Purpose
To evaluate complications and clinical outcomes of Descemet stripping automated endothelial keratoplasty (DSAEK) with intraocular lens (IOL) exchange compared with DSAEK alone.
Design
Retrospective, interventional case series.
Methods
DSAEK was performed in 19 eyes in which the anterior chamber IOL was exchanged for a posterior chamber IOL (study group) and in 188 eyes in which the posterior chamber IOL was left in place (comparison group). The complications of graft dislocations, primary graft failure episodes, and pupillary block were recorded for all eyes. Six-month best spectacle-corrected visual acuity and mean central endothelial cell density were measured prospectively and then compared with preoperative values for all eyes.
Results
Dislocations occurred in 0 (0%) of 19 eyes in the study group and in 5 (3%) of 188 eyes in the comparison group ( P = .47), with 0 primary graft failures and 0 pupillary block episodes in either group. Preoperative mean best spectacle-corrected visual acuity for those eyes without any underlying ocular comorbidities was 20/205 and 20/100 in the study and comparison groups, respectively ( P = .18). Mean best spectacle-corrected visual acuity at 6 months improved to 20/48 in the study group and to 20/34 in the comparison group, a statistically significant difference ( P = .01). Mean donor cell loss at 6 months was 33% in the study group and 26% in the comparison group ( P = .18).
Conclusions
Concurrent IOL exchange with DSAEK surgery does not increase the dislocation, primary graft failure, or pupillary block rates in the immediate postoperative period. Donor endothelial cell loss in DSAEK was not increased significantly by IOL exchange. Visual acuity was slightly worse after combined surgery than after DSAEK alone.
Endothelial keratoplasty is now the leading surgical method used to correct the effects of endothelial dysfunction by selectively removing and replacing diseased endothelium with healthy donor tissue. The evolution and potential benefits of endothelial keratoplasty, and specifically Descemet stripping automated endothelial keratoplasty (DSAEK), where a microkeratome is used to prepare the donor lenticule, have been outlined previously. Compared with full-thickness penetrating keratoplasty (PK), DSAEK provides improved best spectacle-corrected visual acuity (BSCVA) without inducing significant astigmatism. DSAEK also offers increased wound stability and a more rapid recovery than PK surgery.
This new technique of endothelial transplantation comes with new challenges as well, including complications in the form of graft dislocations, pupillary block, and primary graft failure episodes. Multiple studies also have demonstrated that endothelial cell density (ECD) declines after all forms of endothelial keratoplasty, but at a level comparable with that of PK. However, with increasing experience and using the consistent and well-established technique of DSAEK described by Terry and associates, complication rates have been kept at a minimum, with a primary graft failure rate of 0% and dislocation rates of less than 2%, as noted in our initial 500 DSAEK cases.
As the realm of endothelial keratoplasty continues to expand, the indications for transplantation have increased to encompass nearly all causes of endothelial dysfunction. One specific area of interest is the effect of anterior chamber (AC) intraocular lenses (IOLs) on corneal edema. It is well known that ill-fitting AC IOLs can destroy healthy endothelial cells, but the process of implantation itself can also prove to be very destructive and can lead to eventual bullous keratopathy. The most common treatment method in the past for this condition required AC IOL explantation and replacement with a sutured posterior chamber (PC) IOL combined with PK; however, the patient again was exposed to the risks and long-term sequelae of a full-thickness graft.
Endothelial keratoplasty can be combined with cataract surgery and results in faster visual rehabilitation than can be expected from PK combined with cataract surgery. In a similar fashion, can endothelial keratoplasty be combined safely with IOL exchange to provide faster visual recovery than PK with IOL exchange? Will this more extensive surgery lead to greater graft complications such as dislocation, iatrogenic primary graft failure, and pupillary block than what is found with DSAEK alone?
Since September 2005, we have performed DSAEK with IOL exchange and have gathered outcome data in a prospective manner. For this study, we performed a retrospective analysis of that data and compared the complication rates of eyes that underwent DSAEK with IOL exchange with the complication rates of those eyes that were pseudophakic with a PC lens in place at the time of DSAEK surgery. In addition to the complication rates, a comparison of visual acuity and endothelial cell survival between the 2 groups also was performed.
Methods
The patients who underwent both DSAEK with IOL exchange and DSAEK alone are part of our ongoing prospective study of endothelial keratoplasty. All patients signed an 8-page, institutional review board-approved consent form that complied with the Health Insurance Portability and Accountability Act of 1996 before data collection. All surgeries were performed between September 2005 and September 2008.
For this analysis, patient records were reviewed to identify all eyes that underwent DSAEK surgery combined with IOL exchange where the AC IOL was removed and replaced with a PC IOL, either placed in the sulcus alone or sutured to the iris or sclera for additional support. The DSAEK with IOL exchange eyes were designated as the study group. The study group then was compared with an age- and time-matched comparison group of eyes undergoing DSAEK surgery alone in which the PC IOL was left in place. These eyes were designated as the comparison group. The acute complications of dislocations, primary graft failures, and cases of pupillary block were compared between the study and comparison groups. The outcomes of visual acuity and ECD were evaluated at 6 months after surgery for both groups and then compared.
Specular Microscopy Data
The vast majority of donor tissue came from the Lions Eye Bank of Oregon, which used an EB-3000 XYZ Eyebank specular microscope (HAI Laboratories, Inc., Lexington, Massachusetts, USA). These preoperative cell counts were obtained using an apices digitized method and the manufacturer’s calibrations for magnification. The apices of at least 100 cells from the endothelial images of each cornea were counted. Postoperative specular microscopy measurements of ECD (Konan SP4000 noncontact specular microscope; Konan Medical Corp, Fairlawn, New Jersey, USA) were performed in the authors’ clinic at the Devers Eye Institute at 6 months after surgery. A certified ophthalmic technician performed all postoperative testing of patients using the same specular microscope each time. These postoperative cell counts were obtained using the manufacturer’s calibrations for magnification and were counted with a fixed-frame method with the protocol requiring the marking of at least 50 to 100 cells for each image. Specular microscopy measurements with insufficient quality of the image were not included in the analysis. Analysis of endothelial polymorphism and polymegathism was not performed.
Surgical Technique
A simplified and standard technique of DSAEK surgery was used for all cases and has been well described in our previous publication. For the study group, the IOL exchange was performed at the same time as the DSAEK, with the IOL exchange procedure performed just minutes before the DSAEK. The AC IOL was removed through a 7-mm incision placed 1 mm posterior to the temporal limbus, and this incision also was used as the access incision for the insertion of the donor tissue. The replacement PC IOL was placed in the sulcus over intact capsular remnants, was sutured with 10-0 Prolene sutures (Ethicon, Inc., Somerville, New Jersey, USA) to the ciliary sulcus, or was sutured with 10-0 Prolene sutures to the iris. IOLs that were placed into the sulcus alone or sutured to the iris were Alcon 3-piece model MA50BM acrylic lenses (Alcon Laboratories, Fort Worth, Texas, USA), and IOLs sutured to the ciliary sulcus were Alcon model CZ70BD polymethyl methacrylate lenses (Alcon Laboratories). Anterior vitrectomy was performed as necessary to remove formed vitreous from the pupillary zone and AC. Donor tissue was inserted with Charlie insertion forceps (Bausch & Lomb Surgical, St Louis, Missouri, USA) in our usual fashion after completing all the steps of safe IOL exchange. In the 19 eyes of the study group, 11 PC IOLs were sutured with 10-0 Prolene sutures within the ciliary sulcus with knots buried under scleral flaps, 1 IOL had haptics sutured to the iris, and 7 of the IOLs were placed directly into the sulcus without sutures because adequate capsular support was present.
Statistical Analysis
Snellen BSCVA was converted to logarithm of the minimal angle of resolution (logMAR) units to allow averaging and statistical analysis. For normally distributed data, groups were compared using the Student t test and chi-square analysis. SPSS software version 12.0 (SPSS, Inc, Chicago, Illinois, USA) was used for all statistical testing.
Results
Patient Demographics
There were a total of 207 eyes that qualified for this study: 19 eyes in the DSAEK with IOL exchange group and 188 eyes in the comparison DSAEK only group. The patient demographics in both groups were similar, with a mean age ± standard deviation of 76.5 ± 10.8 years (range, 42 to 92 years) in the study group ( Table 1 ) and 75.6 ± 9.4 years (range, 41 to 96 years) in the comparison group. Indications for transplantation were similar in both groups as expected, but the proportions were different (see Table 2 ). In particular, the eyes in the DSAEK with IOL exchange group predominantly underwent endothelial keratoplasty for pseudophakic bullous keratopathy (17/19 eyes, or 89%), whereas most of those in the DSAEK only group underwent surgery secondary to underlying Fuchs endothelial dystrophy (120/188 eyes, or 64%).
| Patient No. | Gender | Age (yrs) | Eye | Indication | IOL Type | Preoperative BSCVA | 6–mo BSCVA | Preoperative ECD | 6–mo ECD |
|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 75 | L | PBK | Ciliary sutured | 20/100 | 20/200 | 2380 | a |
| 2 | F | 85 | L | PBK | Sulcus | 20/400 | 20/70 | 2659 | 2283 |
| 3 | F | 92 | R | PBK | Ciliary sutured | 20/100 | 20/50 | 2423 | 1461 |
| 4 | M | 67 | R | PBK | Ciliary sutured | 20/200 | 20/50 | 2709 | 1309 |
| 5 | F | 83 | R | Fuchs ED | Sulcus | 20/100 | 20/150 | 2315 | a |
| 6 | M | 78 | L | Fuchs ED | Sulcus | 20/30 | 20/25 | 2324 | 1927 |
| 7 | F | 84 | R | PBK | Ciliary sutured | CF 2′ | 20/70 | 3381 | a |
| 8 | M | 79 | L | PBK | Ciliary sutured | 20/70 | 20/80 | 3099 | 2037 |
| 9 | F | 82 | R | PBK | Ciliary sutured | 20/200 | 20/30 | 3162 | 1852 |
| 10 | F | 82 | L | PBK | Ciliary sutured | CF 2′ | 20/70 | 2711 | a |
| 11 | M | 84 | R | PBK | Ciliary sutured | 20/400 | 20/70 | 2792 | 1934 |
| 12 | M | 42 | R | PBK | Ciliary sutured | CF 2′ | 20/40 | 2540 | 1442 |
| 13 | F | 67 | L | PBK | Sulcus | 20/200 | 20/200 | 2968 | a |
| 14 | F | 70 | L | PBK | Ciliary sutured | 20/100 | 20/30 | 3101 | 1855 |
| 15 | M | 69 | L | PBK | Sulcus | 20/100 | 20/30 | 2529 | a |
| 16 | F | 83 | R | PBK | Sulcus | 20/50 | 20/30 | 2791 | 1754 |
| 17 | F | 83 | R | PBK | Iris sutured | 20/200 | 20/100 | 2562 | a |
| 18 | M | 73 | R | PBK | Ciliary sutured | 20/400 | 20/200 | 2543 | 1898 |
| 19 | M | 76 | R | PBK | Sulcus | 20/80 | 20/70 | 2652 | a |
| Mean | 76.5 | 20/205 | 20/48 | 2740 | 1796 |
| Indications for Transplantation | DSAEK with IOL Exchange | DSAEK |
|---|---|---|
| Fuchs endothelial dystrophy | 2 (11%) | 120 (64%) |
| PBK | 17 (89%) | 60 (32%) |
| Other (failed graft, ABK, etc.) | 0 (0%) | 8 (4%) |
| Total | 19 (100%) | 188 (100%) |
Visual Acuity
Preoperative mean BSCVA in the study and comparison groups were 20/202 (logMAR, 1.01 ± 0.53) and 20/125 (logMAR, 0.79 ± 0.59), respectively (see Table 3 ). For all of the 19 study eyes (100%), 6-month visual acuity measurements were obtained. For the 188 control eyes, 128 (68%) eyes were available for 6-month measurements. BSCVA improved significantly in both groups: at the 6-month period, the vision for the study group had improved to 20/73 (logMAR, 0.56 ± 0.32), and for the comparison group, vision had improved to 20/45 (logMAR, 0.33 ± 0.33). There was no statistically significant difference in BSCVA before surgery ( P = .15), but the vision was statistically worse in the study group at 6 months after surgery ( P = .01).
| Group | Preoperative BSCVA | 6–mo BSCVA |
|---|---|---|
| DSAEK with IOL exchange (n = 19) | 20/202 (n = 19) | 20/73 (n = 19) |
| DSAEK (n = 188) | 20/125 (n = 188) | 20/45 (n = 128) |
| P values | .15 | .01 |
Ocular comorbidities, including, but not limited to, age-related retinal disease, cystoid macular edema, and glaucoma, also were examined in both the study and comparison groups before surgery. There was a significantly higher percentage of the above-mentioned comorbidities in the study group (13/19 eyes, or 68%) compared with the DSAEK only group (49/188 eyes, or 26%; P = .01). Because of the disproportionate amount of ocular comorbidities in the study group, a subgroup analysis was performed on those patients without underlying ophthalmic pathologic features.
Six (32%) of 19 eyes in the study group and 139 (74%) of 188 eyes in the comparison group were identified as being free of any ocular comorbidities. Among those patients, preoperative mean BSCVA in the study and comparison groups were 20/205 (logMAR, 1.01 ± 0.62) and 20/100 (logMAR, 0.70 ± 0.56), respectively (see Table 4 ). Of the 6 study eyes, 6-month visual acuity measurements were obtained from all 6 (100%). Of the 139 control eyes, 90 (65%) were available for 6-month measurements. BSCVA improved significantly in both groups: at the 6-month period, the vision for the study group had improved to 20/48 (logMAR, 0.35 ± 0.20), and for the comparison group, vision improved to 20/34 (logMAR, 0.23 ± 0.13). There was no statistically significant difference in BSCVA before surgery ( P = .18), but the vision was statistically worse in the study group at 6 months after surgery ( P = .01).
| Group | Preoperative BSCVA | 6–mo BSCVA |
|---|---|---|
| DSAEK with IOL exchange (n = 6) | 20/205 (n = 6) | 20/48 (n = 6) |
| DSAEK (n = 139) | 20/100 (n = 139) | 20/34 (n = 90) |
| P values | .18 | .01 |
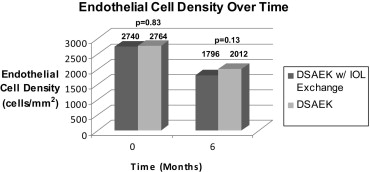
Donor Endothelial Cell Loss
The Figure and Table 5 summarize the analysis of endothelial cell loss over time. The mean preoperative ECD in eyes that underwent combined DSAEK with IOL exchange was 2740 ± 283 cells/mm 2 (range, 2315 to 3381 cells/mm 2 ) and was 2764 ± 353 cells/mm 2 (range, 2193 to 4209 cells/mm 2 ) in the comparison DSAEK only group ( P = .83). Of the 19 study patients, 11 (58%) had 6-month ECD readings available, and of the 188 control patients, 104 (55%) had 6-month ECD readings available.