Cysts and Celes
Developmental Cysts
Dermoid Cysts
This benign tumor runs the gamut from a small, superficial, surgically accessible, easily eradicable lesion to a large, deeply recessed, relentlessly expanding, bone-contouring mass. Because of these frequent extraorbital ramifications, this tumor may defy intact removal. The age range of affected patients is wide, extending from infancy through middle adult life.
Terminology and Classification
The exact pathogenesis of the orbital cyst is not known, but its origin is probably a nidus of embryonic ectoderm entrapped by the juxtaposition of membranous bones and epithelial fusion lines. In this regard, the cyst is a choristoma.
The small, anterior orbital cyst is usually adjacent to the frontolacrimal, frontoethmoidal, or frontozygomatic suture. The larger, more posterior cyst is usually associated with the sphenozygomatic suture and less often with the sphenoethmoidal suture. The origin of the deeper cyst may be obscure, because its base is masked by the large size of the mass, the cyst ruptures during the course of surgical manipulation, or the pedicle of the lesion has atrophied during the long course of development.
At the time of our first edition (1973), this tumor was usually classified on the basis of histologic characteristics into dermoid and epidermoid types. This classification might have been artificial because a tissue specimen from a deeply positioned cyst would be so small or so fragmented that a definitive histologic differentiation was not possible. In addition, some clinicians have argued that histologic classification is academic, inasmuch as complete removal of tumor is the goal of management regardless of histology. In the 1980s, Grove (1981) and several other authors suggested that because the histology of epidermoid and dermoid cysts is so similar, they should be considered as one tumor type, classified on the basis of their anatomic location, superficial or deep. Accordingly, in our third edition, the two histologic types were tabulated as dermoid cysts.
The evolution of high-resolution computed tomography (CT) scan and magnetic resonance imaging (MRI) in the 1990s provided more precise indications about the location of the dermoid cyst in relation to the bone as well as the origin from orbital soft tissue. This is the basis for expanded anatomic classification (Shields et al., 1997) that should improve the management of these cysts. Shields et al. (1997) broadly divided the cysts into juxtasutural, sutural, and soft tissue types.
The juxtasutural type is attached to the bone by dense fibrous bands but does not affect the bone unless unchecked tumor growth causes fossa formation. The cysts presenting along the orbital rim are usually of this type. The nidus of the sutural type is within the synostosis of the orbital bone (intradiploic). The cyst may expand within the cancellous tissue of the bone but more often extends into the soft tissues. The sutural type is associated with bony defects, draining sinuses of the orbital bone (Honig, 1998), and extension into the intracranial cavity (Niederhagen et al., 1998; Meyer et al., 1999). Giant dermoid cysts reported in the literature often are of this type. The soft tissue type includes rarer cysts arising from soft tissues of the orbit (Howard et al., 1994; Atilla et al., 2000; Coevoet et al., 2000). The surgical management related to this classification is discussed later in this section.
Bonavolonta et al. (1995) classified their 145 patients, studied over 16 years, as exophytic and endophytic, according to the site of attachment to the orbital rims. This explains their different presentation. The exophytic cysts are discovered earlier in childhood, whereas the endophytic ones are discovered later in life and are associated with bone damage and invasion of orbital space. Many authors discuss the dermoid cysts without regard to their location, anterior or posterior, relative to orbital septum, lumping them together as orbital in origin. The cyst anterior to the orbital septum should be correctly classified as periorbital, extraorbital, or adnexal in location. Also, these cysts are sometimes referred to incorrectly as “dermoids.” This designation is inappropriate because of the confusion with the solid dermoid (dermolipoma) on the surface of the eye. The older term cholesteatoma should not be used in place of the correct name, dermoid cyst.
Incidence
Forty-one dermoid cysts are tabulated in Table 3.3, an incidence of 2.3% (41/1,795) of total tumors. These 41 cysts include both the histologic types and exclude cysts located anterior to the orbital septum. Many authors who write on the subject of dermoid cysts combine preseptal and postseptal cysts as orbital in location. If we had included the preseptal cysts that we encountered surgically since the beginning of our survey (1948), our present total would be approximately 70 cysts.
The popular, anatomically broad (both preseptal and postseptal types) concept is the basis for an oft-repeated statement that the dermoid cyst is the most common orbital tumor of childhood. In our strictly orbital categorization, dermoid cyst was fifth in frequency in the list of childhood (first decade) tumors (Table 3.4). In the second decade, dermoid cyst was second in frequency. Two thirds of all dermoid cysts in our survey occurred in the first two decades of life. The cysts located in the retrobulbar space occurred more often in the later decades and were usually of the epidermoid types.
Our oldest patient was a 75-year-old woman who presented with a history of “fullness” in the right temporal fossa (see Fig. 4.1) and protrusion of the eye, of 4 years’ duration. The eye had a 3-mm proptosis with inward and downward displacement of 2 mm. The cyst was intradiploic in the orbital portion of the sphenoid bone, causing expansion of bone in both the orbital space and temporal fossa. The sex incidence of our series was 19 men and 22 women. All cysts were unilateral.
The largest series of orbital dermoid cysts published since our last edition is that of Shields et al. (1997). They compiled 195 consecutive, histopathologically proved cases over a 32-year period. They included ten cases of conjunctival cysts and an unspecified number of periorbital cysts. Their incidence data would be more valid had they excluded these conjunctival and periorbital cysts. The value of their article is the anatomic classification they proposed for the management of cysts lined by keratinizing stratified squamous epithelium and containing adnexal elements of hair, sebaceous glands, and sweat glands.
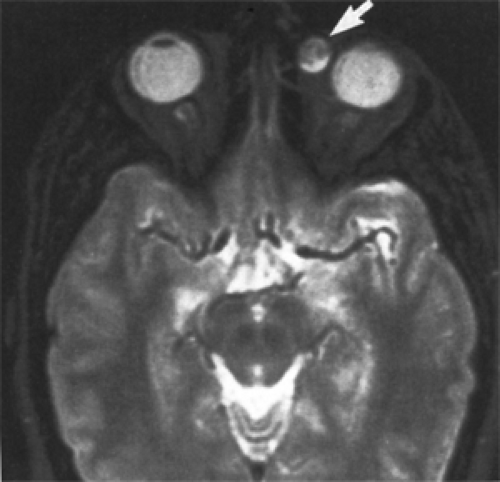
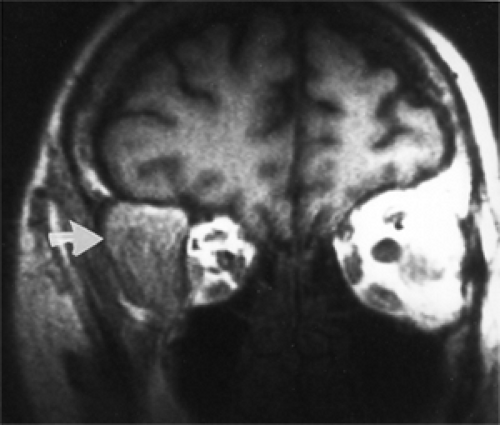 Figure 4.1 Magnetic resonance imaging showing large extension of a cyst into the right temporal fossa (arrow). |
Bonavolonta et al. (1995) reported 145 patients observed between 1976 and 1992. They carefully classified their cases, in relation to the orbital septum, as exophytic (76 patients) and endophytic (69 patients). The median age was 7 years, and the oldest patient was 53 years.
Clinical Features
We follow the current trend of addressing the clinical aspects of these cysts according to their clinical and anatomic location—superficial, palpable, and nonpalpable deep cysts.
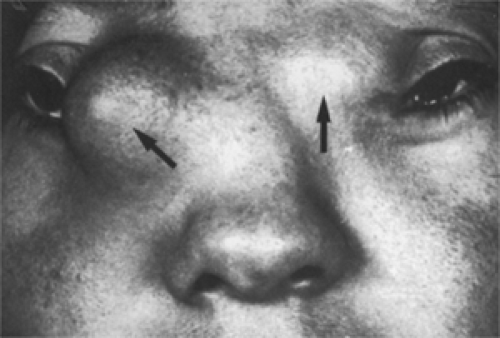
The palpable type (see Fig. 4.2) is a smooth, firm, nontender, oval mass usually located along the superior orbital rim and attached by a pedicle of fibrous tissue to the zygomatic frontal synostosis or maxillofrontal synostosis. In this location, the mass is easily palpable and can be rolled between the thumb and forefinger. The mass is not attached to the skin. Seldom is it larger than 2 cm in length. The tumor is usually diagnosed before the patient reaches 5 years of age.
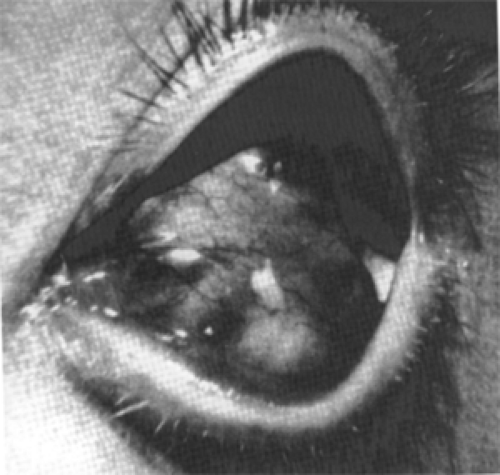
The other superficial cyst (see Fig. 4.3), postseptal in location, requires a longer period of growth before breaching the septum and becoming palpable in the subcutaneous space of the eyelid. This cyst usually pops through a hiatus that develops between the orbital septum and its attachment to the bone, which may not occur until adolescence. In two of our patients, such a cyst was located in the lower orbit, one in the medial quadrant and the other in the lateral quadrant. Proptosis or displacement of the eye, bone
remodeling, (Sathananthan et al., 1993) and diplopia are seldom features of these superficial cysts.
remodeling, (Sathananthan et al., 1993) and diplopia are seldom features of these superficial cysts.
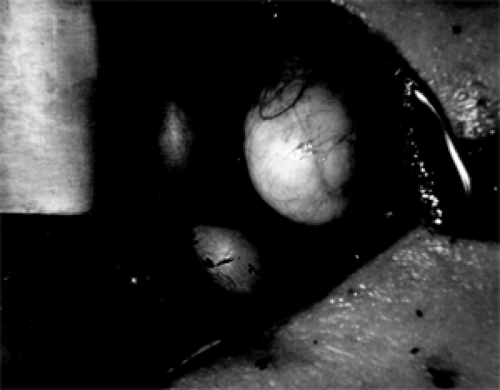 Figure 4.3 An in situ superficial, juxtasutural, endophytic dermoid cyst (3 × 2 × 1 cm) attached to the superolateral wall of the left orbit in a 17-year-old man. Two metal retractors are positioned to the right of the mass. (See Color image.) |
Clinical features of deep (retrobulbar and peribulbar) cysts are another story. Their behavior is contrary to the predictable behavior of superficial cysts. The origin of the deep cyst is either some unseen or unknown embryonic rest attached to a soft tissue structure, usually muscles in the orbital space, or, more often, the diploic space of the frontal, sphenoid, or zygomatic bone. If the cyst selects the frontal or sphenoid bone, a round hiatus eventually develops in the outer table of the affected bone, allowing access to anterior or middle cranial fossa. Less often, the cyst expands into the temporal fossa.
The “giant,” “dumbbell,” and “hourglass” dermoid cysts are usually intradiploic in origin. These cysts have expanding orbital and extraorbital components. If the extraorbital component presents in the infratemporal fossa, the pressure of the temporalis muscle on the cyst may induce proptosis of the eye during mastication (Whitney et al., 1986), “chewing oscillopsia” (Knight et al., 1984), and “bobbing” of the eye (Rootman and Lapointe, 1988). Emerick et al. (1997) reported chewing-induced transient visual impairment in a 29-year-old woman.
An intradiploic cyst rarely presents as a discharging sinus tract. Honig (1998) reported such a case involving the frontozygomatic synostosis. More likely, a chronic draining sinus was associated with recurrent cysts that resisted complete excision.
The origin of a long-standing deep cyst in the medial confines of the orbit is usually obscure. It probably arises from an epithelial nidus attached to some soft tissue structure. This cyst can easily penetrate the thin ethmoid plate, but we are not aware of any report where it blossomed into an ethmoid sinus. Instead, it may expand superiorly, remodel the frontal bone, and ultimately evacuate dirty tan–colored contents with a slightly musky odor through a sinus tract opening under the superior nasal rim near the trochlea. In the years before present imaging technology, the draining sinus was easily mistaken for a tract from a mucocele of the frontal sinus. These cysts may reach a considerable size because of their insidious expansion, with proptosis of 5 to 8 mm. Diplopia and impairment of extraocular muscle motility are common accompaniments, but severe reduction of visual acuity is uncommon.
Imaging Aspects
While discussing this subject, it is important to keep in mind the multiple tissue variables of a dermoid cyst, because relative homogeneity or heterogeneity of the mass influences the ultimate imaging display. The cyst may include all or a few of the following: Keratinizing squamous epithelium, fat, cholesterol clefts, old hemorrhage, hairs, keratin, sebaceous gland, macrophages, lipid globules, multinucleated giant cells, chronic inflammatory cells, and calcium.
MRI of the small superficial cyst located along the orbital rim is rather consistent. Usually, the cyst is a smooth, oval, well-outlined mass with a homogeneous center of low attenuation (see Fig. 4.4). The rim of the mass may show enhancement with contrast injection, but the cyst’s contents show no enhancement. A slight faceting or pressure thinning of adjacent orbital rim is present in some cases.
In the posterior orbit, the cyst is larger and associated with bone erosion that allows many cysts to egress into
the intracranial vault or temporal fossa. The contents show a heterogeneous signal, reflecting different densities of fat, fluid, keratin, and epithelial debris.
the intracranial vault or temporal fossa. The contents show a heterogeneous signal, reflecting different densities of fat, fluid, keratin, and epithelial debris.
Chawda and Moseley (1999) reviewed CT scan images of 160 patients with histologically proved orbital dermoid cysts over a 20-year period. Eighty-five percent (136/160) had adjacent bone changes, 46% (73/160) showed lower signal attenuation than water in the vitreous body, 14% (22/160) had calcification, and 5% (8/160) had fluid levels. They concluded that the range of appearance was wide.
MRI is also useful because of the high signal intensity of fat that provides excellent contrast between the cyst and its environs (see Fig. 4.5A). In large posterior-positioned cysts, the signal may show differing intensities dependent on the content of the tumor. This may vary from one cyst to another. Some tumors emit a T1/T2 signal similar to vitreous or only the T2-weighted signal-weighted signal that shows hyperintensity. The tumor may also be hyperintense on T1-weighted images (Newton and Bilaniuk, 1990). Finally, the image may indicate a fluid level within the cyst that shifts when the patient changes from a supine to decubitus position (Fig. 4.5B).
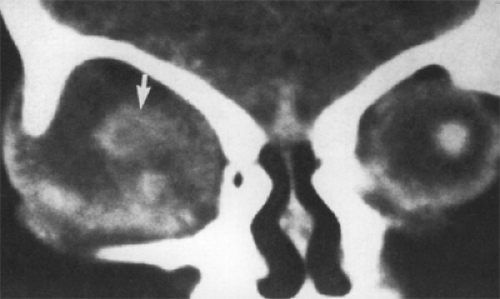
Plain film radiography is seldom used now for diagnosis of an orbital mass unless the tumor invades the bone. Such is the case with a large dermoid cyst in the posterior orbit that erodes the bone and enters the intracranial vault. To a lesser extent, this applies to the intradiploic cyst in the lateral portion of the sphenoid bone that reaches the intracranial vault through the marrow space. Both of these situations require surgical intervention. The size and location of the bony aperture are well outlined in a standard Caldwell or Water projection (see Fig. 4.6). The diagnostic feature, almost unique to the dermoid cyst, is the polished appearance of the bone rim surrounding the protruding cyst wall. A skull film is superior to CT scan in delineating this feature. This sclerosis is the result of long-time, persistent pressure from the expanding mass. Bone sclerosis of such a degree is not seen in other tumors that enter or exit through an unwanted hole in the bone partition between the orbital space and the intracranial vault, such as encephaloceles, meningoceles, various cranial heterotopias, plasmacytoma, hematoceles, Langerhans
histiocytosis, mucoceles of the frontal sinus, malignant neoplasms, and aneurysmal bone cyst.
histiocytosis, mucoceles of the frontal sinus, malignant neoplasms, and aneurysmal bone cyst.
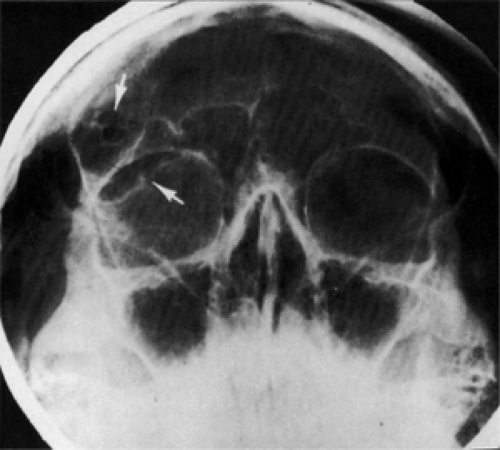 Figure 4.6 Large epidermoid cyst eroding the roof of the right orbit (lower arrow) with a small hiatus (upper arrow) through the inner table of bone into the intracranial vault (Water view). |
Ultrasonography is a practical method for office diagnoses of a superficial dermoid cyst in a small child.
Pathology
Histopathologically, this cyst is divided into dermoid and epidermoid types. In the initial pages of this chapter, we stated our intent based on diagnosis, clinical features, and management, to regard it as one tumor. Both types have a lining of stratified squamous epithelium, but the epidermoid type does not have epidermoid appendages in its wall, which are characteristic of the dermoid type. Both types contain desquamated keratin.
This thick-walled dermoid type contains hair follicles, hair, sebaceous glands, and sweat glands (see Fig. 4.7). The smaller dermoid type has a whitish yellow color and may have shallow fat pads attached to its surface. Anatomically, it is usually located in the anterior orbit.
The larger, thin-walled epidermoid type is filled with a cheesy-textured material (see Fig. 4.8), flecks of calcium, and cholesterol clefts. The walls of these cysts have a lipogranulomatous reaction featuring macrophages, multinucleated giant cells, and hemosiderin deposition (see Fig. 4.9A and B).
In our third edition, two reports are referenced in which squamous cell carcinoma was found in an epidermoid cyst. Both patients were 53 years old. Since then, Holds et al. (1993) reported two patients in whom the lining of their epidermoid cysts underwent malignant transformation into invasive squamous cell carcinomas. Each patient underwent orbital exenteration combined with craniofacial resection.
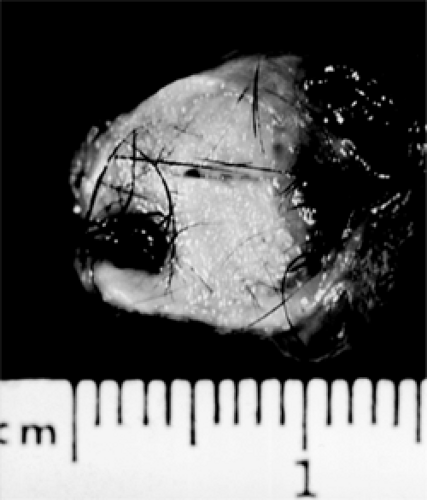 Figure 4.7 Dermoid cyst (opened) showing exuberant growth of hair. (See Color image.) |
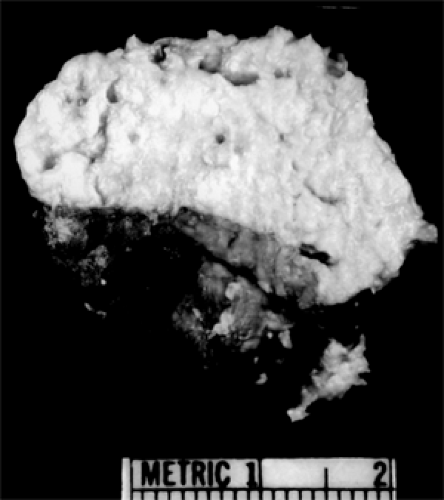 Figure 4.8 Epidermoid cyst (opened) showing copious cheesy material. (See Color image.) |
Treatment
We limit this discussion to the surgical management of cysts posterior to the orbital septum. This excludes superficial cysts located in the adnexal tissues of the upper eyelid. This cyst is usually juxtasutural, seldom larger than 5 mm, and usually removed before school-going age. It can be removed intact through an eyelid crease incision.
Our favorite approach to the common cyst attached to, or adjacent to, the zygomatic frontal suture is the superotemporal, anterior infrabrow incision. The incision can be made directly over the cyst, and the aim of further dissection is intact removal of the mass. Next, the orbicularis muscle and fascia of the eyelid are incised and retracted. At this point, the anterior face of the cyst may be seen through a hiatus in the orbital septum that has been eroded by the expanding tumor. The hiatal opening is enlarged superiorly and inferiorly to accommodate delivery of the cyst.
These cysts are large, probably have been present since birth, but are not accessible for surgical removal until the second decade of life. Figure 4.10 illustrates the surgical exposure necessary to remove a sizable dermoid cyst just before severing its attachment to the bone. To prevent
undue rupture of the cyst, a tag of periorbita is left attached to the bone at the time of excision.
undue rupture of the cyst, a tag of periorbita is left attached to the bone at the time of excision.
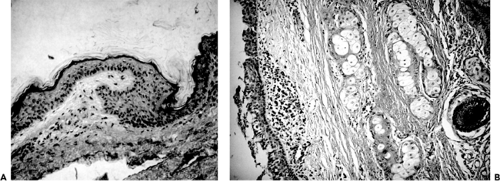 Figure 4.9 A: Section of epidermoid cyst. The lumen is shown at the top. The lining (arrow) is keratinizing squamous epithelium. Thin strands of keratin are shedding into the lumen from the epithelial surface (hematoxylin and eosin, original magnification × 160). B: This section of dermoid cyst wall shows squamous epithelial lining, hair shaft, sebaceous glands, and focal collections of chronic inflammatory cells (hematoxylin and eosin, original magnification × 70). (See Color image.) |
In many cases, because of the size and long duration of growth, the cyst is adherent to the posterior surface of the orbital septum in addition to the adjacent soft tissue components of the anterior orbit. These cysts usually have a thick wall that permits freeing of adhesions to surrounding soft tissues once the mass is detached from the bone. Detaching the mass from the bone is accomplished by grasping the pedicle of the tumor and adjacent periorbita with a small forceps, excising the tumor from the bone, and bringing the specimen forward through the incision in the orbital septum. Thus mobilized, the back and sides of the tumor can be freed of adhesions, and with use of cotton-tipped applicators augmented by a cryoprobe as a traction lever, the mass is peeled away from the orbital septum. Whatever the method of dissection, there is usually immediate, profuse bleeding when the tumor’s vascular pedicle is severed. Bleeding can be controlled by wet-field bipolar cautery. If, on inspection of the surgical field, an embryonic rest of epithelium remains within the bone, it can be reamed out by a small burr attached to a bone drill. A remnant of epithelium in a niche of bone is easily overlooked and may be a nidus for recurrence of the cyst. Recurrent cysts should be imaged radiographically or by CT scan to rule out this possibility.
After suturing the orbital septum to either the periorbita or periosteum, the soft tissue and skin are closed in layers. With present-day sutures, there is little worry about healing with a scar.
Occasionally, aberrant cysts occur somewhere along the inferior or lateral rim of the orbit just beneath the attachment of the lateral palpebral raphe. They are small and present at birth or soon after. A lump or a surface of the mass is frequently visible beneath the conjunctival fornix. A transconjunctival fornix incision is appropriate for removal of this cyst, and intact removal is the rule. The incision should be sutured to prevent symblepharon.
Before the use of CT scan and MRI, surgical removal of an epidermoid-type cyst from the retrobulbar space, without bone involvement, was a frustrating endeavor. The patient’s symptoms suggested that the suspected cyst had been present for many months or several years and was probably quite large. The size of the cyst could be estimated by the amount of proptosis—the greater the numerical value of proptosis, the larger the cyst. The position of the cyst could be guessed by the direction of displacement of
the eye. The size and position of the cyst are now part of the imaging mode display. The surgeon can select the best place to enter the orbit for direct access to the tumor. Once the appropriate approach has been made and the surface of the mass is visible, there remains the frustration of how to remove the tumor intact.
the eye. The size and position of the cyst are now part of the imaging mode display. The surgeon can select the best place to enter the orbit for direct access to the tumor. Once the appropriate approach has been made and the surface of the mass is visible, there remains the frustration of how to remove the tumor intact.
If the cyst has not become large enough or has not been present long enough to have fused with surrounding soft tissue, a cleavage plane is probably present. The surgeon may commence dissection using cotton-tipped applicators and blunt scissors or other dissecting tools. Dissection of tumor from surrounding soft tissue is similar to removal of a cavernous hemangioma. However, application of the cryoprobe to the surface of the cyst for its traction and leverage may not be a useful adjunct because the thin wall of the cyst may rupture, defeating the goal of intact removal.
Many large cysts of long duration in the retrobulbar space fuse to the surrounding soft tissue because of relentless pressure incurred in expansion. The wall of such a cyst is almost paper-thin and has a bluish color in contrast to the flesh color of a thick-walled dermoid-type cyst in the anterior orbit. Also, the cyst is quite firm and filled with cheesy-textured material and semifluid debris almost to the point of rupture. Removing such a cyst intact is impossible.
Instead, we prefer to open the mass widely and decompress it by sucking out the contents. Once the cyst is collapsed, the surgeon can see the important structures in the orbit—muscles, large blood vessels, and nerves—to which the cyst wall is attached. The periphery of the tumor likely is adherent to the dura of the optic nerve. Bits and pieces of the cyst wall can be nibbled away from the muscles, large peripheral nerves, and blood vessels with scissors. However, this dissection should not be attempted along the optic nerve because it inevitably results in some visual loss. It is best to observe the maxim, “The less manipulation of the optic nerve, the better the visual outcome.” At this point, the surgeon wraps several layers of gauze around his forefinger and scoops out, swabs, and scours the remaining lining epithelium. The surgeon may or may not wish to insert a removable drain into the orbital cavity at the time of closure.
Epidermoid cysts that have extension into the intracranial vault through erosion of the frontal or sphenoid bone and dumbbell cysts arising from an embryonic epithelial rest within the synostosis of the sphenoid and zygomatic bones should be approached through a coronal orbitocraniotomy for maximum exposure. This approach allows evacuation of the orbital portion of the cyst, relieving the possibility of any complications caused by penetration of the cyst into the intracranial space. Likewise, along the lateral orbital wall, both orbital and temporal fossa lobes of a dumbbell cyst are visible for removal. Bits of bone surrounding the bony aperture housing the sutural embryonic epithelial nidus can be easily removed, and the marrow passages of the sphenoid can be inspected for any epithelial extension.
Technically, these large epidermoid cysts are only partially or incompletely removed by all these surgical maneuvers, and the patient is subject to recurrence. However, such an excision is not invariably a factor in recurrence. If all the epithelial lining is peeled away, the cyst does not recur.
Conjunctival Cysts
Another congenital cyst closely related to the preceding dermoid cyst, but very rare, is the conjunctival cyst. In the older literature, it is called a simple cyst. Jakobiec et al. (1978) performed a retrospective study of all cystic adnexal lesions that had been classified as epidermoid, dermoids, or cholesteatomas on file in the ophthalmic pathology library at Columbia University’s College of Physicians and Surgeons, accessioned from 1929 to 1977. A total of 128 specimens were reviewed.
Among them were seven cysts lined by stratified, nonkeratinized epithelium, and their walls contained adnexal structures. These cysts, therefore, were neither epidermoid nor dermoid. The name conjunctival dermoid was assigned to this group. Another five had a similar epithelium without adnexal structures in their walls. To this group, Jakobiec et al. (1978) assigned the designation conjunctival cyst. We believe this is the congenital cyst in our classification.
Since 1978, conjunctival dermoid cysts have been reported nearly three times as often as simple conjunctival cysts. The former are a hybrid lot; their pathogenesis is speculative. The congenital conjunctival cyst has a lining of stratified, nonkeratinizing cuboidal epithelium intermixed with mucous-producing goblet cells and containing either a clear or sticky serous fluid. The cyst is thin-walled, translucent, and soft, does not alter the orbital bone, and is usually not evident at birth. The cyst remains hidden in the deeper portions of the superior conjunctival fornix until it enlarges and presents as a puffiness of the adjacent upper eyelid or a palpable mass of indefinite contour. This does not occur until later in infancy, early childhood, or adolescence. If it is possible to evert the patient’s upper eyelid, the cyst comes into view.
Since 1988, several cases of this type have been reported—one case by Tsai et al. (1996), two of four cases by West et al. (1997), two cases among 197 orbital cyst specimens by Shields et al. (1997), and one case by Boynton et al. (1992).
With the present orbital-imaging techniques, cases are now identified, which are attached to the superior oblique tendon (Boynton et al., 1992) and to the common sheath of superior rectus and levator palpebrae superioris muscles (Can et al., 1999; Rose and O’Donnell, 1995). These three references seem to be the first to authenticate the origin of the simple cysts from peribulbar extraocular muscles in the superior anterior orbit. We believe that, in the years
before the CT scan and MRI, the origin of these cysts was unknown.
before the CT scan and MRI, the origin of these cysts was unknown.
In the report by Rose and O’Donnell (1995), the two female patients, aged 14 months and 23 years, and the two male patients, aged 3½ and 39 years, all with unilateral cysts, are of particular interest. A bluish purple mass was visible in the upper conjunctival fornix and palpable through the upper eyelid. All patients had blepharoptosis but normal levator muscle function. The 23-year-old patient had ptosis since birth. The duration of signs and symptoms in the other three patients was shorter.
The authors approached the tumors through a transconjunctival incision along the upper border of the superior tarsal plate. With careful microdissection, the cyst was mobilized superiorly and posteriorly through the orbital septum to its terminus sandwiched between the levator palpebrae superioris and superior rectus muscles, within the sheath encompassing both muscles. In all four patients, the tumor was removed intact.
There is less information about the 37-year-old woman reported by Can et al. (1999). A transconjunctival approach was used, and the approximate dimensions of the cyst were 25 × 19 × 13 mm. The approximate measurement suggests that the tumor might have been decompressed or ruptured during removal. Acquired conjunctival cysts are a frequent postoperative complication of present-day scleral buckling procedures.
Congenital Cystic Eyeball
The present name of this developmental cyst was suggested by Ida Mann (1937) in her text. It is the same tumor described by earlier authors as an anophthalmos with a cyst. It is quite rare. Hayashi et al. (1999) found only 14 histopathologically proved cases, including their two cases, published in the English language literature since 1964. The stage for this tumor is set at about the fourth week of embryonic life, when the primitive optic vesicle should invaginate and form the secondary optic vesicle. Such development is aborted at this point and results in a slowly enlarging primary optic vesicle. Histopathology reflects the primitive nature of the tumor. It is composed of a dysplastic array of primitive neuroectodermal elements, immature mesenchyme, connective tissue, glial and neuroglial tissue, squamous and glandular epithelium, cartilage, bone, and calcium. The cyst may be unilocular or multilocular. A tiny white or yellow dot-like structure corresponding to the optic nerve head may be found along some of the mass. The fluid content is usually clear yellow; a more viscous purple fluid probably represents prior hemorrhage.
The sex incidence is equal. The cystic eye usually affects one orbit of an infant, with a normal eye in the other orbit. Cases of this type reported in the literature since the subject was reviewed in our third edition include those published by Gupta et al. (1990) and Mansour and Li (1996). Hayashi et al. (1999) also observed two infants, a 13-month-old boy and a 2-week-old girl, who were considered to be anophthalmic and developed a cystic lesion in the left orbit with protrusion of lower eyelid. Neonates with cystic eyes are also subject to myriad other congenital anomalies (Wilson et al., 1985; Pasquale et al., 1991).
The cystic eyeball differs from teratoma in the same age-group in its smaller size, it does not have a formed eye, and it is partially covered by an upper eyelid unless the elasticity of the eyelid is overcome by pressure of the expanding mass.
Both CT scan and MRI show a cystic mass containing an admixture of solid components and fluid. The orbit may be enlarged and an optic nerve track visible (Fig. 4.10).
The common management of a cystic eyeball is surgical removal. The conjunctiva is incised and dissected from the underlying cyst, creating flaps. Mansour and Li (1996) removed a 3 × 3.5-mm cyst intact in spite of adhesion of the cyst wall to periorbita. If the wall of a large cyst seems too thin to permit dissection, the fluid content can be aspirated and the cyst extirpated or exenterated by severing the optic nerve with a snare. Conjunctival flaps are used to cover the exposed bone. A noteworthy case reported by Awan (1986) was a 29-year-old woman with a translucent, multiloculated cyst in the left orbit, first noted at birth. After an initial period of slow enlargement in early childhood, the mass became stationary (see Fig. 4.11). No surgical excision was necessary.
Microphthalmos with Cyst
This anomaly of the embryonic eye occurs at a stage immediately after a mishap during fetal development that results in the preceding tumor, the congenital cystic eyeball. The basic defect is improper fusion of margins of the fetal fissure, in essence, a coloboma. If prolapse of primitive ocular tissue into the imperfect cleft occurs soon after the fourth week of embryonic life, the 8-mm stage, the resulting cyst is present at birth. However, if closure of the fetal fissure is complete but defective, ectasia of thin sclera with a cyst-like expansion into the orbit may not occur until some time after birth. A noteworthy case of this type was a 27-year-old man reported by Nowinski et al. (1984). The patient was born with an enophthalmic eye and had worn a cosmetic shell for approximately 20 years. Then, progressive proptosis occurred owing to delayed enlargement of the cystic component.
The end result is a defective small eye to which is attached a herniated cyst-like mass. The cyst may be a small appendage of the larger eye or a large mass that greatly exceeds the size of the attached globe. In the latter, the microphthalmic eye may not be evident at the time of presentation because it is concealed by the oversized cyst. Size may vary between these large and small extremes. If the bilobed structure (eye and cyst) is small, an enophthalmic, strabismic eye with a cystic appendage is the presenting feature. When the cyst becomes larger than the eye, a soft, vascular pink mass occupying the palpebral fissure may be the presenting feature. Finally, a larger bluish, balloon-like mass may push out of the orbit, partially covered by a stretched lower eyelid (see Fig. 4.12). It is these larger cysts that require differential diagnosis from other orbital tumors in the neonatal period.
The incidence of microphthalmos with cyst is greater than that of either teratoma or congenital cystic eyeball but less than that of encephalocele. The laterality of the tumor and its sex incidence are equal. Most of the cases in the literature are unilateral, including two cases in our total series. Since our review of this subject in the third edition, single cases have been recorded by Guterman et al. (1990) and Kodama et al. (1998). Bilateral cases are not common. Mutations in the PAX6 homeobox gene and chromosomal deletion in the 14q, g22g23 region of the genome may be responsible for the congenital defect (Bennett et al., 1991; Bardakjian et al., 1997).
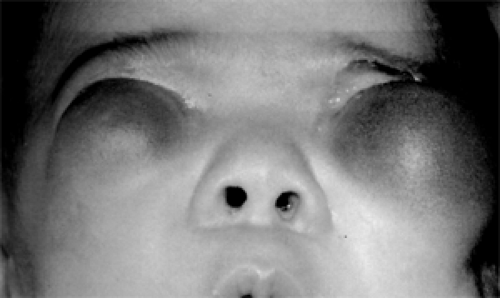 Figure 4.12 Microphthalmos with cyst in a 3-month-old female infant with bilateral orbital cysts that were attached to microphthalmic globes. The cysts were surgically verified. (Courtesy of Cameron JD, Rochester, Minnesota.). (See Color image.) |
Pathologically, the eye is markedly small and displaced superiorly by the inferior nasal colobomatous cyst. The eye may contain immature or dysplastic evidence of cornea, iris, ciliary body, lens, vitreous, choroid, and retina. The colobomatous defect allows protrusion of the cystic structure that is lined by highly vascularized glial tissue mixed with some dysplastic retinal tissue. This inner lining in the case described by Lieb et al. (1990) stained diffusely with glial fibrillary acidic protein. Deep to the glial tissue along the inner wall, some staining for neurofilaments was also noted. Attached to the outer scleral wall posteriorly, some skeletal muscle fibers may be identified.
CT scan outlines the position of the abnormal eye and the size of the cyst. If the cyst is very large, it encompasses the small eye and hides it from view. If the cystic structure is located superiorly, it is probably a congenital cystic eyeball, not a microphthalmic cystic eyeball. If the CT scan display shows specks of calcium, the lesion is probably a cystic teratoma. The colobomatous cyst appears hypotense on T1-weighted MRI and hyperintense on T2-weighted MRI (Kaufman et al., 1998).
In almost all cases, the microphthalmic eye and its appendage must be removed for cosmetic reasons because of continued expansion of the cystic component. After removal of the tumor, some hemifacial deformity may occur due to lack of stimulus for orbital bone growth. To ameliorate this, Gossman et al. (1999) used an orbital tissue expander in five children aged 10 months to 6 years. Gradual inflation of the expander to a diameter of 22 mm reduced the average preoperative orbital dimension deficit of the group from 14.6% to 3.8% after surgery. The average expansion period was 56.8 weeks (range, 20–100 weeks).
Cephaloceles
Cephalocele is a broad term that encompasses herniations into the orbit of the parenchymal brain tissue (encephalocele), membranous brain cover (meningocele), or a mixture of brain and meninges (meningoencephalocele). Technically, the cephaloceles retain some attachment to the brain by a cord or stalk of tissue. Usually, they are associated with a defect of orbital bone at the junction of the frontal and ethmoid bones, or at the lesser and greater wings of the sphenoid bone, or at the skull base.
Incidence
These tumors are not common but are more frequent than teratomas. They are mostly found in the young and
often are visible at birth. Their occurrence in adults is a curiosity. Mahapatra et al. (1994) reported 30 cases of anterior encephaloceles treated between 1973 and 1990. At the time of surgery, >60% of the patients were <2 years old. Only one child was older than 10 years. Twenty-six patients had frontoethmoidal defects. Later, Mahapatra (1997) retrospectively studied 65 children with anterior encephaloceles spanning a 22-year period (1973–1994). Six children were older than 10 years. Nasoethmoid type was the most frequent, encountered in 45 patients. In our 50-year survey, we observed only four cases, three females and one male. Three of these patients were <1 year of age. The fourth patient was 11 years old at the time of presentation.
often are visible at birth. Their occurrence in adults is a curiosity. Mahapatra et al. (1994) reported 30 cases of anterior encephaloceles treated between 1973 and 1990. At the time of surgery, >60% of the patients were <2 years old. Only one child was older than 10 years. Twenty-six patients had frontoethmoidal defects. Later, Mahapatra (1997) retrospectively studied 65 children with anterior encephaloceles spanning a 22-year period (1973–1994). Six children were older than 10 years. Nasoethmoid type was the most frequent, encountered in 45 patients. In our 50-year survey, we observed only four cases, three females and one male. Three of these patients were <1 year of age. The fourth patient was 11 years old at the time of presentation.
Clinical Features
These lesions have been classified according to anatomic location, structural configuration, and presence or absence of external manifestations. Anatomically, in reference to the orbit, cephaloceles are either anterior or posterior. The anterior tumor is usually visible, but the posterior lesion may remain hidden for some months or years. Structurally, the herniations are either sincipital or basal in origin. The sincipital mass is almost always visible. The basal tumor may or may not be manifest in early life, depending on the size and location of the osseous dehiscence.
The anterior sincipital variant is the most common and is usually evident at birth as a paranasal mass located at the nasofrontal–orbital junction (Koopmann and Reynolds, 1981) (see Fig. 4.13). The mass in this illustration is smaller than most of the anterior cephaloceles depicted in the literature. The size of the mass determines the degree of displacement of the eye. The mass is painless and feels less firm than other developmental cysts because the content of soft neural tissue within the mass usually exceeds that of fluid. The mass slowly enlarges during a period of months or years because of an intrinsic cystic degeneration.
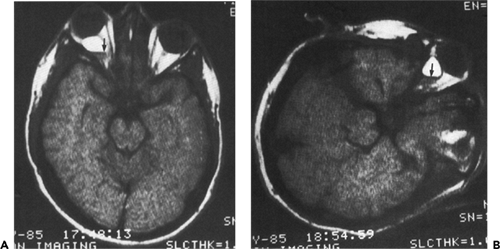
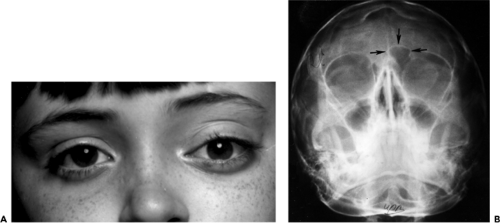
If the dehiscence in bone is located posteriorly along the frontoethmoid suture, the herniation of tissue pushes the eye laterally. In this case, the paranasal mass may not become apparent until some months later. The cephalocele was of this type in the 11-year-old girl in our series of four patients. She presented with a protrusion of the left eye of 8 months’ duration (see Fig. 4.14A). The left eye had a 4 mm proptosis, with 3-mm downward displacement and 7-mm lateral displacement. A bony mass was palpable in the superonasal orbit. CT scan imaging revealed an enlargement of the orbit and an intraorbital mass extending into the anterior cranial fossa (Fig. 4.14B). Anterior orbitotomy uncovered a bluish cyst-like mass covered by a thin shell of bone. The bone was removed, the cyst contents were evacuated, and the membrane-like wall of the cyst was stripped from the exposed dura. Bleeding was marked and difficult to control. Others who approach these tumors surgically have also commented on this bleeding tendency.
The spheno–orbital encephaloceles are oriented to the posterior orbit. Herniation occurs either through the superior orbital fissure, a defect in the bone along the lesser wing of the sphenoid bone, or an aplasia of the greater wing of the sphenoid bone. Most of these are caused by the mesodermal dysplasia associated with neurofibromatosis type 1 (Sugawara et al., 1996; Clauser et al., 1998; de Vries et al., 1998). These tumors, secondary to aplasia of the greater wing of the sphenoid bone, are large and result in a pulsating exophthalmos. A meningoencephalocele arising in the anterior skull base associated with neurofibromatosis and extending through an enlarged superior orbital fissure should also be included in this group (Chapman et al., 2000).
Several cephaloceles with unusual presentations have been recorded (see Fig. 4.15). Soyer et al. (1990) reported a case presenting with trigeminal neuralgia caused by a herniation through a congenital defect in the greater wing of the sphenoid bone associated with angioma of the soft palate. Terry et al. (1993) observed a neonate presenting with a soft mass in the superomedial conjunctival fornix that on surgical exploration was an ependymal cyst filled with cerebrospinal fluid and containing neuroglial and meningeal tissue. Gunduz and Gunalp (1997) reported an 18-year-old man with right congenital symblepharon (abortive cryptophthalmos), cleft palate, visual acuity of no light perception, and an absent right orbital roof. Bilateral cases associated with midline craniofacial abnormalities have been reported by Levy et al. (1989), Cataltepe and Ozcan (1990), and Hershewe et al. (1995). A white, excavated distorted optic disk is often associated with basal encephalocele, the “morning glory” disk (Goldhammer and Smith, 1975).
Imaging Aspects
Unless the bony dehiscence is tiny, CT scan images the defect and outlines the homogeneous appearance and cystic extension of the mass into the orbit. The solid content of the mass is isodense with the brain. MRI also shows features similar to that of the brain. The appearance of the cephalocele differs from that of dermoid cyst and teratoma, which have more fluid content.
Pathology
The basic element of these tumors is glial tissue with supporting connective tissue trabeculae. Occasionally, neurons, nerve fibers, and an ependymal lumen may be present. If the cyst is large, there may be a communication with the ventricular system (see Fig. 4.16). When a cephalocele is of long standing, as in adults, the meningeal layers may fuse and fibrose. The neural tissue also becomes edematous and degenerate, and calcium deposits may appear.
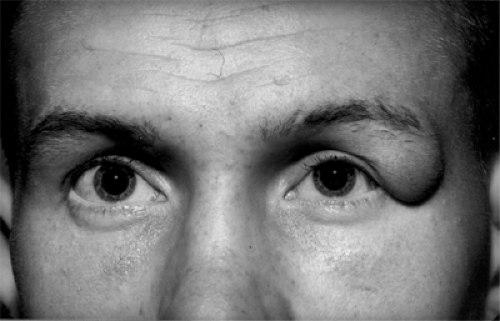
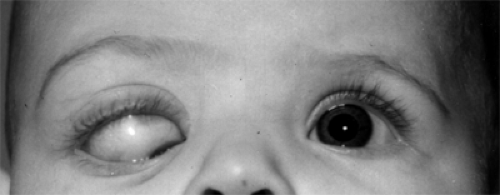 Figure 4.15 Downward and inward displacement of the right eye since birth in a 5-month-old female infant. The displacement was caused by a posterosuperotemporal meningoencephalocele. The cornea is almost covered by her lower eyelid.
Stay updated, free articles. Join our Telegram channel
Full access? Get Clinical Tree
 Get Clinical Tree app for offline access
Get Clinical Tree app for offline access

|