Complications of Pharmacotherapy
Matteo Forlini
Gian Maria Cavallini
Luca Campi
Virgilio Morales-Canton
Paolo Rossini
Adriana Bratu
Cesare Forlini
Giuseppe Di Stefano
INTRODUCTION
Pharmacological therapy of ocular diseases comprises a vast variety of drug classes available for use in treating the many different pathological situations, including antibiotics and antivirals, fibrinolytic molecules (t-PA), new generation antiedema drugs (Triamcinolone acetonide (TA)) and anti-vascular endothelial growth factor (anti-VEGF) agents for the treatment of neovascular and exudative diseases of the retina. All of these drugs are currently used in clinical practice and/or as support to surgery.
The first attempts at intravitreal injections were performed in the early 1900s to treat retinal detachment. Since then, intravitreal injections as treatment for eye disease have progressed significantly (injections of antibiotics, silicon oil, antiviral agents, methotrexate, t-PA, hyaluronidase, corticosteroids, through to the more recent anti-VEGF agents) over the years.
In addition to their undisputed beneficial effects and certain usefulness in resolving even very severe pathological situations, all the drugs used in local eye therapy present a range of side effects and complications on account of the administration route and method, and adverse effects that sometimes trigger pathological reactions and situations, equally or even more severe than the original pathological condition.
The introduction of pharmacological therapy in maculopathy treatment dates from the late 1970s, with the use of intravitreal steroids (triamcinolone and dexamethasone) to treat exudative and neovascular maculopathies.
Since 2001, the use of TA has become increasingly efficacious as a drug with a potent anti-inflammatory and antiedema action for the treatment of exudative diabetic maculopathy,
age-related macular degeneration (AMD), and postsurgical and inflammatory (autoimmune and idiopathic) maculopathy. As of 2004, a new class of anti-VEGF drugs started to become commonly used to treat exudative maculopathy and all forms of intraocular neovascularization (proliferative diabetic retinopathy, neovascular glaucoma, retinopathy of prematurity (ROP), autoimmune diseases, etc.).
Periocular and intraocular injection preparations have been used in clinical practice for many years.
Several molecules are useful for the treatment of the many different ocular conditions by subconjunctival, subtenon, and intravitreal injection.
Molecules, injection techniques, and standard procedures are constantly upgraded. However, differences persist in certain parameters, so that while in Europe, these injections are performed in sterile conditions in the theatre, in the USA and other American countries, intravitreal injections are performed in the surgery in a nonsterile environment.
The drugs used for intraocular or periocular applications can have local and systemic side effects and complications.
These events may be caused by the active substance, administration method and route, the surgeon’s skill, the preparation of drug, surrounding conditions (sterility) and/or self-administration in the postoperative period.
The complications associated to the use of intravitreal drugs can be divided into two groups: 1) those consequential to the injection technique and 2) those caused by the pharmacological effects of the molecules, which can in turn be either systemic or ocular.
TECHNIQUE-RELATED COMPLICATIONS
Subconjunctival-Subtenon Drugs
Some drugs, such as antidiabetic, antiviral, and anti-inflammatory agents, can be administered by the subconjunctival or subtenon routes, which, despite not being complication free, are rarely associated with adverse events during the operative or immediate or late postoperative periods.
The subconjunctival route entails the injection of a drug into the conjunctival stroma; it is not necessary to cut the conjunctiva and topical anesthesia is usually used.
The most common complications are slight, such as corneal abrasions, subconjunctival hemorrhage, the sensation of swelling or presence of a foreign body, and transient conjunctival hyperemia associated with slight irritation.
After injection, the medication passes into vitreous chamber from the subconjunctival space by means of diastatic sclerotomy, even when the mini-invasive sutureless surgery technique is used (1).
For certain classes of aminoglycoside antibiotics (gentamicin), this is associated to a high risk of intravitreal toxicity, such as retinal ischemia and edema, reduction in the amplitude of the electroretinogram (ERG) b-wave caused by photoreceptor necrosis and macular hemorrhage, with poor functional prognosis.
The subtenon technique entails opening a small conjunctival buttonhole and the underlying tenonian capsule, in which the blunt needle is inserted and pushed backwards.
Technique-related complications are usually mild and similar to those of the subconjunctival technique. However, after anti-inflammatory and antiviral drug use, the risk of infection is higher (2, 3, 4 and 5).
The risk of scleral perforation is low, whereas the drug can have serious consequences, such as lid ptosis and orbital fat prolapse (6), local subconjunctival abscess and ocular hypertension.
This technique can be performed using topical anesthesia and some patients may feel pain.
Subconjunctival injection of lidocaine can reduce the risk of pain during the transscleral injection; however, discomfort may be experienced when the subconjunctival injection is made.
Some patients may experience substantial subconjunctival hemorrhage, which resolves in one to two weeks without consequences.
Intravitreal Injection
The use of intravitreal drugs exposes the patient to complications connected to technical risk.
The injection technique is fairly standard and generally consists of a dose of 0.05 ml (for anti-VEGF) to 0.1 ml (for TA) of the agent, except for gas injection, where the dose is higher.
It is performed using a 25-, 27-, or 30-gauge needle, via the transconjunctival route 2 to 5 mm (standard average is 3-3.5-4 mm) from the limbus, depending on whether one is in the presence of a pseudophakic or phakic condition. Although there are no preferential injection quadrants, some practitioners prefer to avoid the lower quadrant, where more germs accumulate, in order to avoid the risk of intravitreal inoculation and consequent inflammation.
Injection can be either direct or performed through a small scleral tunnel incision. The aim of this second technique is to reduce the risk of leakage of the drug and/or vitreal gel into the subconjunctival space.
The conjunctiva can be moved 2 mm from the injection site, to avoid the conjunctival hole from coinciding with the scleral hole, in order to prevent direct contact with the external environment. There are a number of potential injection complications, ranging in severity from very bland to extremely serious.
The subconjunctival hemorrhage is undoubtedly the most frequent of these and although it is usually generally slight and self-limiting, it can be more substantial and a cause of concern to the patient; however, the situation normally resolves spontaneously within a few days.
The ocular surface can be interested by the intravitreal injection technique, as the needle may accidentally touch the cornea causing abrasion; this resolves spontaneously within a few hours when correct therapy is administered.
The potential complications of injection via the pars plana route include touching of the lens or intraocular lens (IOL); the former may cause local (peripheral, paracentral or central) opacity that may later develop into a cataract.
In pseudophakic patients, especially those who have undergone sulcus implantation following capsular rupture, accidental contact may cause IOL dislocation or subluxation with the risk of vitreous prolapse in the anterior chamber.
Vitreous hemorrhage is a relatively common complication of intravitreal injection and occurs in 3% to 5% of patients. Vitreous hemorrhage appears to be more commonly associated with injections performed using a 27-gauge rather than a 30-gauge needle.
Some clinicians prefer using a larger caliber needle to reduce the likelihood of needle obstruction during injection, even at the cost of creating a larger hole (risk of hypotonia and vitreous engagement).
One particular case is that of the vitrectomy patient, where the initial sclerotomy must be avoided and the injection made at a distance or in a different quadrant, to avoid partially reopening previous incisions, producing diastasis, subconjunctival leakage, loss of the remaining vitreous humor, conjunctival chemosis, and consequent hypotonia. This only applies to procedures performed a short time after the vitrectomy and from two months thereafter the only problem is likely to be a greater risk of bleeding due to conjunctival congestion in the area in which the sclerotomy was performed.
Intravitreal injection of an anti-VEGF agent is safe in terms of intraocular pressure (IOP) elevation with a return to levels under 25 mm Hg within 30 minutes from the injection. In rare cases, an increase in pressure may persist beyond this interval and it is in any case good practice to monitor IOP after intravitreal injections. Paracentesis evacuation could be taken into consideration in glaucoma patients to obtain hypotonia (Hollands, 2007).
TRANSCONJUNCTIVAL INTRAOCULAR INJECTION TECHNIQUE
The transconjunctival intravitreal injection procedure is conducted as follows.
In sterile conditions, the surgeon wears a disposable gown and gloves, prepares the surgical field with povidone-iodine 10% vol. and uses sterile drapes and lid speculum. Topical anesthesia is administered with lidocaine 4%.
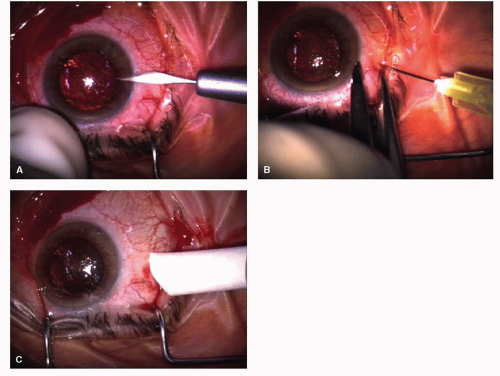
The surgeon uses a caliper to mark a distance of 3.5 mm (in aphakic/pseudophakic patients) or 4 mm (in phakic ones) from the limbus, before moving the conjunctiva to prevent the conjunctival hole coinciding with the scleral hole and preceding with transconjunctival injection of the drug (Fig. 53-1).
TECHNIQUE VARIATION (PERSONAL STRATEGY OF C. FORLINI)
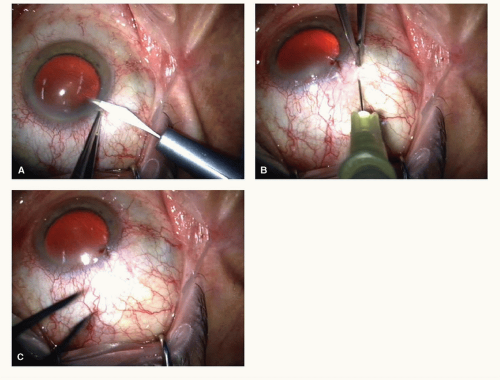
The only variation consists in an anterior chamber paracentesis performed using a 15° slit knife as the first step: a slight emptying of anterior chamber (AC) causes hypotonia, which has two advantages: (a) it prevents IOP elevation and consequently (b) it prevents vitreous fibrils and/or drug fluid from leaking into the subconjunctival space (bleb) after the injection (Figs. 53-2 and 53-3).
The formation of subconjunctival blebs must be avoided, as they would trap part of the drug and/or vitreous fibrils. In the former case, the pharmacological effect is impaired and in the later, vitreous fibril incarceration may cause dangerous late vitreous traction.
In conclusion, AC paracentesis before IV injection offers important advantages:
it avoids intraoperative or short-term IOP elevation
it prevents subconjunctival blebs and consequently there is:
no risk of leakage and loss of some of the drug (→impaired pharmacological action)
no risk of vitreous fibril incarceration (→late vitreoretinal traction)
DRUG-RELATED COMPLICATIONS
Anti-VEGF Agents
VEGF is a protein secreted by retinal pigment epithelium that acts on endothelial cells and is an essential factor in the development of choroidal neovascularization. In physiology, VEGF is essential for normal embryonic development; it plays a role in the female reproductive cycle, is expressed in tissue in the
brain, kidney and gastrointestinal mucosa, contributes to wound healing and bone formation, and promotes the formation of new blood vessels after myocardial ischemia.
brain, kidney and gastrointestinal mucosa, contributes to wound healing and bone formation, and promotes the formation of new blood vessels after myocardial ischemia.
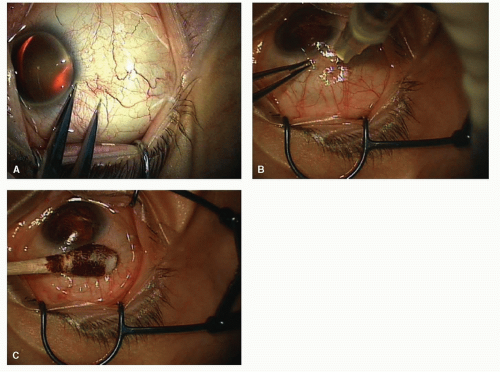 Figure 53-1. A: Marking a distance of 4 mm from the limbus; B: Injection of 0.05 cc of Bevacizumab; C: Massage with cotton bud soaked in povidoneiodine. |
VEGF and VEGF receptors are naturally expressed in the healthy eye and VEGF may play a protective role in maintaining adequate blood flow to the retinal pigment epithelium (RPE) and photoreceptors.
In ocular pathology, VEGF is involved in:
Neovascular AMD
Diabetic retinopathy
Retinal vein occlusion
Retinopathy of prematurity
Corneal neovascularization
Iris neovascularization
VEGF has the following properties:
Angiogenesis stimulant
Potent inducer of vascular permeability
Vascular survival factor
“Fenestration” promoting factor
Pro-inflammatory action
Neuroprotective action
To assess the effects of the intravitreal injection of each drug, both topical and systemic side effects are to be considered.
Despite the small amount of drug injected into the eye, a minimal quantity of it may act at a systemic level. Bearing in mind that VEGF intervenes in several biological processes, including growth, repair, and regeneration, possible side effects may involve the circulatory system.
Pegaptanib (Macugen)
Pegaptanib is a pegylated 28-nucleotide RNA aptamer (aptamers are synthetic oligonucleotides with binding properties similar to those of antibodies). It was the first anti-VEGF agent approved for the treatment of vascular eye disease. By blocking VEGF165, pegaptanib inhibits angiogenesis and vascular permeability.
The VISION study (VEGF Inhibition Study in Ocular Neovascularization) studies the safety and efficacy of pegaptanib. This concurrent, randomized, double-blind, controlled, doseranging study involves 1,190 patients at 117 centers worldwide; 295 receive 0.3 mg pegaptanib and 298 receive usual care with sham injections every 6 weeks. The objective of this study is to establish the safe, efficacious dose of intravitreal
pegaptanib in patients with subfoveal choroidal neovascularization (CNV) secondary to AMD.
pegaptanib in patients with subfoveal choroidal neovascularization (CNV) secondary to AMD.
The ocular adverse events observed in this study are:
Eye pain
Punctuate keratitis
Vitreous floaters
Vitreous opacity
Anterior chamber inflammation
Increased IOP. Pegaptanib injections cause a significant transient rise in IOP that considerably diminishes within 30 minutes post-injection, but can take as long as one hour. This may damage the optic nerve, particularly in patients with advanced glaucoma (7). Pegaptanib injection in this limited series would seem to be safe from an IOP standpoint in the short-term. Postinjection IOP monitoring may not be necessary (8).
Traumatic cataract
Retinal detachment
RPE tears post-pegaptanib (Macugen) injection (9).
Endophthalmitis. After the first year, the prevalence of endophthalmitis drops from 0.18% to 0.07% due to the following factors:
Preinjection antibiotics and povidone-iodine; use of lid speculum, sterile drape and sterile gloves.
Greater experience of clinicians in performing intravitreal triamcinolone (IVT) injections.
The most common systemic adverse events are:
Cardiac disorders
Neoplasia and nervous system disorders
Gastrointestinal disorders
Respiratory, thoracic and mediastinal disorders
Renal and urinary tract disorders
There is no statistically significant difference in the onset of these adverse events between patients who received pegaptanib and those who received usual care with sham injections.
Ranibizumab (Lucentis)
Ranibizumab (Lucentis, Genentech, Inc., South San Francisco, CA) is a specific affinity-matured, recombinant, humanized anti-VEGF-A neutralizing antibody fragment (Fab). The fragment is one third the size of a full-length antibody and readily penetrates all layers of the retina after intravitreal injection. Ranibizumab is
being developed as a potential treatment for various VEGFA-mediated ocular vascular diseases and is being used in phase III trials for treatment of neovascular AMD. The one-year results from a 2-year phase III trial on the treatment of minimally classic and occult-only AMD-related choroidal neovascularization indicate that monthly injection of 0.3 or 0.5 mg ranibizumab halted the growth of the choroidal neovascularization lesion, reduced vascular leakage, and in one quarter to one third of treated patients, respectively, improved visual acuity (VA).
being developed as a potential treatment for various VEGFA-mediated ocular vascular diseases and is being used in phase III trials for treatment of neovascular AMD. The one-year results from a 2-year phase III trial on the treatment of minimally classic and occult-only AMD-related choroidal neovascularization indicate that monthly injection of 0.3 or 0.5 mg ranibizumab halted the growth of the choroidal neovascularization lesion, reduced vascular leakage, and in one quarter to one third of treated patients, respectively, improved visual acuity (VA).
The ANCHOR, MARINA, and SAILOR trials studied the safety and efficacy of Ranibizumab.
The ocular adverse events showed a 1% prevalence in the ANCHOR study and a 1.5% prevalence in the MARINA study:
Endophthalmitis
Uveitis. Another study showed that 500 μg of ranibizumab was the maximum tolerated dose. At the higher dose of 1000 μg, significant intraocular inflammation was observed (10).
Iridocyclitis and injection-site reactions, mild transient ocular inflammation was the most common post-injection adverse event.
Retinal detachment
Retinal rupture
Vitreous and retinal hemorrhage
Sub-retinal fibrosis
RPE tears (11)
Traumatic cataract
Transient IOP elevation
Conjunctival hemorrhage
Ocular pain
Vitreous floaters
Vitreous detachment
Ocular hyperemia
Reduced visual acuity or blurred vision
Dry eye syndrome
The analysis of systemic adverse effects of Lucentis (systemic adverse events) in both studies (ANCHOR, MARINA) has shown:
The incidence of acute elevation of systemic blood pressure in patients treated with lucentis was similar to or lower than the control group
No statistically significant difference in the incidence of thromboembolic diseases (myocardial infarction, stroke,
cerebral infarction) was observed between the various treatment groups
No death has been attributed to Lucentis
Ranibizumab (Lucentis) was approved by the US Food and Drug Administration (FDA) for intravitreal injection at a dose of 0.5 mg; the manufacturer recently issued a letter to physicians warning of the increased risk of stroke at the FDA-approved dose as compared to a lower studied dose of 0.3 mg. An interim analysis of the ongoing SAILOR study revealed a 1.2% risk of stroke in the 0.5 mg arm versus 0.3% in the 0.3 mg arm (p = .02). It is unclear whether the trend toward a higher risk of stroke in patients receiving the 0.5 mg dose of ranibizumab will persist in final analysis, however details such as causality, topography, and severity of stroke in the SAILOR study also need to be defined.
Bevacizumab (Avastin)
Bevacizumab (Avastin) is a recombinant humanized monoclonal IgG1 antibody that inhibits human VEGF. The drug is approved by the US Food and Drug Administration for intravenous use in combination with 5-fluorouracil-based chemotherapy for metastatic colorectal cancer. It has been administered intravitreally in VEGF-mediated diseases, such as choroidal neovascularization, central retinal vein occlusion, proliferative diabetic retinopathy, and other retinal diseases. It has not been approved by the FDA for intravitreal use and therefore it may only be used in an off-label setting. The most commonly used dose for intravitreal injection is currently 1.25 mg (0.05 ml) in the United States, although up to 2.50 mg (0.1 ml) may be used. Because such a small amount of drug from a large vial is required to treat eye disease, ophthalmologists have been obtaining the drug from compounding pharmacies that divide the vial into smaller amounts, at a lower cost.
To date, there are no randomized clinical trials demonstrating the effects of the treatment with bevacizumab for ocular vascular diseases; however, there are several case series concerning this matter.
The ocular complications of intravitreal injection of bevacizumab are:
Bacterial endophthalmitis
Tractional retinal detachments
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree