Purpose
To define a minimum set of outcome measures for tracking, comparing, and improving macular degeneration care.
Design
Recommendations from a working group of international experts in macular degeneration outcomes registry development and patient advocates, facilitated by the International Consortium for Health Outcomes Measurement (ICHOM).
Methods
Modified Delphi technique, supported by structured teleconferences, followed by online surveys to drive consensus decisions. Potential outcomes were identified through literature review of outcomes collected in existing registries and reported in major clinical trials. Outcomes were refined by the working group and selected based on impact on patients, relationship to good clinical care, and feasibility of measurement in routine clinical practice.
Results
Standardized measurement of the following outcomes is recommended: visual functioning and quality of life (distance visual acuity, mobility and independence, emotional well-being, reading and accessing information); number of treatments; complications of treatment; and disease control. Proposed data collection sources include administrative data, clinical data during routine clinical visits, and patient-reported sources annually. Recording the following clinical characteristics is recommended to enable risk adjustment: age; sex; ethnicity; smoking status; baseline visual acuity in both eyes; type of macular degeneration; presence of geographic atrophy, subretinal fibrosis, or pigment epithelial detachment; previous macular degeneration treatment; ocular comorbidities.
Conclusions
The recommended minimum outcomes and pragmatic reporting standards should enable standardized, meaningful assessments and comparisons of macular degeneration treatment outcomes. Adoption could accelerate global improvements in standardized data gathering and reporting of patient-centered outcomes. This can facilitate informed decisions by patients and health care providers, plus allow long-term monitoring of aggregate data, ultimately improving understanding of disease progression and treatment responses.
Macular degeneration is a leading cause of irreversible vision loss, accounting for over 15% of blindness in high-income countries, with the burden of this disease expected to increase with aging populations. Age-related macular degeneration (AMD) is the most prominent macular degeneration in populations of European descent. Even in Asian populations, who are known to have a lower risk of AMD than people of European descent, AMD is one of the leading causes of blindness. Although non-neovascular AMD is much more common, untreated neovascular AMD used to frequently be responsible for severe vision loss. Less common types of neovascular macular degeneration include those secondary to myopia, trauma, inflammation, macular telangiectasia, and idiopathic causes, all of which, when associated with choroidal neovascularization (CNV), are broadly treated with a similar approach as that for neovascular AMD. Intravitreal anti–vascular endothelial growth factor (anti-VEGF) therapy has become the established treatment of neovascular AMD over the last decade, supported by evidence from pivotal randomized clinical trials.
Interventional clinical trials are designed to maximize the likelihood of demonstrating a treatment effect and therefore often use a study population that may be significantly different from the general population of patients with the disease in question. For these and other reasons, including less frequent treatment and less regular follow-up than is inherent in a clinical trial, outcomes achieved in the pivotal trials are not always replicated in routine clinical practice. Interventional clinical trials also suffer from relatively small numbers of patients who are treated and short duration of follow-up, with only 3 major prospective studies reporting data beyond 2 years of treatment. This is particularly problematic with macular degeneration being a lifelong disease, for which the long-term outcomes, especially with treatment, are less well understood.
Various regimens have been developed to address the vast burden of treatment for neovascular macular degeneration. Monthly dosing, which was initially recommended, has been largely supplanted by treat-and-extend or variable as-required (pro re nata) dosing. Decisions regarding initiating treatment and retreatment vary internationally and according to reimbursement schemes, but are generally governed by changes in visual acuity and CNV activity as determined by the detection of intraretinal and subretinal fluid using optical coherence tomography (OCT). There is also increasing choice in anti-VEGF agents (ranibizumab, bevacizumab, aflibercept) and numerous potential treatments for non-neovascular AMD, which are currently being evaluated in clinical trials.
The limitations of clinical trials, variation in treatment approaches and therapies, and ever-growing financial burden of providing long-term treatment despite uncertain long-term gains indicate that the need for systematic measurement of the outcomes of macular degeneration treatment in routine clinical practice is greater than ever. However, only a few care providers around the world routinely record outcome data for the treatment they give to patients for macular degeneration outside of clinical trials. There are, however, emerging “registries”—national or multinational data repositories that assimilate clinical outcome data from large numbers of individual practices. These include the significant projects originating in Sweden (The Swedish Macula Register), Australia (The Fight Retinal Blindness! Project), the United Kingdom (The UK National Ophthalmology Database), and the Czech Republic (The Amadeus Project). The largest is the Luminous Project, which has recently completed enrollment of 30 000 patients across 5 continents (including up to 10% of patients undergoing intravitreal anti-VEGF treatment for diabetic macular edema and retinal vein occlusion); however, this only includes patient receiving ranibizumab. In the United States, the American Academy of Ophthalmology Intelligent Research in Sight (IRIS) project, which was launched in 2014, is the first comprehensive eye disease clinical registry, with macular degeneration outcome measures in development.
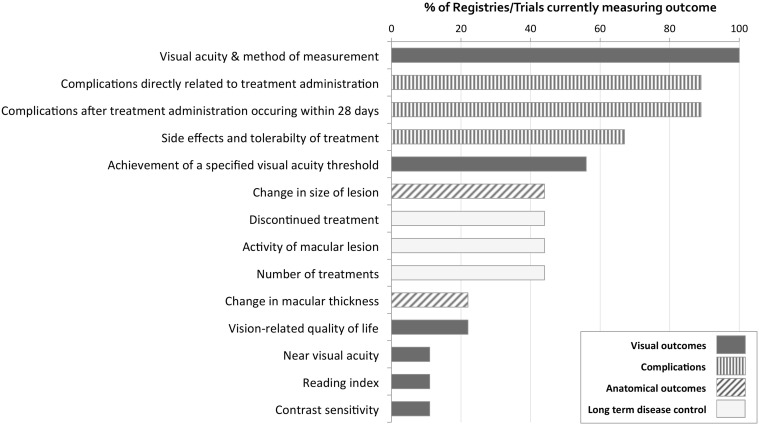
Although existing efforts to measure outcomes of macular degeneration treatment from routine clinical care are a good start, there is no agreed standard approach, and therefore significant variation exists in what outcomes are recorded as well as how they are measured and reported. An overview of the frequency at which main outcomes are currently being measured in existing registries and major clinical trials is provided in Figure 1 . The figure shows that visual acuity and complications of treatment are commonly included. However, long-term disease control and anatomic outcomes are tracked in fewer than half of the registries and trials. The absence of an agreed standard approach limits the ability to perform direct comparisons of different services that could lead to improved outcomes. Most importantly, functional outcomes that are more meaningful to patients may be underrepresented compared to clinical or anatomic outcomes, which are easier to measure but may bear less direct relevance to patients.
Standardized and patient-centered outcome measurement is therefore crucial in order to direct improvements by those providing treatment, promote dissemination of best practices, and ultimately drive competition around quality. Systematic studies of interventions for other diseases in routine practice have exposed significant variation in outcomes that appeared to be dependent on differences in institutional methods, physician preference, or health care systems. Continued data collection and outcome reporting has subsequently led to clear improvements in these outcomes. For these reasons, a Working Group came together, facilitated by the International Consortium for Health Outcomes Measurement (ICHOM, a nonprofit organization with the purpose to transform health care systems worldwide by measuring and reporting patient outcomes in a standardized way), to develop an international, standardized, core set of patient-centered outcomes measures, with common definitions for routine clinical data collection in macular degeneration care. It is hoped that the minimum set of outcomes that we propose will be used as a common platform for routine clinical data collection by those providing care for patients with macular degeneration.
Methods
The ICHOM approach to defining health outcomes for conditions involves forming an international working group that follows a structured approach to compile the key outcomes and clinical characteristics that are recommended to be measured in routine clinical practice, called the “Standard Set.” The work was supported by sponsorship from a number of charitable health care organizations, but has no financial support from any pharmaceutical or health care technology organizations.
International Working Group
ICHOM convened a working group of 18 members, representing 10 different countries from 4 continents ( Figure 2 ). The working group consisted of leading experts in the fields of macular degeneration outcomes and health care registry development, as well as patient advocates. The working group lead, Professor Mark Gillies, has previously led the development of the collaborative Fight Retinal Blindness! (FRB!) Project, which is an efficient, web-based system to track outcomes of patients receiving treatment for neovascular AMD in clinical practice. A project leader from ICHOM (S.S.) managed the project, and an ICHOM Research Fellow (I.R.) supported the content development. The other ophthalmologists among the working group had experience in leading the retinal services in internationally recognized ophthalmic units and established clinical data registries in North America, Europe, and Asia. The patient representatives had direct experience of treatment for macular degeneration and were involved in national charitable organizations for macular disease, which gave them access to the insight of thousands of other patients with macular degeneration.
Development of Standard Set
A modified Delphi technique was used to develop these recommendations, in which a sequence of rounds of discussion followed by an electronic survey were used to arrive at a consensus among the experts who had differing views and perspectives. This enabled the input from participants to be gathered without requiring them to work face-to-face to find consensus.
Six teleconferences were used to discuss outcomes and clinical characteristics for inclusion in the Standard Set. The first teleconference focused on the scope and outcome domains for the Standard Set, supported by a literature review of existing registries and major clinical trials to produce a long list of outcome measures that were being recorded at the time.
The long list of potential outcomes was refined through consensus discussions steered by the working group lead in order to prioritize outcomes that (1) had direct impact on patients; (2) were sensitive to good clinical care; and (3) were feasible to measure in routine clinical practice.
Following the teleconference, members submitted their feedback and final votes on the inclusion of each outcome through electronic surveys. A 75% majority of initial votes was used as the threshold for inclusion of the outcome, and below 50% was the threshold for rejecting outcomes. In cases where a particular outcome received votes for inclusion between 50% and 75%, the contentious point was revisited on the next call and survey. The second teleconference covered the measurement and definition of the selected outcomes.
A similar process was undertaken in teleconferences 3 and 4 to identify the clinical characteristics that are recommended to be measured in order to risk-stratify patients in subsequent analyses. The measurement and definition of the selected clinical characteristics was also agreed upon. Publication, adoption, and implementation strategies were discussed in teleconferences 5 and 6. The final Standard Set was refined and approved unanimously by all members of the working group.
Results
A summary of the ICHOM macular degeneration Standard Set is shown in Tables 1–3 , with additional details in the sections below. A data collection manual that further describes each measure, its definition, suggested reporting format, inclusion and exclusion criteria, and potential data sources is freely available on the ICHOM Website ( http://www.ichom.org/medical-conditions/macular-degeneration/ ).
| Conditions Included | Neovascular macular degeneration:
|
| Treatments Included |
|
| Measure | Details | Timing | Data Source | |
|---|---|---|---|---|
| Visual functioning and vision-related quality of life | Distance visual acuity | Distance visual acuity (best of uncorrected, corrected, or pinhole) in the affected eye. Change in distance visual acuity should be calculated from baseline and previous visual acuity assessments. |
| Clinical data |
| Mobility and independence | Impact of Vision Impairment questionnaire |
| Patient reported | |
| Emotional well-being | Impact of Vision Impairment questionnaire |
| Patient reported | |
| Reading and accessing information | Impact of Vision Impairment questionnaire |
| Patient reported | |
| Disutility of care | Number of treatments | Documentation of individual treatments received for macular degeneration ( Table 1 ) |
| Clinical or administrative data |
| Complications of treatment a | Endophthalmitis: Severe intraocular inflammation within 3 months of last intraocular treatment, due to infectious or noninfectious causes |
| Clinical data | |
| Disease control | Presence of fluid, edema, or hemorrhage a | Presence of intraretinal or subretinal fluid or hemorrhage that is attributable to activity of the neovascular lesion as determined by the treating ophthalmologist. This could be based on clinical examination or imaging. |
| Clinical data |
a Outcomes applicable to neovascular macular degeneration only.
| Measure | Details | Timing | Data Source | |
|---|---|---|---|---|
| Patient demographics | Age | Age in years, calculated from birth date at commencement of therapy |
| Clinical or patient reported |
| Sex | Sex at birth |
| Clinical or patient reported | |
| Ethnicity | Asian, Black, Hispanic, white, mixed, other |
| Patient reported | |
| Smoking status | Smoking status (of cigarettes, cigars, or tobacco) |
| Patient reported | |
| Baseline functional status | Baseline visual acuity | Distance visual acuity (best of uncorrected, corrected using glasses or contact lenses, or pinhole) in the affected eye |
| Clinical data |
| Baseline visual acuity in the fellow eye | Distance visual acuity (best of uncorrected, corrected using glasses or contact lenses, or pinhole) in the fellow eye |
| Clinical data | |
| Clinical status | Type of macular degeneration | Type of macular degeneration as defined in Table 1 |
| Clinical data |
| Geographic atrophy | Presence of geographic atrophy anywhere in the macular area that is not contiguous with the main lesion |
| Clinical data | |
| Subretinal fibrosis | Presence of subretinal fibrosis anywhere in the macular area |
| Clinical data | |
| Pigment epithelial detachment | Presence and type of pigment epithelial detachment anywhere in the macular area |
| Clinical data | |
| Associated clinical history | Previous macular degeneration treatment | Previous macular degeneration treatment in affected eye (multiple options possible) |
| Clinical or administrative data |
| Ocular comorbidities | Ocular comorbidities including retinal vascular disease, other macular pathology, glaucoma or optic neuropathy, amblyopia, or medial opacity in affected eye (multiple options possible) |
| Clinical or administrative data | |
| Additional interventions to be recorded |
|
| Clinical or administrative data | |
Conditions and Treatment Approaches Covered
The Standard Set was developed to include the broad disease categories of macular degeneration and their treatments that were deemed to be sufficiently different to warrant separation, as listed in Table 1 . Other forms of non-neovascular macular degeneration were excluded owing to the large variation in the pathogenesis, prognosis, and treatment of such diseases, which would therefore require measurement of different clinical characteristics and outcomes.
Outcomes Recommended in the International Consortium for Health Outcomes Measurement Macular Degeneration Standard Set
A large number of potential outcomes were considered by the working group before agreement on the final minimum set of outcomes that are recommended for the affected eye, as detailed below. Further details of these potential outcomes, including the final voting decisions and summary of the working group discussions that led to a decision of inclusion or exclusion of individual outcomes, are included in Supplemental Table 1 (Supplemental Material available at AJO.com ).
Visual Functioning and Vision-related Quality-of-Life Outcomes
Because improving or maintaining visual function is the primary goal for most patients with macular degeneration, it is clearly an essential component of any outcome assessment. We recommend recording of distance visual acuity (best of uncorrected, corrected using glasses or contact lenses, or pinhole if required) in the affected eye at each clinical visit. Results should preferably be measured using a logarithm of the minimal angle of resolution (logMAR) or ETDRS chart, although other measurement systems may be used and subsequently converted to logMAR visual acuity.
When analyzing distance visual acuity outcomes of treated eyes, we recommend reporting the following data:
- •
Mean change in visual acuity from baseline
- •
Proportion of eyes gaining vision (≥5 logMAR letters)
- •
Proportion of eyes with stable vision (within 15 logMAR letters of baseline)
- •
Proportion of eyes with visual acuity of ≤0.3 logMAR (20/40 Snellen)
- •
Proportion of eyes with visual acuity of ≥1.0 logMAR (20/200 Snellen)
We chose to recommend measuring mean change in visual acuity after starting treatment because it has become the primary outcome of phase III clinical trials for neovascular AMD. However, observational studies have found that, because of ceiling effects, this may skew results in favor of services that detect the disease and start treatment late, since eyes with worse vision have more to gain compared with eyes starting with good vision, which may not gain anything. For this reason we recommend that the proportion of eyes with stable vision, good vision (≥20/40), and poor vision (≤20/200) should also be measured.
There is mounting evidence that increases in objectively measured distance visual acuity do not necessarily concur with improved visual functioning for patients. Therefore, to fully measure the impact of macular degeneration treatment, it is important to also assess how the patient’s vision-related quality of life has changed. The working group felt that measures of visual function other than distance visual acuity, such as near visual acuity and reading speed, were important outcomes. However, rather than requiring these to be captured as clinical data in a clinical setting, they recommended them to be measured collectively via subjective patient reporting from real life via a patient-reported outcome measure (PROM). The working group felt that use of PROM data would provide a superior reflection of the visual function of patients in the real world than clinical measures such as contrast sensitivity and that PROM data would be of use in directing treatment.
We evaluated a number of existing, validated PROMs that are potentially suitable for assessment of visual function affected by macular degeneration. These included the Daily Living Tasks Dependent on Vision (DLTV), Impact of Vision Impairment (IVI), Macular Disease Quality of Life (MacDQoL), Metamorphopsia Questionnaire, and the National Eye Institute Visual Functioning Questionnaire (NEI-VFQ), which have been recently reviewed by Khadka and associates. The criteria that we used to evaluate all the relevant PROMs consisted of the following: (1) content of the PROM (based on domains of visual function that the working group felt were most important to patients with macular degeneration, including work/hobby continuation, face recognition/social participation, driving/transport, reading, self-care, general enjoyment of life, activities involving near vision, activities involving distance vision, bodily symptoms/functions); (2) quality of the PROM (psychometric properties, content development, reliability, and validity specifically for macular disease); and (3) practical aspects related to using the PROM (available languages, time taken to complete, costs involved with using in routine clinical care).
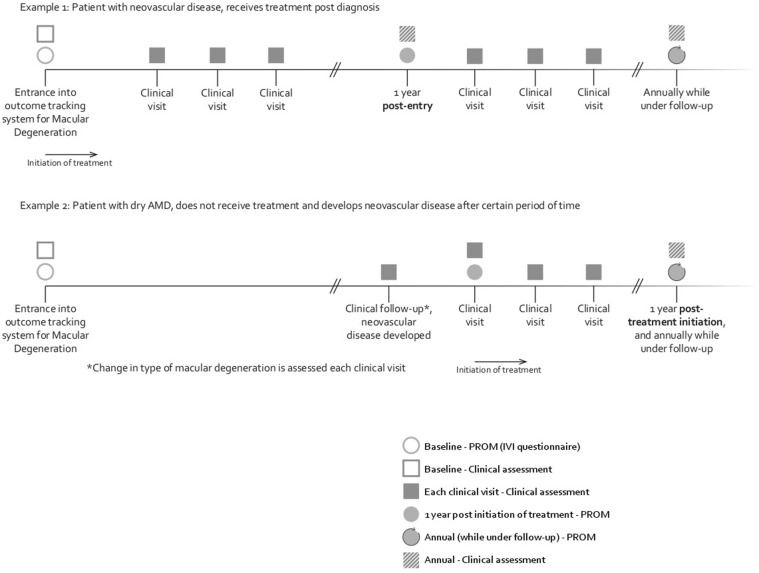
Based on our extensive evaluation and discussions, we recommend the use of the Impact of Vision Impairment (IVI) questionnaire at baseline, prior to initiating treatment and annually while under follow-up. The importance of follow-up assessment is to quantify changes in response to treatments and to allow comparison between different care providers. Annual reassessment has been recommended to minimize data collection burden but enable meaningful changes to be detected in a reasonable period of time. The IVI has been validated specifically in patients with AMD and found to have appropriate content development and reliability, plus has undergone Rasch analysis. It also scores highly on its psychometric properties, including unidimensionality within each domain and high measurement precision.
In terms of content, the IVI consists of 28 items that cover an appropriate and broad range of questions that comprise 3 domains: “mobility and independence,” “emotional well-being,” and “reading and accessing information.” The questions assess bodily symptoms and functions, visual tasks and activities (in particular, those requiring near vision), social participation, and emotional well-being, which were all felt to be true outcomes of high importance and relevance for patients with macular degeneration. We do recognize that other PROMs are widely used and can provide valuable information; however, our decision to recommend the IVI was to ensure that all these important patient needs can be captured.
In practical terms, the IVI is also free to use (for noncommercial purposes) and takes approximately 15 minutes to complete using paper, computer, or touch screen–adapted versions. A guide to scoring and analysis of the IVI is included in the ICHOM Macular Degeneration Standard Set Reference Guide (available at http://www.ichom.org/medical-conditions/macular-degeneration/ ). It has validated translations from the original English version into a number of languages including Chinese, German, Melanesian, Hindi, and Telugu. We recognize that widespread adoption will also require additional translations to ensure comparability across populations. The developers of the IVI are willing to provide advice on the process of translation and validation if needed.
Disutility of Care Outcomes
The burden of care to patients was deemed to be important to measure, as the frequent and regular treatments could have a negative impact on the lives of patients and of those who care for them, in terms of time and money as well as emotionally. It is also possible that the number of treatments received may not be the same as the number that would be recommended if the logistics of clinic capacity and frequent patient visits were not an issue or if there were no financial constraints. We therefore recommend that each individual treatment received for macular degeneration should be recorded at each clinical visit.
Complications related to treatment received for macular degeneration, although occurring infrequently, are of significant concern to patients. The most serious complication of intravitreal injections is endophthalmitis. We therefore recommend capturing the incidence of severe intraocular inflammation within 3 months of the intraocular treatment due to infectious or noninfectious causes. This outcome is applicable for all patients undergoing treatment for neovascular macular degeneration.
Other complications of treatment, such as retinal pigment epithelium tears and retinal detachment, were considered by the working group. However, they were rejected from this minimum outcome set on the basis that these were idiosyncratic complications, with potential variability in detection rates. Further information on the rationale of the working group decisions is included in Supplemental Table 1 .
Disease Control Outcomes
Long-term control of disease was judged to be both significant to patients and often a reflection of the treatment provided. For all types of neovascular macular degeneration, we recommend reporting at each clinical visit the presence of intraretinal or subretinal fluid or hemorrhage that is attributable to activity of the neovascular lesion as determined by the treating ophthalmologist (based on clinical examination or imaging).
Clinical measures such as central retinal thickness or size of lesion were not included because these were not felt to be outcomes of treatment that were important to patients. A summary of all the outcomes that are recommended to be recorded is presented in Table 2 .
Clinical Characteristics and Interventions
In order to make meaningful outcome comparisons between patients, it is important to measure certain baseline and follow-up characteristics to enable subsequent appropriate risk adjustment. The full list of potential clinical characteristics that were considered are provided in Supplemental Table 2 (Supplemental Material available at AJO.com ), along with the final voting decisions of the working group on inclusion or exclusion of individual clinical characteristics. A number of clinical characteristics where there is good evidence of their impact on clinical outcomes following treatment for macular degeneration were discussed and finally selected. These are summarized in Table 3 , grouped under the categories of patient demographics, baseline functional status, clinical status, and associated clinical history. Systemic comorbidities such as cerebrovascular and cardiovascular disease were not included to minimize data collection burden. Similarly, the suggested reporting format for these characteristics and interventions has been simplified to facilitate data collection while still providing useful information.
We also recommend recording of interventions to the affected eye that are likely to have an impact on the outcomes that are recommended to be collected, in particular visual functioning and vision-related quality-of-life outcomes. These are also listed in Table 3 .
Data Collection
The timeline for the suggested recording of the different categories of outcomes and clinical characteristics is summarized in Figure 3 . We also suggest prompts at each clinical visit to check if certain clinical characteristics have changed and if any other relevant interventions have occurred, as indicated in Table 3 . We envisage that this would be performed by some form of electronic patient record and that the prompting, rather than have more mandatory fields, would reduce data collection burden.