Malignant parotid tumors are heterogeneous and diverse. Accurate diagnosis requires a pathologist familiar with the various histologic subtypes, immunohistochemistry stains, and common translocations. Clinical course varies according to tumor subtype, ranging from indolent, slow-growing adenoid cystic carcinoma to rapidly progressive, possibly fatal, salivary ductal carcinoma. Histologic grade is important in prognosis and therapy. Surgery remains the mainstay of treatment when negative margins can be achieved. Radiation improves locoregional control of tumors with high-risk features. Chemotherapy for parotid tumors can be disappointing. Studies of new targeted therapies have not offered significant benefits.
Key points
- •
Histology, immunohistochemistry, and identification of gene translocations are important for parotid tumor diagnosis.
- •
Parotid cancers have diverse histopathology and clinical behavior.
- •
Treatment requires a multidisciplinary approach and surgery to negative margins is a mainstay, supplemented by radiation for better locoregional control and chemotherapy, usually in the palliative recurrent or metastatic setting.
- •
Targeted therapies are under active investigation; however, they are not yet proven in the clinical setting.
Introduction
Salivary gland cancers are rare with an annual incidence of approximately 3.0 cases per 100,000 in the United States accounting for less than 3% of all head and neck cancers. They are the most heterogeneous of any group of cancers and pose diagnostic challenges due to their rarity of occurrence, diversity of types, and the limited experience of most cytopathologists. New molecular tests are being identified and may add to the diagnostic accuracy.
Approximately 70% of salivary cancers occur in the parotid gland. This article discusses primary salivary cancers with emphasis on the parotid gland. It provides an overview of select parotid cancers and up-to-date trends in management.
Introduction
Salivary gland cancers are rare with an annual incidence of approximately 3.0 cases per 100,000 in the United States accounting for less than 3% of all head and neck cancers. They are the most heterogeneous of any group of cancers and pose diagnostic challenges due to their rarity of occurrence, diversity of types, and the limited experience of most cytopathologists. New molecular tests are being identified and may add to the diagnostic accuracy.
Approximately 70% of salivary cancers occur in the parotid gland. This article discusses primary salivary cancers with emphasis on the parotid gland. It provides an overview of select parotid cancers and up-to-date trends in management.
Causes
The causes of salivary gland tumors are not well understood. Radiation exposure has been clearly established as a risk factor for parotid malignancies. This association was first observed in atomic bomb survivors but has also been seen in cancer survivors who were treated with radiation. Even patients with exposure to low-dose radiation or those treated with radioactive iodine may be at risk. A possible dose–response relationship of cellular telephone use with epithelial parotid gland malignancy has also been described. Despite parotid communication with the oral cavity, human papilloma virus has no proven role in parotid carcinogenesis. Benign tumors, if untreated, can transform into malignant forms over time. There is generally no ethnic propensity for salivary cancers, although a higher incidence of malignant oncocytoma has been reported in Eskimos.
Histology and immunohistochemistry
An understanding of the normal architecture and histology of the salivary gland aids in tumor categorization. The glandular structure is composed of 2 types of cells: luminal (acinar and ductal) and abluminal (myoepithelial and basal). The gland is further organized into lobules containing several acini, which then connect into intercalated followed by striated ducts. Cuboidal cells line the acini and may produce serous, mucoserous, or mucous secretions. Parotid acini secretions are predominantly serous. Intercalated and striated ducts are lined with cuboidal and simple columnar epithelium, respectively. Each cell type expresses a variety of markers that allows for identification and characterization ( Table 1 ).
| Antigen | Luminal Cells | Abluminal Cells | ||
|---|---|---|---|---|
| Acinar | Ductal | Myoepithelial | Basal | |
| CK (AE1/AE3) | + | + | + | + |
| EMA | + | + | − | − |
| CEA | + | + | − | − |
| CK14 | − | − | + | |
| p63 | − | − | + | + |
| α-Smooth muscle actin | − | − | + | − |
| Muscle-specific actin | − | − | + | − |
| Calponin | − | − | + | − |
| Podoplanin | − | − | + | − |
| Vimentin | − | − | + | − |
| S-100 | Variable | Variable | Variable | Variable |
Translocations and fusion genes
Although salivary cancers are uncommon, growing understanding of their unique biologic characteristics is paving the way toward specific and personalized treatment. It is now known that most mucoepidermoid carcinoma (MEC) harbors the MECT1-MAML2 gene rearrangement. The MYB-NFIB translocation has recently been identified in adenoid cystic carcinoma (ACCa). Finally, the newly described mammary analogue secretory carcinoma harbors the ETV6-NTRK3 translocation. Although these translocations are mainly of diagnostic value, they may evolve as potential therapeutic targets in the future ( Table 2 ).
| Tumor Type | Chromosomal Translocation | Fusion Oncogenes |
|---|---|---|
| MEC | t(11;19) (q21;p13) t(6;22) (p21;q12) | CRTC1 a -MAML2; CRTC3-MAML2 EWSR1-POU5F1 |
| ACCa | t(6;9) (q22–23;p23–24) | MYB-NFIB |
| Carcinoma ex pleomorphic adenoma b | t(3;8) (p21;q12) or t(5;8) (p13;q12) t(3;12) (p14.2;q14–15) | PLAG1 HMGA2 b |
| Mammary analogue secretory carcinoma | t(12;15) (p13q25) | ETV6-NTRK3 |
| Hyalinizing clear cell carcinoma | t(12;22) (q13;q12) | EWSR1-ATF1 |
b Translocations are found in benign pleomorphic adenoma and carry over into carcinoma ex pleomorphic adenoma.
Classification of malignant parotid tumors
The last formal histologic classification of salivary cancers was published by the World Health Organization (WHO) in 2005 and included 24 separate entities. Since then, new subtypes have been described and are included in a modified WHO list of 28 separate entities ( Box 1 ). This classification helps facilitate accurate and consistent diagnoses.
Acinic cell carcinoma
Mucoepidermoid carcinoma
Adenoid cystic carcinoma
Polymorphous low-grade adenocarcinoma
Epithelial-myoepithelial carcinoma
Clear cell carcinoma, not otherwise specified, and hyalinizing variant
Basal cell adenocarcinoma
Malignant sebaceous tumors
Cystadenocarcinoma
Low-grade cribriform cystadenocarcinoma
Mucinous adenocarcinoma
Oncocytic carcinoma
Salivary duct carcinoma
Adenocarcinoma, not otherwise specified
Myoepithelial carcinoma
Carcinoma ex pleomorphic adenoma
Carcinosarcoma
Metastasizing pleomorphic adenoma
Squamous cell carcinoma
Small cell carcinoma
Large cell carcinoma
Lymphoepithelial carcinoma
Sialoblastoma
Carcinoma arising in Warthin tumor
Sebaceous carcinoma
Sebaceous lymphadenocarcinoma
Malignant transformation of ductal papilloma
Signet ring cell adenocarcinoma
Histology and grade are important determinants of local, regional, and distant disease control. For the purpose of study and reporting, each cancer can be categorized into low-grade, intermediate-grade, or high-grade categories ( Table 3 ). A detailed discussion of each cancer type is beyond the scope of this article. Nonetheless, a select group of parotid cancers is highlighted.
| Malignant Salivary Tumors | |
|---|---|
| Low-grade to intermediate-grade | High-grade |
| Low-grade MEC a | High-grade MEC a |
| Tubular or cribriform ACCa a | Solid ACCa a |
| Low-grade adenocarcinoma, NOS a | High-grade adenocarcinoma, NOS a |
| Acinic cell carcinoma | Salivary duct carcinoma |
| Polymorphic low-grade adenocarcinoma | Carcinoma ex pleomorphic adenoma |
| Mucinous adenocarcinoma | Metastasizing pleomorphic adenoma |
| Clear cell carcinoma, NOS | Squamous cell carcinoma |
| Epithelial-myoepithelial carcinoma | Undifferentiated carcinoma |
| Myoepithelial carcinoma | Mucinous adenocarcinoma |
| Basal cell adenocarcinoma | Oncocytic carcinoma |
| Cystadenocarcinoma | Carcinosarcoma |
| Sialoblastoma | Small cell carcinoma |
| Sebaceous carcinoma | Large cell carcinoma |
| Sebaceous lymph adenocarcinoma | — |
| Mammary analogue secretory carcinoma | — |
| Signet ring cell adenocarcinoma | — |
| Carcinoma arising in Warthin | — |
| Malignant transformation of ductal papilloma | — |
| Low-grade cribriform cystadenocarcinoma (intraductal carcinoma) | — |
Mucoepidermoid carcinoma
MEC is the most common salivary cancer affecting the parotid gland and comprising approximately 30% of malignant tumors. It occurs uniformly across all ages. It is composed of mucus-secreting, intermediate, and epidermoid (squamous) cells in varying proportion with low-grade MEC containing more cystic spaces lined with mucous cells and high-grade MEC containing more squamous cells ( Figs. 1–10 ).
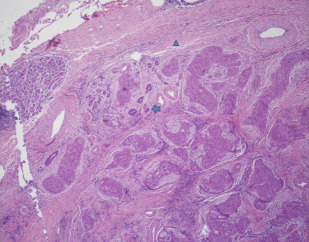
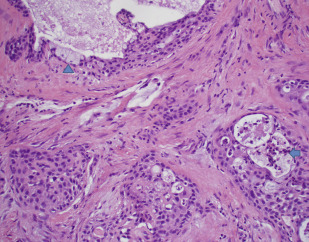
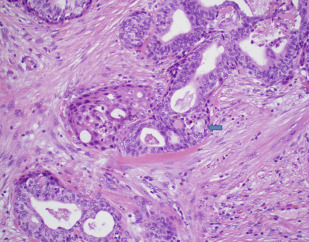
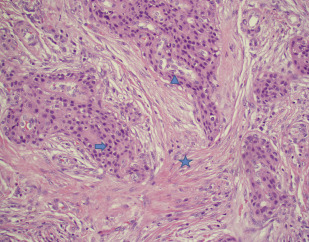
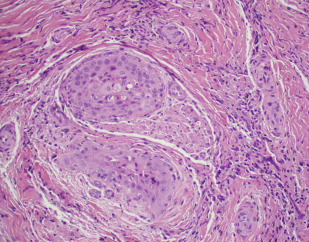
Several molecular markers have both diagnostic and prognostic value in MEC. These include a variety of membrane-bound secretory mucins. For example, MUC1 is associated with a high histologic grade, high rate of recurrence, metastasis, and short disease-free interval. MUC4, on the other hand, is expressed in low-grade MEC and is associated with a low recurrence rate and a long disease-free interval. In addition, the t(11;19) chromosomal translocation seems to be specific to MEC and predicts a better prognosis. The translocation results in CRTC1 (formerly MECT1)-MAML2 and CRTC3-MAML2 fusion proteins that disrupt the NOTCH signaling pathway. MAML2 rearrangements occur in up to 80% of MEC tumors and are more common in low-grade tumors. CRTC1/MAML2 translocation imparts a better prognosis even when found in high-grade MEC tumors. Epidermal growth factor receptor (EGFR) is also expressed in approximately two-thirds of MECs and may be associated with higher grade irrespective of MAML2 fusion status.
Prognosis is largely based on age, clinical stage, and grade. A lack of consistency among grading systems has led to a discrepancy in reports on prognosis for intermediate-grade MEC. This discrepancy can be explained by the use of multiple grading systems, including the Armed Forces Institute of Pathology (AFIP) system, Brandwein system, and the Modified-Healy classification. Chen and colleagues found that the Brandwein grading system ( Table 4 ) predicted low-grade behavior in intermediate-grade MEC; however, Aro and colleagues used the AFIP grading system and suggested intermediate-grade MEC be treated like high-grade tumors.
| Parameter | Points |
|---|---|
| Intracystic component<25% | +2 |
| Neural invasion | +3 |
| Necrosis | +3 |
| 4 or more mitoses/10 high-power field | +3 |
| Anaplasia | +2 |
| Lymphovascular invasion | +3 |
| Invasive front: small nest and islands | +2 |
| Bone invasion | +3 |
| Grade (G) | Sum of Points (Score) |
|---|---|
| Low (G1) | 0 |
| Intermediate (G2) | 2–3 |
| High (G3) | 4 or more |
Higher grade is associated with male sex, older age, larger tumors, locally aggressive tumors, and higher rates of nodal and distal metastases. A review of the Surveillance, Epidemiology, and End Results (SEER) database reveals a significant increase in nodal level I-III metastases with increasing grade (3.3%, 8.1%, and 34.0% in low-grade, intermediate-grade, and high-grade MEC, respectively). Furthermore, 8.9% of patients with high-grade MEC have level IV-V nodes. These findings suggest that treatment should be tailored to the grade and predicted behavior. Overall prognosis is favorable with a 5-year overall survival of 79% but depends upon grade.
Adenoid cystic carcinoma
ACCa is the second-most common cancer of the parotid gland; however, the incidence seems to be decreasing over time. ACCa typically occurs in between the fourth and sixth decade of life with a slight predisposition toward women. The behavior of ACCa is unique in that it is indolent, yet often highly fatal, with a propensity for neural and occult bony invasion. Neurotropism is a hallmark of ACCa. ACCa presents with pain and facial nerve dysfunction more often than other types of malignant salivary gland tumors. There is a strong correlation between pain and perineural invasion. Perineural invasion occurs in 29.2% to 62.5% of patients with ACCa and is associated with local tumor recurrence but not distant failure.
ACCa is a biphasic salivary gland tumor containing myoepithelial or basal cells with a minor population of epithelial or luminal cells growing in gland-like cystic architecture ( Figs. 11–13 ). Histologically, ACCa may easily be confused with low-grade adenocarcinoma, basal cell adenoma, basal cell adenocarcinoma, or cellular pleomorphic adenoma (PA). Three histologic growth types are appreciated in varying patterns: tubular, cribriform, and solid. A grading system was developed based on the percent solid component ranging from no solid component in grade 1 to less than 30% solid component in grade 2 and a predominantly solid component in grade 3. A solid pattern is associated with aggressive behavior and a higher rate of distant metastasis. The aggressive nature of the solid growth subtype is observed even in early stage I or II ACCa. Transformation of ACCa into poorly differentiated cribriform adenocarcinoma or solid, undifferentiated carcinoma has been described and portends much worse prognosis. The reported median survival in such cases is only 12 months.
Immunohistochemical markers for ACCa include nonspecific markers for basal cells and ductal cells (see Table 1 ) as well as CD43, a sialoglycoprotein that may assist in narrowing down the differential diagnosis. Ki-67 is both a diagnostic and prognostic marker found in ACCa. Neural cell adhesion molecule (NCAM) expression suggests ACCa and is associated with a higher incidence of perineural invasion. A unique tumor-specific translocation, t(6;9) (q22–23;p23–24), has been described. This translocation fuses the MYB oncogene to the transcription factor gene NFIB, leading to the potential activation of MYB targets, including genes associated with apoptosis, cell cycle control, and cell growth. The MYB-NFIB gene fusion is identified by fluorescence in situ hybridization (FISH) in 52.9% of tumors, although there is not always a chimeric transcript. MYB overexpression is correlated with poor survival.
Several growth factor receptor proteins and ligands have been identified within ACCa; however, none are specific to this particular disease. A significant number of cases have abnormalities in receptor tyrosine kinases (c-kit, EGFR, HER2/neu). C-kit is expressed in 80% to 90% of tumors but is not pathognomonic of ACCa. Although c-kit expression is common, gene mutations are rare, resulting in a poor response to tyrosine kinase inhibitors. There is no predictive value of c-kit mutation in terms of prognosis. In addition, phase II trials using imatinib in ACCa have been disappointing with significant toxicity. Similarly, phase II clinical trials with sunitinib, a multityrosine kinase inhibitor that includes vascular endothelial growth factor (VEGF), have failed to show any objective response.
The overall prognosis of ACCa varies but is generally poor due to late distant failures. Nodal metastases are rare, ranging between 3% and 10% in most studies. Conversely, distant metastases are common even after treatment with surgery and locoregional radiation, and usually occur in the lungs (80%) or bones (15%). Distant metastases occur in 31% of patients, and at least 20% of patients with early-stage disease will later develop metastases without local recurrence. Distant failures in early-stage ACCa present within a median of 31.5 months.
The indolent course is usually drawn out over 10 to 15 years. A review of the SEER database from 1973 to 2007 showed optimistic 5, 10, and 15 years overall survival rates of 90.3%, 79.9%, and 69.2%, respectively. Another study reports overall survival rates of 5, 10, and 20 years at 85.6%, 67.4%, and 50.4%, respectively, for patients without distant metastasis at presentation; and 69.1%, 45.7%, and 14.3%, respectively, for patients who presented with distant metastasis. Median survival after detection of metastasis is 36 months (range 1–112 months). Treatment of distant metastatic recurrences does not seem to offer any survival benefit based on historical data. Survival seems to be better for women and married persons. Older age (age>60 years), advanced stage, positive resection margin, high histologic grade, and high expression of Ki-67 were significantly correlated with poor prognosis.
Acinic cell carcinoma
Acinic cell carcinoma (AcCC) is a low-grade malignancy that accounts for 10% to 15% of parotid tumors. In the past, this was regarded as a benign tumor; however, late recurrences and metastases led to reclassification as a malignancy. AcCC may occur at any age; however, the mean age of occurrence tends to be younger than other parotid malignancies. Bilateral tumors are unusual but may occur.
The hallmark of AcCC is serous acinar cell differentiation; however, multiple cell types and histologic growth patterns are recognized ( Figs. 14–20 ). Thus, AcCC can present as several variants, which may confound the diagnosis. Cytologic diagnosis by fine-needle aspiration (FNA) hinges on the identification of acinar cells, which are more apparent in better differentiated AcCC, and the elimination of other histologies. DOG1 was recently described as a marker for acinar tissue and may aid in the diagnosis of AcCC. The rarity and indolent nature of AcCC has made it difficult to develop a grading system. Although AcCC generally follows an indolent course, it tends to be more aggressive when found in the parotid gland in comparison to the minor salivary glands.
AcCC is unpredictable in behavior. Prognosis is determined by recurrence and cervical lymph node metastases with some recurrences occurring late. Historically, recurrence was reported as high as 44%. In more recent studies, recurrence occurs in approximately one-third of patients with a disease-associated death rate of about 15%. This may reflect the difficulty in following patients out far enough to capture the late recurrences or, possibly, better surgical techniques. Stage, positive margins, local and neural invasion, high Ki-67 expression, desmoplasia, anaplasia, and a paucity of inflammation are also markers for worse prognosis.
Mammary analogue secretory carcinoma
Mammary analogue secretory carcinoma is a newly described salivary cancer that, as it sounds, is an analogue of AcCC but lacks acinar differentiation. More than 100 cases have been reported in the literature since its original description in 2010. Microscopically, the tumors have a lobulated growth pattern and are composed of microcystic and glandular spaces with abundant eosinophilic homogenous or bubbly secretory material positive for periodic acid-Schiff, mucicarmine, MUC1, MUC4, and mammaglobin. The neoplasms also show strong vimentin, S-100 protein, and STAT5a positivity. This tumor is unique for the chromosomal translocation, t(12;15) (p13;q25), which leads to a fusion gene between the ETV6 gene on chromosome 12 and the NTRK3 gene on chromosome 15. Data are limited on prognosis but suggests that these tumors are indolent similar to AcCC with a risk for late recurrences.
Salivary ductal carcinoma
Salivary ductal carcinoma (SDC) is rare but worth mentioning because it is notorious for its rapid and locally aggressive growth, most commonly in the parotid gland. SDC most commonly occurs in older men and is associated with poor prognosis. Histologically, SDC seems reminiscent of high-grade ductal carcinoma of the breast composed of pleomorphic, epithelioid cells with a cribiform growth pattern. In the authors’ institution, most cases seem to have arisen from a pre-existing PA, although de novo cases undoubtedly occur. Generally, cases arising in a PA may remain confined by the original capsule. Frequently, however, the tumor has extended beyond the capsule and is grossly infiltrative ( Fig. 21 ). The cut surfaces are solid and remarkable for foci of necrosis. Microscopically, marked pleomorphism and necrosis is the rule ( Fig. 22 ), rendering a rather straightforward cytologic diagnosis, provided the diagnosis is entertained ( Figs. 23–25 ).

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


