Purpose
To compare the lamina cribrosa thickness, measured by swept-source optical coherence tomography (SS OCT), between each eye of normal tension glaucoma (NTG) patients with unilateral visual field (VF) defect and to investigate the correlation between lamina cribrosa thickness and VF loss.
Design
Prospective, cross-sectional study.
Methods
Optic nerve heads were scanned using SS OCT, and laminar thickness was measured on mid-superior, central, and mid-inferior regions of vertical midline of the optic disc. The inter-eye differences of lamina cribrosa thickness in NTG patients with unilateral VF defect and the intra-eye difference of lamina cribrosa thickness in VF-affected eyes were analyzed using the paired t test. We evaluated the correlation between lamina cribrosa thickness and mean deviation, measured using standard automated perimetry, in NTG patients.
Results
This study included 102 eyes in 51 NTG patients with unilateral VF defect and 47 eyes in 47 normal subjects without glaucomatous change in either eye. The mean lamina cribrosa thickness of normal fellow eyes was thicker than VF-affected eyes in NTG patients ( P < .001), but thinner than normal subject eyes ( P < .001). Within VF-affected eyes, lamina cribrosa thickness of regions correlated with visual field defect was thinner than horizontally contralateral locations ( P < .001). The mean deviation was statistically correlated with inter-eye difference of lamina cribrosa thickness in NTG patients (n = 51; r 2 = 0.12; P = .01).
Conclusions
The lamina cribrosa was thinner in VF-unaffected eyes of NTG patients than in normal subject eyes, in VF-affected eyes than in normal fellow eyes of NTG patients, and in regions correlated with visual field loss than in horizontally contralateral ones in VF-affected eyes.
Glaucoma is the most common cause of irreversible blindness worldwide. However, the pathophysiology of glaucomatous optic atrophy remains unclear. Many studies have confirmed that elevated intraocular pressure (IOP) is one of the most important risk factors for glaucoma development and progression. However, even when IOP is maintained within the normal range, glaucomatous optic neuropathy such as normal tension glaucoma (NTG) can develop. Conversely, ocular hypertension with a high IOP can have normal functional and structural configurations. Therefore, non-IOP-related risk factors such as biomechanical, autoimmune, or blood flow issues must be considered and studied, especially in NTG. In regard to biomechanical factors, the optic nerve head (ONH) is considered a weak region owing to discontinuity in the corneoscleral shell. This discontinuity concentrates the stress or strain on the optic nerve. Therefore, the ONH, especially the lamina cribrosa, has come into focus as a key pathophysiologic site in glaucomatous optic neuropathy.
The emergence and advancement of optical coherence tomography (OCT) has facilitated lamina cribrosa morphology and thickness evaluation. Studies performed using enhanced-depth imaging (EDI) OCT have shown that glaucoma patients have a thinner lamina cribrosa than glaucoma suspects or normal subjects, and that lamina cribrosa thickness is significantly correlated with the mean deviation (MD) in standard automated perimetry (SAP). However, there is little knowledge about the factors that could affect lamina cribrosa biomechanics and thickness, such as age or sex. Furthermore, biomechanical, vascular, and immunologic factors could contribute to glaucomatous optic neuropathy development. Human patients can have similar or identical blood flow, autoimmunity, cerebrospinal fluid (CSF) pressure, and extracellular matrix properties in both eyes. If 1 eye is diagnosed with NTG and the other has a normal configuration, many of the contributing factors would be controlled, and these patients could serve as an ideal model to assess the relationship between lamina cribrosa and NTG.
Recently, swept-source OCT (SS OCT) became commercially available, and this instrument uses a longer wavelength and a higher scanning speed compared with currently used OCT. SS OCT was suitable for evaluating deeper structures such as the choroid and lamina cribrosa. Therefore, we measured lamina cribrosa thickness using SS OCT in both eyes of NTG patients with unilateral visual field (VF) defect to test the following hypotheses. First, when compared intra-individually, the lamina cribrosa would be thinner in the VF-affected eyes than in the normal fellow eyes. Second, when analyzed by intra-eye comparison in VF-affected eyes, the lamina cribrosa would be thinner in locations corresponding to the VF defect than in horizontally contralateral locations.
Methods
This prospective, cross-sectional study was approved by the Institutional Review Board of the Samsung Medical Center. All participants provided informed consent, and the study was carried out in accordance with the tenets of the Declaration of Helsinki. Fifty-one NTG patients with unilateral VF defect and 47 normal subjects were included in this study. The subjects were evaluated using SS OCT between May 2013 and April 2014 in the Department of Ophthalmology, Samsung Medical Center, Sungkyunkwan University in Seoul, South Korea.
All participants underwent comprehensive ophthalmologic examinations including slit-lamp biomicroscopy, best-corrected visual acuity (BCVA), refractive error, central corneal thickness, and Goldmann applanation tonometry. An automated VF test was evaluated using the 30-2 Swedish interactive threshold algorithm standard program on a Humphrey 740 Visual Field Analyzer (HFA 30-2; Carl Zeiss Meditec, Dublin, California, USA). To identify glaucomatous ONH, retinal nerve fiber layer (RNFL) defect, and retinal pathology, a dilated stereoscopic examination of the optic disc and fundus and red-free fundus photography (TRC-50IX; Topcon Corp, Tokyo, Japan) were performed and reviewed for all participants. A single well-trained operator performed all SS OCT (DRI OCT-1 Atlantis; Topcon Corp) and Cirrus OCT (Carl Zeiss Meditec Inc) and screened all the OCT scans during imaging. Rescanning was performed if the image was determined to be poor quality or to have off-center locations on ONH, or if there were scans with missing data.
NTG with unilateral VF defect was defined as having 1 eye diagnosed with NTG, and the fellow eye showing no VF defect, glaucomatous ONH, or RNFL defect. Normal subjects were defined when they did not have any VF defect, glaucomatous optic disc, or significant eye disease in either eye. Randomly selected eyes in normal subjects were assigned as normal control group. NTG was diagnosed using the following criteria: glaucomatous ONH or RNFL defects with corresponding VF loss, open angle configuration on gonioscopy, and IOP recordings ≤21 mm Hg during the follow-up period. Glaucomatous ONH was defined as neural rim thinning, focal notching, excavation, and cup-to-disc ratio difference of greater than 0.2 determined by color stereoscopic photography. A localized RNFL defect was defined as a defect wider than the width of a major retinal vessel at 1 disc diameter distance from the edge of the disc, which widened into an arcuate or wedge shape and reached the edge of the disc as determined by red-free fundus photography. Glaucomatous VF defects were defined as the presence of a cluster of 3 or more contiguous non-edge points on the pattern deviation probability plot with a probability less than 5% and with at least 1 of these points having a probability less than 1%. This was confirmed on 2 consecutive tests. Test results were considered unreliable and excluded if the fixation loss was more than 30% or the false-positive or false-negative was greater than 33%. All examinations were evaluated and recorded by 2 experienced observers (Y.K., J.C.H.) in a masked fashion.
Exclusion criteria were as follows: (1) older than 80 years old or younger than 20 years old, (2) BCVA less than 20/40, (3) refractive error less than -6 diopters or more than +6 diopters, (4) history of intraocular surgery including laser treatment and refractive surgery, (5) media opacity that reduced image quality, (6) non-glaucoma conditions that affect VF, (7) glaucomatous VF defect in both eyes, or (8) an invisible lamina cribrosa posterior border in the scanned images.
Swept-Source Optical Coherence Tomography of the Optic Nerve Head
We used SS OCT with a 1050-nm wavelength and a 100 nm tuning range swept source to scan the ONH in each patient. The scans yielded an approximately 8 μm depth and a 20 μm transverse resolution. The tunable laser source allowed for quick scans through the range of relevant frequencies, and the longer wavelength could visualize deep fundus structures, including the choroid and lamina cribrosa. This technique provided high-quality images with constant signal strength of the posterior pole, even in eyes with cataracts that affected image quality because of light scattering. The instrument provided up to 100 000 A-scans per second and an invisible scanning line owing to the long wavelength, which could reduce an eye movement and provide more successful scans. The lamina cribrosa was captured using the 3-dimensional (3D) optic disc scan protocol that covered a 3 × 3-mm area with a depth of 2.6 mm centered on the ONH. Each dataset consisted of 256 cross-sectional B-scan images of 512 × 256 pixels. The total data acquisition time for a 3D optic disc scan was 0.9 seconds. An internal nasal fixation light was used to center the ONH in the rectangle imaging mode.
Measurement of Lamina Cribrosa Thickness
The lamina cribrosa thickness was defined as the distance between the anterior and posterior borders of the highly reflective region visible beneath the ONH in a cross-sectional B-scan SS OCT image. For identifying the anterior and posterior borders of the lamina cribrosa, horizontal SS OCT B-scans were assessed in Adobe Photoshop CS2 (version 9.0; Adobe Systems, Inc, San Jose, California, USA). A quantitative lamina cribrosa thickness measurement was performed on 3 lines including the mid-superior, central, and mid-inferior lines of the optic disc using the manual caliper tool in DRI OCT Viewer 9.01 ( Figure 1 ). Mid-superior and mid-inferior lines were identified as the horizontal line on the middle of the vertical line connecting the center to the margin of the optic disc. At each line, laminar thickness was measured at 3 locations along a perpendicular line from the reference line connecting the end of the Bruch membrane: the center of the reference line, 50 μm temporally, and 100 μm temporally. Laminar thickness was the average of those 3 measurements. In this study, beta, “β,” was defined as the average laminar thickness measured on the vertical center of the optic disc. Alpha, “α,” was defined as the average laminar thickness measured at the halfway point of a vertical line connecting the optic disc center to the margin in the direction of the VF defect in VF-affected eyes or the corresponding location in fellow eyes. Gamma, “γ,” was defined as the average laminar thickness measured at points horizontally contralateral to “α.” For evaluating the inter-observer reproducibility of our measurements, lamia cribrosa thickness from 30 randomly selected SS OCT cross-sectional B-scans was independently measured by both observers (Y.K., J.C.H.) without clinical information.
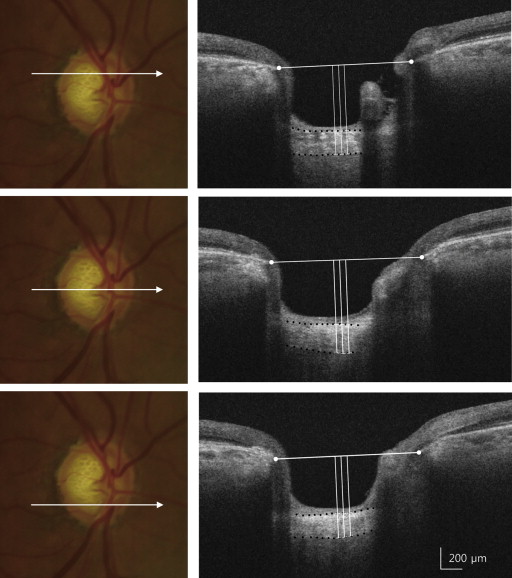
Statistical Analysis
Statistical analyses were performed using SPSS (version 21.0; SPSS Inc, Chicago, Illinois, USA). A paired t test was used to compare related data values measured from both eyes of NTG patients. In cases of multiple comparisons, Bonferroni correction was applied to control the family-wise error rate. The inter-observer reproducibility of lamina cribrosa thickness measurements was evaluated by calculating the intraclass correlation coefficient (ICC). We used the t test to compare the means of data from VF-unaffected NTG patient eyes and normal control eyes. A linear regression analysis was performed to evaluate the statistical correlation between MD measured by SAP and lamina cribrosa thickness. P values less than .05 were considered statistically significant.
Results
The optic disc OCT images were collected for 180 eyes from 120 patients (120 eyes in 60 NTG patients and 60 eyes in 60 normal subjects). In 11 of the eyes, vascular shadowing prevented clear lamina cribrosa structure imaging. In 21 of the eyes, the posterior borders of the highly reflective region beneath the optic disc cup were unresolvable. Images from these eyes were excluded from analyses. If 1 eye of an NTG patient was excluded then data from both eyes were excluded from the analysis. The remaining 149 eyes in 98 subjects were evaluated and analyzed. The NTG group included 102 eyes in 51 patients who had been treated with IOP-lowering eye drops for VF-affected eyes. The average age of the NTG group was 56.2 ± 11.3 years (range: 27–79 years; 30 men and 21 women). The normal control group included 47 eyes in 47 subjects with an average age of 57.4 ± 12.1 years (range: 23–80 years; 23 men and 24 women). There was no statistically significant difference in age or sex between the 2 groups ( Table 1 ). Within the NTG group, there was no difference in central corneal thickness ( P = .661), refractive error ( P = .124), or intraocular pressure at baseline ( P = .093) between VF-affected and fellow eyes. Mean lamina cribrosa thickness was 178.9 ± 28.3 μm in VF-affected eyes and 196.4 ± 28.6 μm in fellow eyes; therefore, lamina cribrosa in VF-affected eyes were significantly thinner than in fellow eyes ( P < .001). The mean lamina cribrosa thickness for the normal control group was 219.5 ± 28.4 μm, which was significantly thicker than the VF-unaffected eyes of the NTG group ( P < .001). The lamina cribrosa thickness measurement reproducibility was excellent (ICC = 0.831, 95% confidence interval, 0.812–0.847).
| NTG (n = 51) | Controls (n = 47) | P Value b | |||
|---|---|---|---|---|---|
| Affected Eyes | Unaffected Eyes | P Value a | |||
| Age (y) | 56.2 ± 11.3 | 57.4 ± 12.1 | .605 | ||
| Male/female | 30/21 | 23/24 | .417 | ||
| Central corneal thickness (μm) | 519.6 ± 36.3 | 518.7 ± 35.3 | .661 | 536.0 ± 31.0 | .013 |
| Refractive error (diopters) | −1.3 ± 2.3 | −1.2 ± 2.1 | .124 | −1.2 ± 2.2 | .975 |
| Intraocular pressure (mm Hg) | |||||
| Initial visit | 16.4 ± 2.9 | 15.9 ± 2.7 | .093 | 17.1 ± 3.1 | .059 |
| At scanning disc | 14.0 ± 2.3 | 14.3 ± 2.7 | .274 | 16.7 ± 3.1 | <.001 |
| Automated perimetry (dB) | |||||
| MD | −5.7 ± 5.3 | −0.1 ± 1.5 | <.001 | −0.1 ± 1.6 | .884 |
| PSD | 10.8 ± 12.0 | 2.0 ± 0.7 | <.001 | 2.0 ± 0.6 | .635 |
| RNFL average thickness (μm) | 71.5 ± 11.3 | 84.6 ± 10.4 | <.001 | 87.9 ± 10.4 | .174 |
| Mean LCT (μm) | 178.9 ± 28.3 | 196.4 ± 28.6 | <.001 | 219.5 ± 28.4 | <.001 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


