical features in greater depth than supplied in earlier studies. We particularly focus on individual or multiple immunohistochemical biomarkers (algorithms) that might have prognostic value with regard to the 2 major genetic subtypes, GCB and ABC, as alleged for DLBCL of other sites. Finally, we assessed treatment options based on the data derived from the current study as well as from published ophthalmic and nonophthalmic series, in order to discover the optimal therapeutic approach for orbital DLBCL.
Methods
Twenty cases of DLBCL accumulated from 3 different institutions (Massachusetts Eye and Ear Infirmary, Emory Eye Center, and the Cole Eye Institute, Cleveland Clinic) between the years of 1996 and 2011 were selected for this series. All histopathologic and immunohistochemical slide preparations were independently reviewed by F.A.J. and H.E.G. for accuracy of diagnosis, with a 100% concordance. Two specimens were obtained by means of a conjunctivectomy of the leading edges of an unequivocal orbital mass; the remaining 18 specimens were obtained through orbitotomies.
The tissues were embedded in paraffin and stained with hematoxylin-eosin. Immunohistochemical biomarkers were then employed for further characterization and included Bcl-2, Bcl-6, CD3, CD5, CD10, CD20, CD21, CD23, CD43, cyclin D1, FOXP1, GCET1, MUM1, Pax5, and Ki-67, using standard staining protocols as described elsewhere on Ventana Benchmark automated immunostainers ( Table 1 ).
| Antibody | Usual Cell Stained | Protein Location or Function | Staining Pattern | Source | Dilution |
|---|---|---|---|---|---|
| Bcl-2 | B lymphocytes (surrounding germinal centers in normal tissue) | Antiapoptotic protein | Cytoplasm | Mouse monoclonal a , f | Prediluted |
| Bcl-6 | B lymphocytes (follicular center cells in normal tissue) | Transcription factor | Nucleus | Rabbit polyclonal b , mouse monoclonal c | 1:100/1:5 |
| CD3 | T lymphocytes | T cell receptor complex | Cell membrane | Rabbit polyclonal, mouse monocloncal a | Prediluted |
| CD5 | T lymphocytes | Transmembrane glycoprotein | Cell membrane | Rabbit monoclonal a , Mouse monoclonal d | Prediluted/1:10 |
| CD10 | T lymphocytes and germinal center B cells | Membrane endopeptidase | Cell membrane | Mouse monoclonal a , d | Prediluted/1:5 |
| CD20 | B lymphocytes | Calcium channel membrane protein | Cell membrane | Mouse monoclonal a , c | Prediluted/1:50 |
| CD21 | Follicular dendritic cells | Membrane protein | Cell membrane/dendritic processes | Mouse monoclonal a , c | Prediluted/1:10 |
| CD23 | Follicular dendritic cells | Membrane protein | Cell membrane/dendritic processes | Mouse monoclonal a , e | Prediluted/1:20 |
| CD79a | B lymphocytes | Membrane protein | Cell membrane | Rabbit monoclonal a | Prediluted |
| Cyclin D1 | Mantle cell lymphoma | Cell cycle regulator | Nucleus | Rabbit polyclonal a | Prediluted |
| FOXP1 | B lymphocytes | Transcription factor – overexpressed in DLBCL | Nucleus | Rabbit polyclonal g | 1:400 |
| GCET1 | B cells in germinal center stage | Protease inhibitor | Cytoplasm | Rabbit polyclonal g | 1:80 |
| MUM1 | Differentiating germinal B center cells | Transcription factor | Nucleus | Mouse monoclonal c | 1:40 |
| Pax5 | B lymphocytes | Transcription factor | Nucleus | Rabbit monoclonal a | Prediluted |
| Ki-67 | Cells in premitotic state (S-phase) | Cell proliferation | Nucleus | Rabbit monoclonal a | Prediluted |
| Kappa | Plasma cells | Ig light chain | Cytoplasm | Rabbit polyclonal c | 1:2000/1:32 000 |
| Lambda | Plasma cells | Ig light chain | Cytoplasm | Rabbit polyclonal c | 1:1400/1:64 000 |
a Ventana Medical Systems, Arizona, USA.
b Leica Microsystems, Illinois, USA.
c DAKO Corporation, California, USA.
d Novocastra Laboratories Ltd, United Kingdom.
e Binding Site Inc, California, USA.
f Cell Marque Corporation, California, USA.
After immunostaining the slides, the samples were evaluated using 2 different, previously described algorithms, which differentiate between the GCB and ABC subtypes of DLBCL by comparing the results of 3 or 5 immunohistochemical biomarkers employed in a stepwise fashion: CD10, Bcl-6, MUM1, FOXP1, and GCET. In brief, the “Hans algorithm” uses CD10 positivity as an initial determining factor, and then further stratifies specimens based on Bcl-6 and MUM1 positivity. The end result of the Hans algorithm defines the GCB subtype as any lesion that exhibits CD10+, or that is CD10-Bcl6+MUM1-, using 30% positivity as a cutoff. ABC subtypes are either CD10-Bcl6- or CD10-Bcl6+MUM1+. The “Choi algorithm” uses 2 additional biomarkers, FOXP1 and GCET1, both at 80% cutoff for positivity, to supplement interpretation of the CD10, Bcl-6, and MUM1 markers (using 30%, 30%, and 80% cutoffs, respectively). The Choi algorithm starts by analyzing GCET positivity and then continues across a flow chart to produce the following results: GCB cases are GCET1+MUM1-, GCET1-CD10+, or GCET1-CD10-Bcl6+FOXP1-; ABC cases are GCET1+MUM1+, GCET1-CD10-Bcl6-, or GCET1-CD10-Bcl6+FOXP1+. All of our specimens were analyzed by both algorithms and, once determined to be either GCB or ABC subtype, were then correlated with clinical findings and outcomes.
Patients’ clinical histories and follow-ups were obtained by reviewing the patients’ hospital charts and/or the attending ophthalmologists’ office records and clinical photographs. Data on lesion location, tissue collection, and patient demographics were available in all cases. Detailed clinical histories on presentation were available on 12 patients, imaging studies on 12 patients, and information on treatment and clinical follow-up overseen by an oncologist on 10 patients. Systemic investigations included computed tomographic (CT) scans of the chest, abdomen, and pelvis; magnetic resonance imaging (MRI) of the brain and orbit; and a bone marrow biopsy on 4 patients. Flow cytometry was performed in 11 cases and polymerase chain reaction (PCR) for gene rearrangement in 1.
Results
Clinical Features
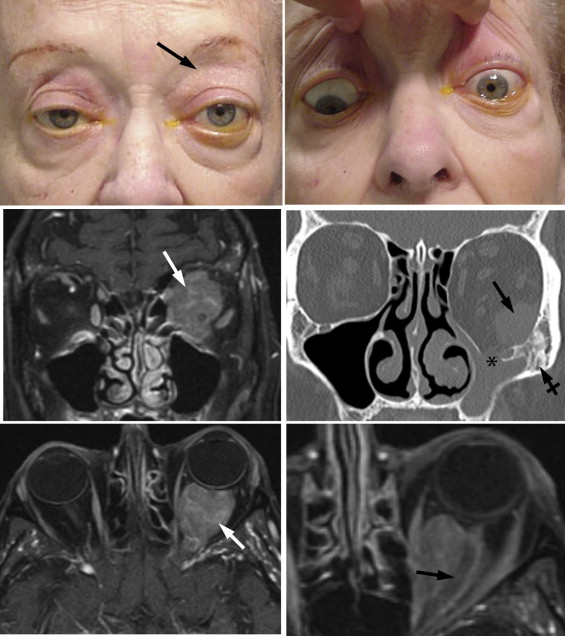
The patients included 10 men and 10 women; the median age at the time of presentation was 75 years (range 31−88 years). Two patients presented with subconjunctival extensions of an orbital tumor; the remaining 18 cases represented deeper orbital lesions. Four patients had bilateral disease; of the unilateral cases, 12 were located on the left and 4 on the right. Nine patients had obvious orbital masses on clinical inspection, 4 of which were documented to be in the superior orbit ( Figure 1 , Upper left). Two patients complained of a foreign body sensation and 2 of diplopia. On examination, 2 patients had decreased visual acuity, 4 patients had limited eye movements on the involved side ( Figure 1 , Upper right), 4 had measurable proptosis between 1 and 5 mm, and 2 had ptosis. MRI and CT scanning disclosed orbital masses that in the majority of cases were located superiorly and occupied intraconal, extraconal, or both compartments ( Figure 1 , Middle left); 3 involved the lacrimal glands. In 3 out of 12 cases evaluated with imaging studies, there was bone erosion or remodeling ( Figure 1 , Middle right). Bone erosion occurred in 2 patients with localized orbital disease at presentation; the third had an already diagnosed systemic lymphoproliferative disorder. In 2 cases an intraconal lesion encased the optic nerve ( Figure 1 , Lower left and right). One such patient with a perioptic nerve mass had decreased visual acuity of 20/30 and an afferent pupillary defect. Eleven patients had total-body. Four patients had bone marrow biopsies, which were negative for DLBCL. In 10 cases flow cytometry was performed; 7 samples showed peripheral blood monoclonality with light chain restriction (2 kappa and 5 lambda) but 3 did not. In 1 case, PCR and fluorescence in situ hybridization (FISH) revealed IGH – BCL2 gene rearrangement but no c-myc rearrangement.
Three patients had prior histories of lymphoma and another was diagnosed with chronic myelocytic myelogenous leukemia (CMML) before the orbital presentation of lymphoma. A 69-year-old patient had a B-cell lymphoma, not classifiable (no further detail was given and her biopsy is unavailable for further review), situated behind the left eustachian tube 14 years earlier; another, aged 54 years, had abdominal DLBCL diagnosed 2 years earlier; and the third, aged 65 years, was diagnosed with a B-cell lymphoma displaying lymphoplasmacytic features that was discovered to be widely metastatic 8 months before the orbital lesion manifested. This particular patient then developed a left upper eyelid mass 2 months later, which was treated empirically and without biopsy with 4 gray of radiation. However, the mass recurred 2 months later, which prompted the biopsy that is included in this series. A fourth patient, aged 77 years with a history of CMML for 3 years, developed DLBCL of the orbit while undergoing decitabine chemotherapy for his CMML. This patient was 1 of the 3 who had bony erosion on a head CT scan. The other 11 patients with ascertainable clinical histories had no prior history of lymphoma.
Histopathologic Findings
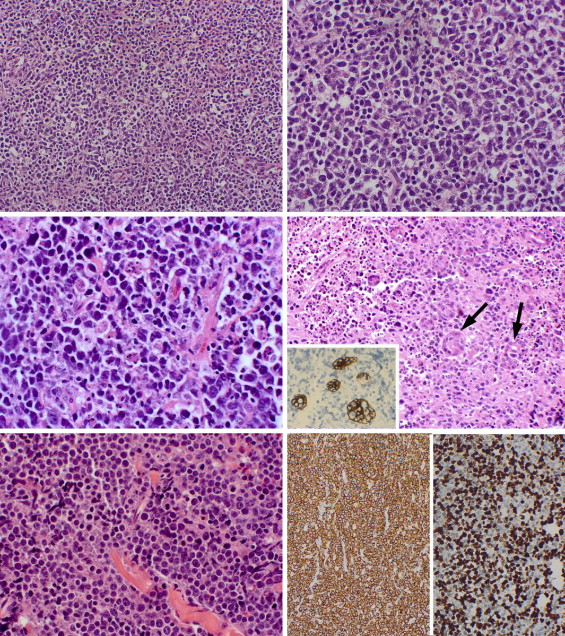
The 20 tumors were composed of large to medium-sized lymphocytes, usually 2 times the dimensions of small lymphocytes and approximating the size of histiocytes, arranged in featureless sheets without follicular organization ( Figure 2 , Upper left). Infiltration of the orbital fat was typical; focal, coagulative necrosis was identified in about half the cases. The lymphocytes usually displayed centroblastic-type nuclei that were round to oval or vesicular; they possessed multiple small nucleoli located at the nuclear membrane ( Figure 2 , Upper right). Smaller centrocytes with cleaved or indented nuclei were identified as a minority population. Mitotic figures, karyorrhexis, and apoptosis were discovered in all lesions ( Figure 2 , Middle left). In specimens that involved the lacrimal gland, degenerating acini obscured by necrosis and surrounding tumor cells were highlighted by pancytokeratin immunostaining ( Figure 2 , Middle right, and inset). In none of the 20 DLBCL specimens were there cytologic signs of immunoblastic lymphoma (cells exhibiting a dominant nucleolus in a large round nucleus with a broad rim of basophilic to amphophilic cytoplasm) or plasmablastic differentiation. Lesions composed of cells with anaplastic bizarre nuclei and more ample, clear cytoplasm were also absent. In 1 patient in whom a prior biopsy demonstrated a lymphoplasmacytic lymphoma ( Figure 2 , Lower left), the newly diagnosed orbital lesion was different, with less distinct amphophilic cytoplasm, in keeping with a conventional DLBCL. In another patient, evidence of a lower-grade, preexisting lymphoma (probably an extranodal marginal zone or follicular lymphoma) with attenuated follicular centers possessing dendritic cells (see below) was discovered. Review of the slides obtained from the earlier nonophthalmic biopsies confirmed their accuracy.
Immunohistochemical Findings
Of the 18 specimens stained for CD20, all were positive, thereby establishing a B-cell proliferation ( Figure 2 , Lower right, left panel) rather than a large T-cell neoplasm. Variable expression was noted for MUM1, Bcl-2, Bcl-6, CD-10, CD79a, FOXP1, and GCET1; scattered T cells were also stained by CD3 or CD5. In 1 case from a subset of 6 specimens, CD5 coexpression on neoplastic B cells was observed. Cyclin D1 was negative. Ki-67 proliferation index was high (usually 60%−80%, Figure 2 , Lower right, right panel). In 1 case, CD21 and CD23 highlighted dendritic cells suggestive of a residual follicular pattern, consistent with a transformation from either a local orbital extranodal marginal zone or a follicular lymphoma into a DLBCL ( Figure 3 , Upper left). Bcl-2 staining was positive in 9 out of 10 cases. Kappa and lambda in situ hybridization in 2 cases showed kappa restriction but in general demonstrated only sparse staining in the remaining specimens.
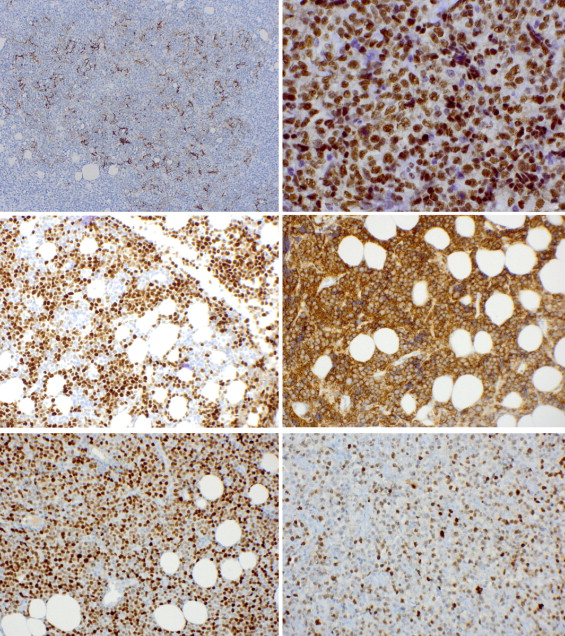
The specimens were further analyzed using the stains proposed in the Hans and Choi algorithms: GCET1, FOXP1, Bcl-6, CD10, and MUM1 ( Figure 3 ). The results are tabulated in Table 2 . Using the algorithm described by Choi and associates, 15 cases were subclassified as GCB and 5 as ABC, whereas the Hans algorithm resulted in 11 GCB and 9 ABC cases. The 2 algorithms concurred on 10 GCB and 4 ABC cases. Five cases were classified as ABC by the Hans algorithm but GCB by the Choi algorithm; 1 case was classified as GCB by the Hans algorithm but ABC by the Choi algorithm. Two of the patients with a prior history of lymphoma were classified as GCB by both algorithms, and 1 as ABC. The patient with a prior history of CMML was characterized as GCB by Choi and ABC by Hans. Among the patients presenting with stage I disease, there were 4 GCB subtypes, 1 ABC subtype, and 2 cases in which the algorithms did not agree on a subtype. Among patients presenting with stage IV disease, there were 4 GCB and 1 ABC cases.
| Germinal Center B-Cell-Like (N = 10) | Activated B-Cell-Like (N = 4) | Nonconcordant Subclassification a (N = 6) | |
|---|---|---|---|
| GCET1 | 8/10 (80%) | 4/4 (100%) | 6/6 (100%) |
| MUM1 | 0/10 (0%) | 4/4 (100%) | 1/6 (17%) |
| CD10 | 8/10 (80%) | 0/4 (0%) | 1/6 (17%) |
| Bcl-6 | 8/10 (80%) | 3/4 (75%) | 1/6 (17%) |
| FOXP1 | 9/10 (90%) | 4/4 (100%) | 5/6 (83%) |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


