Purpose
To compare 3-year endothelial cell loss and graft survival following Descemet stripping automated endothelial keratoplasty (DSAEK) using the EndoGlide (AngioTech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK) donor insertion device compared to donor insertion using the Sheets glide technique.
Design
Retrospective comparative case series.
Methods
Study involved consecutive patients who underwent DSAEK with Fuchs endothelial dystrophy or pseudophakic bullous keratopathy at a single tertiary center. Clinical data with outcomes and donor and recipient characteristics were obtained from our ongoing prospective cohort from the Singapore Corneal Transplant Study. Main outcome measures were percent endothelial cell loss and graft survival up to 3 years.
Results
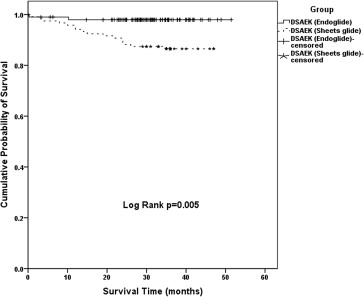
Overall percent endothelial cell loss was significantly lower in the EndoGlide group (100 eyes) compared to the Sheets glide group (119 eyes) at 1 year (16.3% ± 16.6% vs 29.5% ± 22.2%, P < .001), 2 years (23.8% ± 17.8% vs 35.7% ± 22.9%, P = .001), and 3 years (29.7% ± 20.9% vs 38.5% ± 24.1%, P = .015) postoperatively. Overall graft survival was greater in the EndoGlide compared to Sheets glide group up to 3 years postoperatively (97.9% vs 86.5%, log-rank P value = .005). In eyes with Fuchs endothelial dystrophy, endothelial cell loss was significantly lower in the EndoGlide group (3-year: 28.2% ± 17.9% vs 43.4% ± 27.1%, P = .032). In eyes with pseudophakic bullous keratopathy, the EndoGlide group had a superior graft survival compared to Sheets glide (log-rank P = .031).
Conclusion
Endothelial cell loss was lower using a donor insertion device during DSAEK, compared to using the Sheets glide technique for DSAEK, in Asian eyes with Fuchs endothelial dystrophy, and resulted in better graft survival in eyes with pseudophakic bullous keratopathy.
Descemet stripping automated endothelial keratoplasty (DSAEK) is progressively becoming the surgery of choice for endothelial diseases such as Fuchs endothelial dystrophy and pseudophakic bullous keratopathy. However, early studies reveal significant donor endothelial cell trauma from surgical manipulation, leading to a high primary (iatrogenic) graft failure and a greater initial endothelial cell loss (up to 6 months) postoperatively. Longer-term studies have, however, suggested that the rate of this endothelial cell loss decreases after the initial period to plateau and stabilize up to 5 years after DSAEK. Most studies (albeit with limited follow-up) suggest that the average endothelial cell loss is around 37% (range 25%-54%) at 6 months and 41% (range 24%-61%) at 1 year—with an average graft survival around 94% at 1 year.
Recent efforts have focused on further reducing endothelial cell damage associated with surgical manipulation upon insertion of the donor during DSAEK. The evolution of techniques from forceps insertion to pull/push-through techniques has led to an apparent reduction in postoperative endothelial cell loss over the years ( Supplementary Table , available at AJO.com ). Although the technique of donor insertion appears to be a major factor, other factors that have been found to contribute to greater endothelial cell loss in DSAEK include glaucoma, longer or smaller incisions, and donor dislocation. We had previously described a pull-through technique using a trimmed anterior chamber intraocular lens (ACIOL) Sheets glide, with relatively good graft survival results and reduced endothelial cell loss. Although donor insertion using folding techniques reported good results in eyes with deeper anterior chambers, we observed that the Sheets glide technique was more useful in Asian eyes with shallow anterior chambers and high vitreous pressures. This technique evolved after our initial experience with the Busin glide device, an open-ended device that often resulted in chamber collapse or iris prolapse when inserted into the wound; the Sheets glide essentially prevented iris prolapse while enabling an anterior chamber (AC) maintainer to be used to prevent chamber collapse. A major advantage of this technique is the relatively low cost of the Sheets glide.
The observations from our early studies using the Sheets glide led to the development of the US Food & Drug Administration–approved EndoGlide (AngioTech, Reading, Pennsylvania, USA/Network Medical Products, North Yorkshire, UK). The flat anterior surface (akin to the Sheets glide), modified to have a complete wound seal, protects the donor, prevents iris prolapse, and maintains the anterior chamber using a sealed back plate. This design was especially useful in Asian eyes with shallow anterior chambers and high vitreous pressures, or even in complex eyes with previous glaucoma filtration surgeries or floppy irises, or to hold down an ACIOL. Our preliminary studies found endothelial cell loss of 13.5% at 6 months and 14.9% at 12 months, with a low rate of postoperative complications in uncomplicated eyes. Here, we report and compare longer-term (3-year) graft survival and endothelial cell loss between the EndoGlide inserter and our initial Sheets glide technique.
Materials and Methods
In this study, we reviewed consecutive patients who underwent DSAEK at a single tertiary referral center (Singapore National Eye Centre) from April 1, 2006, to June 1, 2010, whose surgical indication was either Fuchs endothelial dystrophy or pseudophakic bullous keratopathy (PBK). Our study followed the principles of the Declaration of Helsinki, with ethics approval obtained from the Singapore Health Services Centralized Institutional Review Board. In this study, we excluded eyes that underwent DSAEK in the presence of any concomitant anterior segment pathology such as anterior chamber intraocular lenses, primary angle-closure glaucoma, or previous intraocular surgery such as trabeculectomy or glaucoma drainage devices, as well as those with complicated cataract surgery with secondary anterior chamber abnormalities. We obtained all clinical data from our ongoing Singapore Corneal Transplant Study, a prospective cohort study that tracks and records all clinical data, surgical complications, and outcomes in patients who undergo corneal transplants at the Singapore National Eye Centre. The Singapore Eye Bank provides all donor corneas with standard guidelines for DSAEK grafts and we obtained all donor information from their database, including donor endothelial cell density. Specular microscopy of the donor tissue was performed preoperatively by a certified eye bank technician at the Singapore Eye Bank and, using a noncontact specular microscope (Konan Medical Corp, Hyogo, Japan), at 1, 2, and 3 years postoperatively by masked trained ophthalmic technicians, ensuring good image quality and accurate cell counts, as previously described. Our main outcome measure was graft survival and percent endothelial cell loss up to 3 years postoperatively.
Surgical Technique
All DSAEK surgeries were performed using either a Sheets glide or the EndoGlide technique (1-year noncomparative results previously reported). All surgeries were performed by 5 consultant corneal surgeons with similar surgical experience at our center, which included cases performed or partially performed by corneal fellows in training under direct supervision. Donors were prepared using an automated lamellar therapeutic keratoplasty system (ALTK; Moria SA, Antony, France). In summary, after insertion of AC maintainer, Descemet membrane stripping, inferior peripheral iridectomy, and preplaced venting incisions, EndoGlide insertion forceps (ASICO, Westmont, Illinois, USA) were used to pull the donor cornea through a 5-mm scleral tunnel incision over a trimmed Sheets glide (BD Visitec, Waltham, USA) or via the EndoGlide through a 4.5-mm scleral tunnel incision. The main wound and the AC maintainer wound were secured with 10/0 nylon interrupted sutures, and a full air tamponade was performed for at least 6 minutes. Following this, some air was removed, leaving a smaller air bubble approximating the size of the DSAEK graft in the AC, and a bandage contact lens was placed.
Postoperative Care
All patients received a standard postoperative regime: prednisolone acetate 1% (Pred Forte; Allergan, Inc, Irvine, California, USA) or dexamethasone sodium phosphate 0.1% (Minims; Bausch & Lomb, Inc, Rochester, New York, USA), and a topical antibiotic. Eyedrops were prescribed to be applied every 3 hours for 1 month, then 4 times daily for 2 months, then were tapered by 1 drop per 3 months down to a dose of 1 drop per day by 1 year, and thereafter were continued indefinitely.
Statistical Analysis
Statistical analysis included descriptive statistics, where the mean and standard deviation (SD) were calculated for the continuous variables, while frequency distribution and percentages were used for categorical variables. Comparisons between categorical variables were conducted by χ 2 test/Fisher exact tests, whereas for continuous variables independent t test and Mann-Whitney U tests were conducted. The trend of endothelial cell density with 95% confidence intervals (95% CI) was evaluated over 3 years of follow-up using a linear mixed-model analysis. Kaplan-Meier survival analysis was conducted to determine survival probabilities of EndoGlide and Sheets glide DSAEK groups. The survival period of failed grafts was defined as the time between the date of surgery and recorded date of graft failure, defined as irreversible loss of optical clarity, sufficient to compromise vision for a minimum of 3 consecutive months. Statistical Package for the Social Sciences version 17.0 (SPSS Inc, Chicago, Illinois, USA) was used to analyze the data. A P value <.05 has been considered statistically significant.
Results
During the study period, we included 219 eyes of 219 patients that underwent DSAEK using either EndoGlide (100 eyes) or Sheets glide (119 eyes) insertion technique. The overall mean age of our study cohort, of which more patients were female (118 patients, 53.9%) and predominantly Chinese in our Asian cohort (157 patients, 71.7%), was 67 ± 11 years ( Table 1 ). There were more eyes with PBK (78 eyes, 65.5%) in the Sheets glide group compared to the EndoGlide group (43 eyes, 41.0%, P < .001). However, there were no significant differences in demographics or characteristics comparing Fuchs endothelial dystrophy or PBK within the EndoGlide or Sheets glide DSAEK groups.
| Characteristics | Total (n = 219) | DSAEK | P Value | |
|---|---|---|---|---|
| EndoGlide (n = 100) | Sheets Glide (n = 119) | |||
| Mean age, y (±SD) | 67.4 (10.5) | 69.5 (9.1) | 65.5 (11.3) | .005 |
| Sex (%) | ||||
| Male | 101 | 43 (43) | 58 (48.7) | .396 |
| Female | 118 | 57 (57) | 61 (51.3) | |
| Race | .680 | |||
| Chinese | 157 | 69 | 88 | |
| Malay | 13 | 8 | 5 | |
| Indian | 8 | 4 | 4 | |
| Other | 31 | 19 | 12 | |
| Surgical indication | ||||
| Fuchs endothelial dystrophy | 98 | 57 | 41 | .001 |
| Pseudophakic bullous keratopathy | 121 | 43 | 78 | |
| Surgery | ||||
| DSAEK alone | 132 | 52 | 82 | <.001 |
| DSAEK + phacoemulsification/IOL | 75 | 48 | 27 | |
| Donor characteristics | ||||
| Donor age (y), median (range) | 56 (17-79) | 56 (17-75) | 57 (18-79) | .438 |
| Donor graft diameter (mm), mean (SD) | 8.7 (0.4) | 8.7 (0.3) | 8.6 (0.5) | .474 |
Endothelial Cell Density
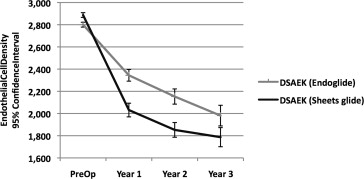
We compared postoperative percent endothelial cell loss between EndoGlide and Sheets glide DSAEK at 1, 2, and 3 years and found that percent endothelial cell loss was significantly lower in the EndoGlide group at each time point of comparison ( Table 2 ). Overall percent endothelial cell loss was significantly lower in the EndoGlide group compared to Sheets glide at 1 year (16.3% ± 16.6% vs 29.5% ± 22.2%, P < .001), 2 years (23.8% ± 17.8% vs 35.7% ± 22.9%, P < .001), and 3 years postoperatively (29.7% ± 10.9% vs 38.5% ± 24.1%, P = .015) ( Figure 1 ). Of note, the endothelial cell loss was also significantly lower in the EndoGlide compared to Sheets glide group up to 3 years postoperatively for the Fuchs endothelial dystrophy subgroup (28.2% ± 17.9% vs 39.5% ± 23.5%, P = .015). In patients with PBK, the EndoGlide group had significantly lower endothelial cell loss at 1 year postoperatively (17.9% ± 16.7% vs 28.4% ± 22.2%, P = .023) but not subsequently after 1 year ( Table 2 ).
| DSAEK (EndoGlide) | DSAEK (Sheets Glide) | P Value | |||
|---|---|---|---|---|---|
| Mean (SD) | Eyes a | Mean (SD) | Eyes a | ||
| Endothelial cell density (cells/mm 2 ) | |||||
| Baseline | 2800 (216) | 99 | 2887 (225) | 113 | .004 |
| 1 year | 2344 (480) | 83 | 2031 (652) | 112 | .001 |
| 2 years | 2153 (526) | 59 | 1852 (662) | 100 | .004 |
| 3 years | 1981 (602) | 42 | 1786 (720) | 70 | .105 |
| Overall | |||||
| Endothelial cell loss (%) | |||||
| 1 year | 16.3 (16.6) | 83 | 29.5 (22.2) | 106 | <.001 |
| 2 years | 23.8 (17.8) | 58 | 35.7 (22.9) | 94 | .001 |
| 3 years | 29.7 (20.9) | 42 | 38.5 (24.1) | 65 | .015 |
| Fuchs endothelial dystrophy | |||||
| Endothelial cell loss (%) | |||||
| 1 year | 15.2 (16.6) | 49 | 31.5 (24.2) | 37 | .002 |
| 2 years | 23.3 (17.3) | 38 | 36.1 (22.5) | 34 | .009 |
| 3 years | 28.2 (17.9) | 24 | 39.5 (23.5) | 28 | .015 |
| Pseudophakic bullous keratopathy | |||||
| Endothelial cell loss (%) | |||||
| 1 year | 17.9 (16.7) | 34 | 28.4 (22.2) | 69 | .023 |
| 2 years | 24.7 (18.9) | 20 | 35.5 (23.3) | 60 | .052 |
| 3 years | 31.5 (24.7) | 18 | 37.8 (24.9) | 37 | .351 |
a Number of eyes calculated for endothelial cell loss are only from eyes with available endothelial cell density values at baseline.
Graft Success
Overall graft survival was greater in the EndoGlide compared to Sheets glide group (cumulative graft survival at 3 years: 97.9% vs 86.5%, respectively; log-rank P value =.005) ( Figure 2 ). There was 1 case of primary graft failure in each of the EndoGlide (1.0%) and Sheets glide (0.8%) DSAEK groups. In the EndoGlide DSAEK group, 1 case of endothelial rejection at 6 months led to eventual graft failure at 14 months. Overall, remaining causes of graft failure in DSAEK were late endothelial decompensation, attributable to immunologic graft rejection (n = 4), and nonimmunologic reasons such as glaucoma or glaucoma surgery (n = 5), herpetic endotheliitis (n = 3), or late endothelial failure (n = 6). Subgroup analysis revealed a significant superior graft survival comparing EndoGlide DSAEK vs Sheets glide DSAEK in the PBK group (log-rank P = .031, Figure 3 ) but not in the Fuchs endothelial dystrophy group ( P = .303, Figure 4 ).