Purpose
To review the diagnostic categories of a group of conditions referred to as “primary acquired melanosis.”
Design
Literature review on the subject and proposal of an alternative diagnostic schema with histopathologic and immunohistochemical illustrations.
Methods
Standard hematoxylin-eosin–stained sections and immunohistochemical stains for MART-1, HMB-45, microphthalmia-associated transcription factor (MiTF), and Ki-67 for calculating the proliferation index are illustrated.
Results
“Melanosis” is an inadequate and misleading term because it does not distinguish between conjunctival intraepithelial melanin overproduction (“hyperpigmentation”) and intraepithelial melanocytic proliferation. It is recommended that “intraepithelial melanocytic proliferation” be adopted for histopathologic diagnosis. Atypical proliferations are characterized either by bloated dendritic melanocytes with enlarged cell components (dendrites, cell bodies, and nuclei) or by epithelioid melanocytes without dendrites. Atypical polygonal or epithelioid pagetoid cells may reach higher levels of the epithelium beyond the basal layer. Immunohistochemistry defines the degree of melanocytic proliferation or the cellular shape (dendritic or nondendritic) (MART-1, HMB-45) or identifies the melanocytic nuclei (MiTF). Intraepithelial melanocytic proliferation without atypia represents increased numbers of normal-appearing dendritic melanocytes (hyperplasia or early neoplasia) that generally remain confined to the basal/basement membrane region. Intraepithelial nonproliferative melanocytic pigmentation signifies the usually small number of conjunctival basal dendritic melanocytes that synthesize increased amounts of melanin that is transferred to surrounding keratinocytes.
Conclusion
All pre- and postoperative biopsies of flat conjunctival melanocytic disorders should be evaluated immunohistochemically if there is any question regarding atypicality. This should lead to a clearer microscopic descriptive diagnosis that is predicated on an analysis of the participating cell types and their architectural patterns. This approach is conducive to a better appreciation of features indicating when to intervene therapeutically. An accurate early diagnosis should forestall unnecessary later surgery.
Conjunctival melanomas are seen in 2–8 individuals per million in predominantly white populations and constitute only 2% of ocular melanomas, the remainder developing in the uveal tract. There is some evidence that ultraviolet radiation plays a role in causing many conjunctival melanocytic lesions, which develop most often in the interpalpebral zone. Around 25% of conjunctival melanomas arise in pre-existing nevi (which appears not to influence prognosis) and 70%–75% in pre-existing “primary acquired melanosis,” which is somewhat more common than melanomas. Primary acquired melanosis (PAM) is one of the few diseases in ophthalmology that is on a trajectory capable of culminating in fatality (13%–30% lethality for melanomas at 10 years). An important exercise in ophthalmic pathology, therefore, is the prediction of which patients with PAM are at high risk to develop melanoma, a progressively metastasizing lesion once the invasive nodule exceeds a thickness of 0.8 mm.
Despite ongoing investigations, including many clinically valuable and biologically insightful studies and reviews, the subject of flat, acquired pigmented lesions of the conjunctiva continues to be challenging and to retain a certain degree of mystery. This is particularly true of the group of conditions collectively referred to as primary acquired melanosis, a designation with limitations that has been in circulation for over half a century and that is the main focus of this article. Zimmerman (1920–2013) has given a reminiscence of a gentleman’s difference of opinion regarding the fate of PAM between him and Algernon Reese (1900–1980), who took a special interest in the subject as the father of American ophthalmic oncology. It is proposed herein that the term PAM, which departs from descriptions of analogous skin conditions in dermatopathology, be abandoned because it is insufficiently specific and nondescriptive of the underlying cellular events. Over the years, attempts have been made to grade PAM with respect to mild, moderate, or severe degrees of atypia, but intraobserver and interobserver variability has often confounded reproducible accuracy. I have therefore come to the conclusion that the term primary acquired melanosis creates an unnecessary impediment for clearly communicating to ophthalmologists, dermatopathologists, and general pathologists the true biologic nature of the family of conditions it is meant to encompass.
The objectives of this review are 3-fold: (1) to describe in uncomplicated terms the origin and biology of melanocytes; (2) to describe and illustrate the clinical, histopathologic, and immunohistochemical features of conjunctival PAM; and (3) to offer an alternative pathologic diagnostic terminology and schema based on current concepts and language in dermatopathology. The prognosis for cutaneous and conjunctival melanomas has improved over the years, less as a consequence of advances in therapy, but more as a result of the treatment of lesions at the earliest stages before melanomatous evolution has occurred. For the purposes of this presentation, the term primary acquired melanosis has been episodically used, even though an argument is simultaneously made for the adoption of what is regarded as a more appropriate diagnostic nosology based on the vocabulary that is frequently employed today for cutaneous precursor lesions, namely “intraepithelial melanocytic proliferations (IMP).” The expedient of a dual description of the conjunctival disease (ie, IMP/PAM) in this exposition is intended to ease the intellectual transition to the new proposed diagnostic framework.
Any effort to revise an entrenched terminology will inevitably be considered as quixotic or even provocative by some experts and consequently encounter variable resistance. This writer believes, based on the rationale provided below, that this undertaking is nonetheless worthwhile. The current conjunctival terminology is isolated and parochial, failing to reflect that used in dermatopathology, nor does it convey a sufficiently precise impression of the underlying histopathologic features. Any reaction to the proposed new nosology described herein is considered a positive development, especially if it stimulates better communication and improved diagnostic accuracy. Since it is generally conceded that there are no clinical features that can distinguish premalignant from totally benign flat pigmented conjunctival lesions (although progressive clinical spread must be regarded with high suspicion), this article is focused mostly on the histopathologic aspects of diagnosis.
Melanocytic Origins and Biology
As the midline embryonic ectoderm invaginates to form the neural tube, cells also delaminate to create aggregates on the dorsolateral aspects of the tube called the neural crest ( Figure 1 , Top panel, left side). Among its many roles, the neural crest contributes Schwann cells that wrap around the axons of peripheral nerves; it also persists postembryonically as the paraspinal ganglia; and most germane to the current discussion, it spawns melanocytes, which undergo far-flung migrations to ultimately arrive in the skin and mucosae (including the conjunctiva) ( Figure 1 , Top panel), but also in other sites like the uvea of the eye (the retinal pigment epithelium derives not from the neural crest, but instead from the outer neuroectodermal layer of the optic cup).
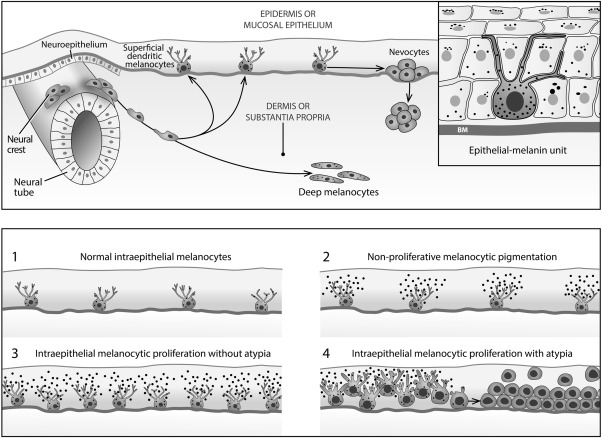
In the conjunctiva, most migrating melanocytes reach the epithelium to lodge among the basal germinal keratinocytes (squamous or epidermoid cells) at the level of the basement membrane. In more darkly complexioned individuals, some melanocytes may get “hung up” or arrested in their migration without ever settling in an epithelium ( Figure 1 , Top panel). Such melanocytic collections in the episclera or conjunctival substantia propria appear slate gray to blue, are stationary through life, and are postmitotic and therefore not responsible for generating conjunctival melanomas; they may, however, rarely create diffuse loose aggregates of cells resulting in the nevus of Ota or focal collections such as the blue nevus or cellular blue nevus.
The density of the basal dendritic melanocytes in the skin (intraepidermal) differs little among the various races, but it may vary to some degree among the races in mucosal epithelia. Melanocytes are normally found singly and widely spaced along the basement membrane rather than in nests ( Figure 1 , Top panel and Bottom panel #1). Non-nesting hyperplasias of normal-appearing dendritic melanocytes of whatever causation are referred to as manifesting a lentiginous growth pattern when confined to the basal layer of the epithelium. Their dendrites serve to distribute melanin granules by functioning as physiologic syringes that inject the granules into the surrounding keratinocytes (melanin–keratinocytic unit) ( Figure 1 , Top panel, inset on right side).
In more darkly complexioned individuals, bilateral, asymmetrical small golden or chocolate brown macules in the sun-exposed, interpalpebral epibulbar conjunctiva may frequently be seen. Such a lesion in the skin is designated as an ephelis or freckle. These totally benign lesions in the conjunctiva have been referred to as epithelial melanoses or hypermelanoses; because of the argument advanced below regarding confusion engendered by the term melanosis, I would prefer to call them nonproliferative melanocytic pigmentary spots, macules, or patches. These spots are movable upon the sclera and are formed as a result of the increased manufacture and transfer of melanin granules to keratinocytes rather than from melanocytic proliferation ( Figure 1 , Bottom panel #2). The pigment is most prominent in the cytoplasm of the basal germinal keratinocytes and becomes diluted in the suprabasal layers of the epithelium, where it is aggregated as supranuclear caps to protect the keratinocytic nuclear DNA from incident ultraviolet radiation. As long as there is preservation of functioning sinuous dendrites, melanin can additionally be transferred to higher-level keratinocytes and goblet cells. Unlike hyperplasia ( Figure 1 , Bottom panel #3), melanocytic neoplasia ( Figure 1 , Bottom panel #4) denotes an irreversible genetic alteration that allows cells to escape the usually operative negative feedbacks designed to arrest proliferation. Except when there is frank atypia, morphology alone is probably not adequate for separating these 2 processes when neoplasia is in its earliest phase.
Intraepithelial Melanocytic Proliferation/Primary Acquired Melanosis: Definitions and Preceding Studies
It cannot be overemphasized that the lesions subsumed under the rubric of IMP/PAM share one overarching feature: melanocytic proliferation ( Figure 1 , Bottom panel #3 and Bottom panel #4). Most melanomas (75%) arise from the dispersed dendritic melanocytes of IMP/PAM; a minority arise from persistent intraepithelial junctional mitotic activity of more rounded nevocytes that have lost their dendrites and are responsible for conventional nevomelanocytic nevi ( Figure 1 , Top panel, right side). Extremely rare melanomas can develop from a small number of melanocytes with an evanescent intraepithelial phase and are called nodular. The subepithelial nevocytes are postmitotic in contrast to the junctional nevocytes, and therefore do not play a significant role in generating melanomas. The stage of IMP/PAM corresponds to what has been characterized in dermatopathology as a variably prolonged radial growth phase within the epidermis, which represent in the skin precursors for lesions such as lentigo maligna, acral lentiginous, and superficial spreading subtypes of melanoma. Because of differences in anatomy, they are precluded from being reliably identified in the conjunctiva. A vertical growth phase develops when a nodule forms in the subepithelial connective tissues (the dermis of skin or the substantia propria of the conjunctiva), at which stage metastases may develop from increasing access to lymphatic channels as the nodule thickens. As the vertical thickness of the nodule progresses beyond 0.8 mm, metastases become more common.
The most pressing issue is to determine if a given lesion of IMP/PAM is likely to progress to invasive melanoma. In this connection the term “primary acquired melanosis” poses several difficulties. For example, today “primary” in oncology refers to the original site of a tumor as distinguished from its metastases (secondary lesions). In conjunctival PAM, on the other hand, “primary” is employed to rule out certain local extrinsic stimuli such as inflammation, climatic factors, or ultraviolet radiation that provoke melanin granule overproduction or deposition in melanophages but generally do not stimulate a conspicuous melanocytic proliferation. “Acquired” means that the lesion is not congenital, as in congenital ocular melanosis and oculodermal melanosis (nevus of Ota), conditions due to embryologically incompletely migrated melanocytes resident within the episcleral tissues (melanosis oculi) or conjunctival substantia propria. Via the Tyndall effect (light scattering of higher-energy blue wavelengths from deep cells) these locations confer a bluish or slate-gray discoloration that is immovable ( Figure 1 , Top panel). In practice, melanocytes in these locations do not serve as precursors for conjunctival melanoma, although conceptually this is a long-shot possibility. Unfortunately, “melanosis” is too noncommittal a term for a proliferative condition of the melanocytes. One can reserve the use of melanosis for the strictly nonproliferative synthesis of melanin granules, for which, however, the descriptive term of “intraepithelial nonproliferative melanocytic pigmentation” would seem to be more appropriate and accurately descriptive.
The precursor lesions to melanoma in the skin often survive as an intraepithelial radial growth phase at the edges of the invasive nodule. As mentioned, the precise histologic features of these radial extensions are not identically reproduced in the conjunctival epithelium, which is thinner than the epidermis and lacks the rete pegs and adnexal structures displayed in the skin. Moreover, the definition of the radial growth phase in the skin includes extension of atypical cells from the epithelium into the papillary dermis, which is not a microanatomic modification of the conjunctival substantia propria. Any degree of involvement of the superficial substantia propria is thus considered to be synonymous with microinvasion. The nomenclature for melanocytic intraepidermal proliferations that has been employed in dermatopathology includes the following: atypical melanocytic hyperplasia, pagetoid melanocytic proliferation, atypical lentiginous hyperplasia, pagetoid melanocytosis, precancerous melanosis, severe melanocytic dysplasia, dysplastic atypical nevus, and melanoma in situ. Pagetoid spread, according to dermatopathologic usage, indicates the percolation of individual atypical melanocytes throughout the multilaminar epidermis (buckshot appearance), typically reaching higher levels beyond the basal cell/basement membrane zone. This feature is more difficult to appreciate in the conjunctival epithelium, which can be as thin as 2–3 cell layers in some topographic zones such as the fornices. Two terms that are sufficiently encompassing and more clearly descriptive and that consequently seem preferable are “melanocytic intraepidermal neoplasia” and “intraepithelial melanocytic proliferation,” with or without atypia.
The latter term is preferred by some but not all dermatopathologists; it has been recommended herein for diagnosing conjunctival and periocular cutaneous eyelid intraepithelial lesions manifesting increased numbers of melanocytes. “Proliferation” is less ominous in terms of insurability issues than “neoplasia.” Both “proliferation” and “neoplasia” confined to the epithelium signify a nonmetastasizing condition because of a lack of access to lymphatic channels. Other efforts to bring conjunctival lesions into conformity with the criteria of dermatopathology have been discussed and alternative classifications have been offered. The closest new classification scheme to the one outlined in this article has been described by Damato and Coupland and restated by Kenawy and associates. They conducted a critical review of the term primary acquired melanosis and pointed out its failure to convey the biologic substrate behind the histopathologic diagnosis, similar to the critique already provided. They curiously do retain, however, the terms primary hypermelanosis for complexion-associated pigmentation and secondary melanosis for Addison disease and other externally provoked melanin granule production. Even if one makes an argument for salvaging melanosis as a histopathologic term, it is not obvious what diagnostic benefit accrues from the use of the intensified term hypermelanosis (hyperpigmentation), since the conjunctiva is normally nonpigmented, so that melanosis (pigmentation) should suffice.
Damato and Coupland recommend adoption of the term conjunctival melanocyte intraepithelial neoplasia (C-MIN) when there are increased numbers of melanocytes. The problem with invoking neoplasia rather than proliferation is that not all flat pigmented lesions are necessarily autonomously neoplastic or clonal—an indeterminate number may in fact be preneoplastic idiopathic hyperplasias or reactive hyperplasias due to an external stimulus (eg, ultraviolet radiation, postinflammatory) that occur within a normal range of physiologic responses and can be modulated by negative and suppressive feedback. They therefore are nonclonal and conceivably reversible or, at the most, stationary. The sharp definition of the interface between hyperplasias and neoplasias awaits further genetic studies. The presence of cytologic atypicality, however, can be employed as an indirect morphologic sign for inferring clonality. A significant virtue of Damato and Coupland’s approach is that they have devised a 12-category grading system of potentially unfavorable histopathologic features, which reflects an attempt to objectify microscopic risk factors, as further alluded to below.
Historically, the cornerstone of contemporary histopathologic interpretation of conjunctival acquired flat pigmentations was first developed by Zimmerman in 1966 and 1978, then elaborated upon by Folberg and associates in an outstanding series of articles from the Armed Forces Institute of Pathology (AFIP) in the 1980s and recently briefly summarized in a major pathologic oncologic textbook. Zimmerman’s first set of histopathologic diagnostic recommendations divided acquired melanosis into benign melanosis (subgroup A, represented either by pigmentation or by a few clusters of basal “nevus” cells) or marked “junctional” activity (subgroup B, exhibiting many nests of “disturbing” nevus cells). Cancerous melanosis was employed for invasive lesions. Subsequently he adopted the term benign acquired melanosis either with minimal or no intraepithelial melanocytic activity or moderate to marked activity with atypia, the latter typically associated with chronic inflammation. Malignant acquired melanosis was used for invasive lesions. The work of Folberg and associates is based on the determination of the presence of melanocytic atypia or its absence in biopsy specimens. The identification of epithelioid cells and their creation of discohesive and often confluent nests, and of pagetoid spread of individual cells or small cell clusters into higher levels of the epithelium, are their criteria for establishing risk for the eventual emergence of melanoma. In a defense of the term primary acquired melanosis Folberg has written: “to change nomenclature in the pathology report interrupts the chain of clinical logic…pathologists should strive to assist surgeons in the management of their cases, not by imposing on them a classification that may be biologically accurate but clinically confusing.”
There is no reason, however, why clinical and pathologic diagnoses should rely on the same vocabulary. Clinicians in all specialties are accustomed in their practices of medicine to making a descriptive and vaguely interpretive diagnosis using one vocabulary, and receiving post-biopsy a more specific, biologically grounded histopathologic diagnosis employing another more technical vocabulary. In place of “primary acquired melanosis” for a clinical diagnosis, one could describe the lesion simply as a pigmented macule, a flat pigmented patch, or another kindred term with accompanying measurements of the greatest horizontal and vertical dimensions. A clinical photograph is invaluable for documentation, especially if the lesion reflects from the epibulbar conjunctiva through the fornix onto the palpebral conjunctiva when straightforward planar measurements are more difficult to obtain. Furthermore, one microscopic aspect that has not been heavily stressed in previous work is an objective characterization with adequate nonimpressionistic specificity of the elements of cytologic atypia—eg, nuclear size, the presence of nucleoli, the prevalence of mitoses, etc. Until recently I employed the AFIP diagnostic scheme, which I still believe to be fundamentally sound, but I have rethought the terminology employed, which I believe can be redesigned to better communicate what is actually going on biologically.
Sugiura and associates have followed up on the work of Folberg and associates. While retaining the term primary acquired melanosis with atypia, they attempted to analyze in 29 cases the type of atypicality manifested by the lesions and came up with 2 forms of cytologic atypia along with 2 categories of architectural atypia concerning growth pattern. They did not employ the term melanoma in situ. Melanocytic hyperplasia along the basement membrane (lentiginous or basilar hyperplasia) was detected in 13 cases considered to be low risk for developing melanoma (although recurrences were often seen). It was composed of small to medium-sized polyhedral melanocytes with inconspicuous cytoplasm, a high nuclear-to-cytoplasmic ratio, and small to medium-sized hyperchromatic nuclei bereft of nucleoli. High-risk lesions (16 cases) exhibited larger epithelioid cells with pagetoid features, patently visible eosinophilic cytoplasm, medium to low nuclear-to-cytoplasm ratios, large clearly observable nucleoli, and often nuclei with a vesicular character. These lesions had a 63% rate of local recurrence and a metastatic rate of 25%. Microinvasion or frank invasion of the substantia propria were apt to be associated with intraepithelial epithelioid cells. Four cases were cytologically and architecturally mixed, in that disparate biopsies from separate regions of the same lesion could display either low-risk or high-risk characteristics. An overall ominous behavior (metastases) was aligned with the more worrisome cytopathologic and architectural features.
Most authors in this field (Damato and Coupland, Folberg, and Sugiura and associates ) have recommended treatment for all preinvasion lesions of primary acquired melanosis with atypia. Sugiura’s observations unaccountably were not carried forward by one of his co-authors and are not included in that author’s revised textbook completely dedicated to cutaneous and mucosal melanocytic proliferations. This textbook offers a few terminological changes. Flat pigmentations are divided into 2 main groups, conjunctival hypermelanosis (either without perceptible melanocytic hyperplasia or with cytologically “banal” hyperplasia) and conjunctival hypermelanosis with atypical melanocytic hyperplasia (either lentiginous [basal] or pagetoid). In essence, the term primary acquired melanosis was replaced by hypermelanosis, which continues to obscure the precise underlying pathologic and biologic substrate as discussed above.
Melanoma in situ is a morphologic entity denoting virtually total replacement of the squamous epithelium with sparing of a monolayer of flattened surface keratinocytes. Invasive disease, however, may apparently arise more commonly from less extreme forms of intraepithelial melanocytic proliferation and has also been considered to be potentially equivalent to melanoma in situ. This situation parallels that of squamous lesions—namely, do squamous carcinomas obligatorily arise from full-thickness carcinomas in situ, or can squamous dysplasias of moderate severity also serve as precursor lesions? Damato and Coupland have circumvented subjective grading of intraepithelial conjunctival melanocytic disease by offering a cogent stratification scheme in the form of a grading list that attempts to evaluate 12 parameters regarding atypia. Individual consideration is given to intraepithelial melanocytic patterns of growth, various degrees of vertical extension upwards within the epithelium, the characterization of nuclear size, ampleness of cytoplasm, and the presence of nucleoli and mitoses, among other features. Since it requires more concentrated attention for analyzing cases, time will tell whether ophthalmic pathologists will expend the intellectual energy entailed in its use, which will determine whether it will be widely adopted. Anyone with a deep interest in the subject under discussion should read this article. I came across it in preparing the bibliography for this paper after I had formulated and finalized my own thoughts, which have substantial terminological overlap with the contribution of Damato and Coupland. My minor quibbles should not detract from the historical precedence and credit that must be accorded to their work, which has received less attention in the United States than it ought to have, owing to its publication in the Australian ophthalmic literature.
Intraepithelial Melanocytic Proliferation/Primary Acquired Melanosis: Definitions and Preceding Studies
It cannot be overemphasized that the lesions subsumed under the rubric of IMP/PAM share one overarching feature: melanocytic proliferation ( Figure 1 , Bottom panel #3 and Bottom panel #4). Most melanomas (75%) arise from the dispersed dendritic melanocytes of IMP/PAM; a minority arise from persistent intraepithelial junctional mitotic activity of more rounded nevocytes that have lost their dendrites and are responsible for conventional nevomelanocytic nevi ( Figure 1 , Top panel, right side). Extremely rare melanomas can develop from a small number of melanocytes with an evanescent intraepithelial phase and are called nodular. The subepithelial nevocytes are postmitotic in contrast to the junctional nevocytes, and therefore do not play a significant role in generating melanomas. The stage of IMP/PAM corresponds to what has been characterized in dermatopathology as a variably prolonged radial growth phase within the epidermis, which represent in the skin precursors for lesions such as lentigo maligna, acral lentiginous, and superficial spreading subtypes of melanoma. Because of differences in anatomy, they are precluded from being reliably identified in the conjunctiva. A vertical growth phase develops when a nodule forms in the subepithelial connective tissues (the dermis of skin or the substantia propria of the conjunctiva), at which stage metastases may develop from increasing access to lymphatic channels as the nodule thickens. As the vertical thickness of the nodule progresses beyond 0.8 mm, metastases become more common.
The most pressing issue is to determine if a given lesion of IMP/PAM is likely to progress to invasive melanoma. In this connection the term “primary acquired melanosis” poses several difficulties. For example, today “primary” in oncology refers to the original site of a tumor as distinguished from its metastases (secondary lesions). In conjunctival PAM, on the other hand, “primary” is employed to rule out certain local extrinsic stimuli such as inflammation, climatic factors, or ultraviolet radiation that provoke melanin granule overproduction or deposition in melanophages but generally do not stimulate a conspicuous melanocytic proliferation. “Acquired” means that the lesion is not congenital, as in congenital ocular melanosis and oculodermal melanosis (nevus of Ota), conditions due to embryologically incompletely migrated melanocytes resident within the episcleral tissues (melanosis oculi) or conjunctival substantia propria. Via the Tyndall effect (light scattering of higher-energy blue wavelengths from deep cells) these locations confer a bluish or slate-gray discoloration that is immovable ( Figure 1 , Top panel). In practice, melanocytes in these locations do not serve as precursors for conjunctival melanoma, although conceptually this is a long-shot possibility. Unfortunately, “melanosis” is too noncommittal a term for a proliferative condition of the melanocytes. One can reserve the use of melanosis for the strictly nonproliferative synthesis of melanin granules, for which, however, the descriptive term of “intraepithelial nonproliferative melanocytic pigmentation” would seem to be more appropriate and accurately descriptive.
The precursor lesions to melanoma in the skin often survive as an intraepithelial radial growth phase at the edges of the invasive nodule. As mentioned, the precise histologic features of these radial extensions are not identically reproduced in the conjunctival epithelium, which is thinner than the epidermis and lacks the rete pegs and adnexal structures displayed in the skin. Moreover, the definition of the radial growth phase in the skin includes extension of atypical cells from the epithelium into the papillary dermis, which is not a microanatomic modification of the conjunctival substantia propria. Any degree of involvement of the superficial substantia propria is thus considered to be synonymous with microinvasion. The nomenclature for melanocytic intraepidermal proliferations that has been employed in dermatopathology includes the following: atypical melanocytic hyperplasia, pagetoid melanocytic proliferation, atypical lentiginous hyperplasia, pagetoid melanocytosis, precancerous melanosis, severe melanocytic dysplasia, dysplastic atypical nevus, and melanoma in situ. Pagetoid spread, according to dermatopathologic usage, indicates the percolation of individual atypical melanocytes throughout the multilaminar epidermis (buckshot appearance), typically reaching higher levels beyond the basal cell/basement membrane zone. This feature is more difficult to appreciate in the conjunctival epithelium, which can be as thin as 2–3 cell layers in some topographic zones such as the fornices. Two terms that are sufficiently encompassing and more clearly descriptive and that consequently seem preferable are “melanocytic intraepidermal neoplasia” and “intraepithelial melanocytic proliferation,” with or without atypia.
The latter term is preferred by some but not all dermatopathologists; it has been recommended herein for diagnosing conjunctival and periocular cutaneous eyelid intraepithelial lesions manifesting increased numbers of melanocytes. “Proliferation” is less ominous in terms of insurability issues than “neoplasia.” Both “proliferation” and “neoplasia” confined to the epithelium signify a nonmetastasizing condition because of a lack of access to lymphatic channels. Other efforts to bring conjunctival lesions into conformity with the criteria of dermatopathology have been discussed and alternative classifications have been offered. The closest new classification scheme to the one outlined in this article has been described by Damato and Coupland and restated by Kenawy and associates. They conducted a critical review of the term primary acquired melanosis and pointed out its failure to convey the biologic substrate behind the histopathologic diagnosis, similar to the critique already provided. They curiously do retain, however, the terms primary hypermelanosis for complexion-associated pigmentation and secondary melanosis for Addison disease and other externally provoked melanin granule production. Even if one makes an argument for salvaging melanosis as a histopathologic term, it is not obvious what diagnostic benefit accrues from the use of the intensified term hypermelanosis (hyperpigmentation), since the conjunctiva is normally nonpigmented, so that melanosis (pigmentation) should suffice.
Damato and Coupland recommend adoption of the term conjunctival melanocyte intraepithelial neoplasia (C-MIN) when there are increased numbers of melanocytes. The problem with invoking neoplasia rather than proliferation is that not all flat pigmented lesions are necessarily autonomously neoplastic or clonal—an indeterminate number may in fact be preneoplastic idiopathic hyperplasias or reactive hyperplasias due to an external stimulus (eg, ultraviolet radiation, postinflammatory) that occur within a normal range of physiologic responses and can be modulated by negative and suppressive feedback. They therefore are nonclonal and conceivably reversible or, at the most, stationary. The sharp definition of the interface between hyperplasias and neoplasias awaits further genetic studies. The presence of cytologic atypicality, however, can be employed as an indirect morphologic sign for inferring clonality. A significant virtue of Damato and Coupland’s approach is that they have devised a 12-category grading system of potentially unfavorable histopathologic features, which reflects an attempt to objectify microscopic risk factors, as further alluded to below.
Historically, the cornerstone of contemporary histopathologic interpretation of conjunctival acquired flat pigmentations was first developed by Zimmerman in 1966 and 1978, then elaborated upon by Folberg and associates in an outstanding series of articles from the Armed Forces Institute of Pathology (AFIP) in the 1980s and recently briefly summarized in a major pathologic oncologic textbook. Zimmerman’s first set of histopathologic diagnostic recommendations divided acquired melanosis into benign melanosis (subgroup A, represented either by pigmentation or by a few clusters of basal “nevus” cells) or marked “junctional” activity (subgroup B, exhibiting many nests of “disturbing” nevus cells). Cancerous melanosis was employed for invasive lesions. Subsequently he adopted the term benign acquired melanosis either with minimal or no intraepithelial melanocytic activity or moderate to marked activity with atypia, the latter typically associated with chronic inflammation. Malignant acquired melanosis was used for invasive lesions. The work of Folberg and associates is based on the determination of the presence of melanocytic atypia or its absence in biopsy specimens. The identification of epithelioid cells and their creation of discohesive and often confluent nests, and of pagetoid spread of individual cells or small cell clusters into higher levels of the epithelium, are their criteria for establishing risk for the eventual emergence of melanoma. In a defense of the term primary acquired melanosis Folberg has written: “to change nomenclature in the pathology report interrupts the chain of clinical logic…pathologists should strive to assist surgeons in the management of their cases, not by imposing on them a classification that may be biologically accurate but clinically confusing.”
There is no reason, however, why clinical and pathologic diagnoses should rely on the same vocabulary. Clinicians in all specialties are accustomed in their practices of medicine to making a descriptive and vaguely interpretive diagnosis using one vocabulary, and receiving post-biopsy a more specific, biologically grounded histopathologic diagnosis employing another more technical vocabulary. In place of “primary acquired melanosis” for a clinical diagnosis, one could describe the lesion simply as a pigmented macule, a flat pigmented patch, or another kindred term with accompanying measurements of the greatest horizontal and vertical dimensions. A clinical photograph is invaluable for documentation, especially if the lesion reflects from the epibulbar conjunctiva through the fornix onto the palpebral conjunctiva when straightforward planar measurements are more difficult to obtain. Furthermore, one microscopic aspect that has not been heavily stressed in previous work is an objective characterization with adequate nonimpressionistic specificity of the elements of cytologic atypia—eg, nuclear size, the presence of nucleoli, the prevalence of mitoses, etc. Until recently I employed the AFIP diagnostic scheme, which I still believe to be fundamentally sound, but I have rethought the terminology employed, which I believe can be redesigned to better communicate what is actually going on biologically.
Sugiura and associates have followed up on the work of Folberg and associates. While retaining the term primary acquired melanosis with atypia, they attempted to analyze in 29 cases the type of atypicality manifested by the lesions and came up with 2 forms of cytologic atypia along with 2 categories of architectural atypia concerning growth pattern. They did not employ the term melanoma in situ. Melanocytic hyperplasia along the basement membrane (lentiginous or basilar hyperplasia) was detected in 13 cases considered to be low risk for developing melanoma (although recurrences were often seen). It was composed of small to medium-sized polyhedral melanocytes with inconspicuous cytoplasm, a high nuclear-to-cytoplasmic ratio, and small to medium-sized hyperchromatic nuclei bereft of nucleoli. High-risk lesions (16 cases) exhibited larger epithelioid cells with pagetoid features, patently visible eosinophilic cytoplasm, medium to low nuclear-to-cytoplasm ratios, large clearly observable nucleoli, and often nuclei with a vesicular character. These lesions had a 63% rate of local recurrence and a metastatic rate of 25%. Microinvasion or frank invasion of the substantia propria were apt to be associated with intraepithelial epithelioid cells. Four cases were cytologically and architecturally mixed, in that disparate biopsies from separate regions of the same lesion could display either low-risk or high-risk characteristics. An overall ominous behavior (metastases) was aligned with the more worrisome cytopathologic and architectural features.
Most authors in this field (Damato and Coupland, Folberg, and Sugiura and associates ) have recommended treatment for all preinvasion lesions of primary acquired melanosis with atypia. Sugiura’s observations unaccountably were not carried forward by one of his co-authors and are not included in that author’s revised textbook completely dedicated to cutaneous and mucosal melanocytic proliferations. This textbook offers a few terminological changes. Flat pigmentations are divided into 2 main groups, conjunctival hypermelanosis (either without perceptible melanocytic hyperplasia or with cytologically “banal” hyperplasia) and conjunctival hypermelanosis with atypical melanocytic hyperplasia (either lentiginous [basal] or pagetoid). In essence, the term primary acquired melanosis was replaced by hypermelanosis, which continues to obscure the precise underlying pathologic and biologic substrate as discussed above.
Melanoma in situ is a morphologic entity denoting virtually total replacement of the squamous epithelium with sparing of a monolayer of flattened surface keratinocytes. Invasive disease, however, may apparently arise more commonly from less extreme forms of intraepithelial melanocytic proliferation and has also been considered to be potentially equivalent to melanoma in situ. This situation parallels that of squamous lesions—namely, do squamous carcinomas obligatorily arise from full-thickness carcinomas in situ, or can squamous dysplasias of moderate severity also serve as precursor lesions? Damato and Coupland have circumvented subjective grading of intraepithelial conjunctival melanocytic disease by offering a cogent stratification scheme in the form of a grading list that attempts to evaluate 12 parameters regarding atypia. Individual consideration is given to intraepithelial melanocytic patterns of growth, various degrees of vertical extension upwards within the epithelium, the characterization of nuclear size, ampleness of cytoplasm, and the presence of nucleoli and mitoses, among other features. Since it requires more concentrated attention for analyzing cases, time will tell whether ophthalmic pathologists will expend the intellectual energy entailed in its use, which will determine whether it will be widely adopted. Anyone with a deep interest in the subject under discussion should read this article. I came across it in preparing the bibliography for this paper after I had formulated and finalized my own thoughts, which have substantial terminological overlap with the contribution of Damato and Coupland. My minor quibbles should not detract from the historical precedence and credit that must be accorded to their work, which has received less attention in the United States than it ought to have, owing to its publication in the Australian ophthalmic literature.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


