Purpose
To characterize the level of depression in patients with ocular inflammatory disease and to determine predictors of depression in this population.
Design
Prospective cross-sectional survey and medical record review.
Methods
Participants were consecutive patients with noninfectious ocular inflammatory disease in a university-based tertiary referral center. Subjects were given the self-administered Beck Depression Inventory-II (BDI-II), National Eye Institute Visual Function Questionnaire-25 (NEI VFQ-25), and additional supplemental questions. Medical records were reviewed for clinical characteristics. Univariate analyses were conducted to compare clinical characteristics between patients with and without a positive screen for depression, and a multivariate regression model was performed to determine the most significant predictors of depression.
Results
Of the 104 participants, 26.9% screened positive for depression with the BDI-II. Of these subjects, only 39.3% had been previously diagnosed with depression. NEI VFQ-25 scores were significantly lower in depressed patients in all subscales except driving and color vision. Predictors of depression were inadequate emotional support, lower visual functioning (VFQ composite score), history of changing immunomodulatory treatment, and current oral corticosteroid use.
Conclusions
Depression may be a significant but underrecognized comorbid condition in patients with ocular inflammatory disease. Worse visual function was associated with depression. The authors recommend heightened awareness of potential depression in patients with ocular inflammatory disease.
Uveitis affects people of all ages and accounts for up to 30 000 new cases of blindness annually and 10% of blindness in the United States. It has been shown to negatively impact both physical and mental health. Depressive symptoms and poorer self-reported visual functioning, as assessed with self-administered questionnaires, have been found in age-related macular degeneration, in retinitis pigmentosa, and in newly diagnosed glaucoma patients. However, to date, there have been few studies looking at depression and vision-related quality of life in patients with ocular inflammatory diseases.
For patients with ocular inflammatory diseases, in addition to the burden of the disease and potential for vision loss, there are several factors that may contribute to the development of depression. The drugs used to treat ocular inflammatory diseases, including corticosteroids and other immunosuppressive agents, can cause mania, depression, and other behavioral changes. Furthermore, in diseases with chronic inflammation there is evidence that cytokines are able to cross the blood-brain barrier and induce behavioral changes. Patients with chronic inflammatory diseases such as rheumatoid arthritis, atopy, Alzheimer disease, and multiple sclerosis can have significant comorbid depression disproportionate to disease-specific disability. A combination of these factors may place patients with ocular inflammatory diseases at a greater risk for depression. The purpose of this study was to determine the level of depression in patients with ocular inflammatory disease, as measured by a self-administered depression screening questionnaire, and the risk factors associated with depression, including self-reported visual functioning and clinical characteristics.
Material and Methods
Participants
Consecutive patients between March 29, 2010 and July 19, 2010 seen in the Uveitis and Ocular Inflammatory Disease Clinic at the Francis I. Proctor Foundation, a tertiary care referral center at the University of California, San Francisco, were included in the study. Eligible individuals included patients at least 18 years of age with a diagnosis of noninfectious ocular inflammatory disease, who were English-speaking and able to provide written informed consent. Patients seen at the Francis I. Proctor Foundation clinic undergo a standard evaluation, which includes screening for tuberculosis and syphilis as well as targeted evaluation for other infectious and noninfectious causes of ocular inflammatory disease.
Design and Procedures
After informed consent was obtained, participants completed a self-administered depression screening questionnaire and vision-related quality-of-life questionnaire, as well as a supplemental questionnaire. Participants were given the option to complete the questionnaires at the clinic or complete the questionnaires at home and return them by mail with a prepaid, self-addressed envelope. Medical records were reviewed for each patient in order to gather general demographic information on age, sex, and self-reported race. Additional chart review was conducted to gather information on type of ocular inflammatory disease (uveitis, scleritis, or mucous membrane pemphigoid), location of disease, chronicity and recurrence of disease, associated systemic diseases, current best-corrected visual acuity (BCVA), presence of active inflammation (defined as greater than 0.5+ anterior chamber cell, greater than 0.5+ vitreous haze, active vasculitis, retinitis, choroiditis, or active scleritis), and current and past treatments. The status (active or inactive), location, and chronicity of ocular disease were described and recorded as outlined by the Standardization of Uveitis Nomenclature criteria. Anatomic location of inflammation was noted as anterior, intermediate, posterior/panuveitis, scleritis, cicatrizing conjunctivitis, or orbital inflammation. The type of uveitis was noted as acute (new onset and singular event), recurrent (multiple episodes of inflammation marked by medication-free inactive periods of greater than 3 months), or chronic (persistent inflammation and inability to remain free of inflammation for at least 3 months without medications). History of changing immunomodulatory therapy for any reason was recorded. Immunomodulatory medications are typically prescribed to patients with ocular inflammation using a stepladder approach, starting with antimetabolites and adding T-cell inhibitors and biologics for refractory cases. Alkylating agents are also used in particularly severe cases. The necessity to change therapies may result from poorly controlled inflammation, inability to taper concomitant corticosteroids, intolerance to an agent because of undesirable side effects or safety concerns, discontinuation secondary to cost, or reduced effectiveness of an agent over time, as can happen with tumor necrosis factor α (TNF-α) inhibitors. Socioeconomic status, approximated by median household income, was determined by using geocodes ( http://www.ffiec.gov/Geocode/default.aspx , accessed July 20, 2010).
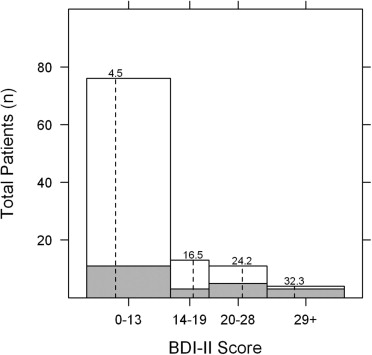
The Beck Depression Inventory II (BDI-II), a validated 21-item self-administered questionnaire, was used to measure depression in this study. Each question has 4 choices, ranging in point value from 0 to 3. Total scores of 0 to 13 represent no depression, 14 to 19 mild depression, 20 to 28 moderate depression, and 29 to 63 severe depression. Using a cutoff value of > 13 as a positive screen for depression yields 90% sensitivity and 99% specificity.
Vision-related quality of life was determined using the National Eye Institute Visual Function Questionnaire-25 (NEI VFQ-25), a self-administered 25-question survey. An overall composite score is generated with the lowest score of 0 to the highest score of 100, along with 12 subscales each with scores from 0 to 100 relating to general health, general vision, ocular pain, near activities, distance activities, social functioning, mental health, role difficulties, dependency, driving, color vision, and peripheral vision.
An additional supplementary questionnaire was created to capture information regarding past history of depression, level of perceived emotional support, knowledge about uveitis and participation in uveitis support groups, and smoking history.
Statistical Considerations
Conservatively assuming that 10% of incoming patients would be classified as depressed, a sample size of 100 patients was chosen to give us 80% power to detect a 20-point difference in composite NEI-VFQ score using a 2-sided t test. This estimate assumes a 95% confidence level and a pooled standard deviation of 21.6 points. If as many as 25% were classified as depressed, as seen in another study of chronic eye disease, we would be able to detect a somewhat smaller difference of 14 points.
Statistical analyses were conducted using R statistical software (The R Foundation for Statistical Computing, Vienna, Austria). The Fisher’ exact test was used to determine differences in categorical variables between the depressed and not-depressed groups, while continuous variables were analyzed using a 2-sided t test; all tests were performed at a 95% confidence level. Pearson correlations were included where appropriate. A backward stepwise linear regression model using all variables was used to determine significant predictors of BDI-II score. A backward stepwise logistic regression model using all variables was used to determine significant predictors of whether a subject screened positive or negative for depression according to BDI-II. Visual acuity using a Snellen eye chart was recorded individually for each eye and then converted into logarithm of the minimal angle of resolution (logMAR) scale. Low vision of count fingers, hand motion, light perception, and no light perception was recorded as logMAR 1.7, 1.8, 1.9, and 2.0, respectively.
Results
Of 151 eligible subjects for the study, 31 declined, 8 consented but did not mail in their questionnaires, and 8 consented but had incomplete questionnaires, resulting in 104 consecutive patients in the analysis. Twenty-eight subjects (26.9%) scored greater than 13 on the BDI-II (“depressed group”) and 76 subjects (73.1%) scored 13 or less (“not-depressed group”). There were no significant demographic differences between the depressed and not-depressed groups. Overall, 54.8% of participants were female and 49.0% were white ( Table 1 ). Inflammation-specific clinical characteristics were compared between the depressed and nondepressed groups. Twenty-four of the 28 subjects (85.7%) in the depressed group and 44 of the 76 subjects (57.9%) in the not-depressed group had chronic ocular inflammatory disease ( P = .01). Twenty-five subjects (89.3%) in the depressed group and 52 subjects (68.4%) in the not-depressed group had bilateral disease ( P = .04). Fifteen subjects (53.5%) in the depressed group and 16 subjects (21.1%) in the not-depressed group were currently taking oral corticosteroids ( P = .003). Twelve subjects (42.9%) in the depressed group and 13 subjects (17.1%) in the not-depressed group were currently taking an antimetabolite ( P = .01). Nine subjects (32.1%) in the depressed group and 6 subjects (7.9%) in the not-depressed group had been treated with more than 1 immunomodulatory therapy ( P = .004).
| Overall (N=104) | Depressed (N=28) | Not Depressed (N=76) | P Value | |
|---|---|---|---|---|
| Demographics | ||||
| Female | 57 (54.8%) | 16 (57.1%) | 41 (53.9%) | .83 b |
| Race | .01 b | |||
| White | 51 (49.0%) | 12 (42.9%) | 39 (51.3%) | |
| Hispanic | 11 (10.6%) | 8 (28.6%) | 3 (3.9%) | |
| Asian | 23 (22.1%) | 3 (10.7%) | 20 (26.3%) | |
| Black | 10 (9.6%) | 2 (7.1%) | 8 (10.5%) | |
| Indian subcontinent | 6 (5.8%) | 1 (3.6%) | 5 (6.6%) | |
| Native American | 2 (1.9%) | 1 (3.6%) | 1 (1.3%) | |
| Other | 1 (1.0%) | 1 (3.6%) | 0 (0.0%) | |
| Current smoker | 9 (8.7%) | 2 (7.1%) | 7 (9.2%) | .95 b |
| Median household income in dollars (IQR) | 94 293 (77 255, 121 348) | 80 268 (73 158, 105 766) | 95 919 (81 114, 125 831) | .06 c |
| Median age in years (IQR) | 41.0 (32.0, 51.3) | 41.5 (30.8, 51.3) | 40.5 (32.8, 50.5) | .79 c |
| Mean logMAR vision in the better eye (range) | 0.07 (-0.14, 0.6) | 0.12 (0, 0.54) | 0.05 (-0.14, 0.6) | .02 c |
| Any systemic inflammatory disease | 42 (40.4%) | 13 (46.4%) | 29 (38.2%) | .50 b |
| Ankylosing spondylitis | 12 (11.5%) | 1 (3.6%) | 11 (14.5%) | |
| Behçet disease | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Inflammatory bowel disease | 2 (1.9%) | 1 (3.6%) | 1 (1.3%) | |
| Juvenile idiopathic arthritis | 1 (1.0%) | 1 (3.6%) | 0 (0%) | |
| Mucous membrane pemphigoid (MMP) | 3 (2.9%) | 2 (7.1%) | 1 (1.3%) | |
| Multiple sclerosis | 1 (1.0%) | 1 (3.6%) | 0 (0%) | |
| Parry Romberg syndrome | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Reactive arthritis | 2 (1.9%) | 1 (3.6%) | 1 (1.3%) | |
| Rheumatoid arthritis | 2 (1.9%) | 1 (3.6%) | 1 (1.3%) | |
| Rosacea | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Sarcoidosis | 7 (6.7%) | 1 (3.6%) | 6 (7.9%) | |
| Systemic lupus erythematosus | 2 (1.9%) | 0 (0%) | 2 (2.6%) | |
| Tubulointerstitial nephritis and uveitis | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Vogt-Koyanagi-Harada syndrome | 6 (5.8%) | 4 (14.3%) | 2 (2.6%) | |
| Clinical course | ||||
| Median duration of disease in years (IQR) | 5.8 (2.0, 12.0) | 3.9 (1.6, 9.3) | 6.3 (3.0, 12.3) | .18 c |
| Chronic | 68 (65.4%) | 24 (85.7%) | 44 (57.9%) | .01 b |
| Bilateral | 77 (74.0%) | 25 (89.3%) | 52 (68.4%) | .04 b |
| Location of inflammation | .01 b | |||
| Anterior | 49 (47.1%) | 8 (28.6%) | 41 (53.9%) | |
| Intermediate | 8 (7.7%) | 0 (0%) | 8 (10.5%) | |
| Anterior + intermediate | 11 (10.6%) | 6 (21.4%) | 5 (6.6%) | |
| Posterior/pan | 27 (25.9%) | 11 (39.2%) | 16 (21.1%) | |
| Scleritis | 4 (3.8%) | 1 (3.5%) | 3 (3.9%) | |
| Conjunctiva (MMP) | 3 (2.9%) | 2 (7.1%) | 1 (1.3%) | |
| Cornea (peripheral ulcerative keratitis) | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Orbit | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Current active inflammation | 26 (25.0%) | 7 (25.0%) | 19 (25.0%) | .99 b |
| Current treatment | ||||
| Oral corticosteroid | 31 (29.8%) | 15 (53.5%) | 16 (21.1%) | .003 b |
| Topical corticosteroid | 51 (49.0%) | 13 (46.4%) | 38 (50.0%) | .92 b |
| Antimetabolite | 25 (24.0%) | 12 (42.9%) | 13 (17.1%) | .01 b |
| Methotrexate | 13 (12.5%) | 6 (21.4%) | 7 (9.2%) | |
| Mycophenolate mofetil | 11 (10.6%) | 6 (21.4%) | 5 (6.6%) | |
| Azathioprine | 1 (1.0%) | 0 (0%) | 1 (1.3%) | |
| Cyclosporine | 1 (1.0%) | 1 (3.6%) | 0 (0%) | .27 b |
| Cyclophosphamide | 2 (1.9%) | 2 (7.1%) | 0 (0%) | .07 b |
| Biologic | 10 (9.6%) | 3 (10.7%) | 7 (9.2%) | .99 b |
| Infliximab | 6 (5.8%) | 1 (3.6%) | 5 (6.6%) | |
| Adalimumab | 4 (3.8%) | 2 (7.1%) | 2 (2.6%) | |
| History of changing immunomodulatory therapy | 15 (14.4%) | 9 (32.1%) | 6 (7.9%) | .004 b |
a Beck Depression Inventory II: patients in the depressed group scored >13 and patients in the not-depressed group scored ≤13.
b Analyzed using a Fisher exact test at a confidence level of 95%.
c Analyzed using a 2-sided t test at a confidence level of 95%.
In the depressed group, 13 patients had a BDI-II score of 14 to 19 (mild depression), 11 patients had a score of 20 to 28 (moderate depression), and 4 patients had a score > 28 (severe depression). The mean BDI-II score in the depressed group was 21.8 (range 14–40) and the mean BDI-II score in the not-depressed group was 4.5 (range 0–12) ( Figure 1 ) . The mean logMAR vision in the better-seeing eye was 0.12 (Snellen equivalent 20/25−1) in the depressed group and 0.05 (Snellen equivalent 20/25+2) in the not-depressed group ( P = .02) ( Table 1 ). The mean composite NEI VFQ-25 scores for the depressed and not-depressed groups were 63.6 and 85.6, respectively ( P < .001) ( Table 2 ). All subscale scores were significantly lower in the depressed group except for driving and color vision. There was a moderate inverse correlation between BDI-II score and VFQ composite score (Pearson correlation −0.53, P < .001) and between logMAR BCVA in the better-seeing eye and VFQ composite score (Pearson correlation −0.58, P < .001) ( Figures 2 and 3 ) . Among patients with anterior, anterior + intermediate, or posterior/panuveitis, logMAR BCVA in the better-seeing eye was slightly lower in the depressed group but not significantly different ( Table 3 ). VFQ composite scores were significantly lower for depressed patients with anterior + intermediate ( P = .006) and posterior/panuveitis ( P = .03), but not for depressed patients with strictly anterior uveitis ( P = .30).
| Overall (N=104) | Depressed (N=28) | Not Depressed (N=76) | P Value c | |
|---|---|---|---|---|
| Mean composite score (range) | 79.7 (16, 100) | 63.6 (16, 98.1) | 85.6 (31.1, 100) | <.001 |
| Mean subscale scores (range) | ||||
| General health | 60.3 (0, 100) | 46.4 (0, 75) | 65.5 (0, 100) | <.001 |
| General vision | 72.8 (20, 100) | 65.2 (20, 100) | 75.5 (20, 100) | .03 |
| Ocular pain | 73.9 (0,100) | 60.3 (12.5, 100) | 78.9 (0, 100) | .002 |
| Near activities | 79.2 (16.7, 100) | 61.6 (16.7, 100) | 85.6 (16.7, 100) | <.001 |
| Distance activities | 78.8 (8.3, 100) | 59.8 (8.3, 100) | 85.7 (33.3, 100) | <.001 |
| Social functioning | 89.9 (25, 100) | 76.3 (25, 100) | 94.9 (37.5, 100) | <.001 |
| Mental health | 70.8 (0, 100) | 44.0 (0, 93.8) | 80.7 (12.5, 100) | <.001 |
| Role difficulties | 74.2 (0, 100) | 50.9 (0, 100) | 82.7 (0, 100) | <.001 |
| Dependency | 84.9 (8.3, 100) | 66.4 (8,3, 100) | 91.8 (25, 100) | <.001 |
| Driving | 77.4 (0, 100) | 62.0 (0, 100) | 82.4 (0, 100) | .007 |
| Color vision | 94.9 (25, 100) | 91.7 (25, 100) | 96.1 (25, 100) | .24 |
| Peripheral vision | 81.3 (0, 100) | 66.1 (0, 100) | 86.8 (25, 100) | .002 |
a Assessed by the National Eye Institute Visual Function Questionnaire-25.
b Beck Depression Inventory II: patients in the depressed group scored >13 and patients in the not-depressed group scored ≤13.
c Analyzed using a 2-sided t test at a confidence level of 95%.
| Overall | Depressed | Not Depressed | P Value c | |
|---|---|---|---|---|
| Anterior | N=49 | N=8 | N=41 | |
| Mean VFQ-25 composite score (range) | 85.2 (21.2, 100) | 76.1 (21.2, 98.1) | 86.9 (33.1, 100) | .30 |
| Mean logMAR vision in the better eye (range) | 0.04 (−0.14, 0.48) | 0.07 (0, 0.28) | 0.03 (−0.14, 0.60) | .37 |
| Intermediate | N=8 | N=0 | N=8 | |
| Mean VFQ-25 composite score (range) | 85.6 (60.8, 100) | NA | 85.6 (60.8, 100) | NA |
| Mean logMAR vision in the better eye (range) | 0.09 (0, 0.30) | NA | 0.09 (0, 0.30) | NA |
| Anterior + intermediate | N=11 | N=6 | N=5 | |
| Mean VFQ-25 composite score (range) | 68.3 (27.1, 94.2) | 54.4 (27.1, 69.8) | 85.0 (64.8, 94.2) | .006 |
| Mean logMAR vision in the better eye (range) | 0.15 (0, 0.40) | 0.20 (0, 0.40) | 0.08 (0.02, 0.10) | .12 |
| Posterior/panuveitis | N=27 | N=11 | N=16 | |
| Mean VFQ-25 composite score (range) | 73.7 (15.9, 99.4) | 62.7 (15.9, 82.1) | 81.3 (31.1, 99.43) | .03 |
| Mean logMAR vision in the better eye (range) | 0.09 (−0.04, 0.60) | 0.11 (0, 0.54) | 0.07 (−0.04, 0.60) | .54 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


