Purpose
To examine bevacizumab and ranibizumab utilization and disease monitoring patterns in patients with neovascular age-related macular degeneration (neovascular AMD) in clinical practice.
Design
Retrospective medical claims analysis.
Methods
Patients receiving ≥1 ranibizumab or bevacizumab injection during the 12 months after initial neovascular AMD diagnosis were included. Annual bevacizumab and/or ranibizumab injection utilization was assessed by year of first injection cohorts: 2006 and 2007 (received either agent because of billing code overlap), 2008, 2009, and January-June 2010 (received each agent). Outcome measures were time to first injection relative to neovascular AMD diagnosis and mean numbers of intravitreal injections, ophthalmologist visits, and optical coherence tomography (OCT) and fluorescein angiography (FA) examinations in 12 months.
Results
In the 2006 and 2007 cohorts (n = 8767), mean annual numbers of bevacizumab or ranibizumab injections were 4.7 and 5.0, respectively. Over 92% of patients in all cohorts received first treatment within 3 months of neovascular AMD diagnosis. In the 2008-2010 cohorts (n = 10 259), mean annual number of injections remained low (bevacizumab: 4.6, 5.1, and 5.5; ranibizumab: 6.1, 6.6, and 6.9), as did mean numbers of ophthalmologist visits (bevacizumab only) and OCT examinations (both agents), but there was no such trend in FA examinations.
Conclusions
Compared with treatment paradigms validated by clinical trials published at the time, in clinical practice, patients with neovascular AMD received fewer bevacizumab or ranibizumab injections and less-frequent monitoring from 2006 to mid-2011. Factors contributing to this lower injection frequency and visual outcomes associated with reduced utilization need to be researched.
Age-related macular degeneration is the leading cause of blindness among Americans over 40 years of age and irreversible vision loss in those over 65 years of age in the United States. Neovascular age-related macular degeneration (neovascular AMD) is one of several ocular diseases that may result in pathologic neovascularization leading to significant vision loss. Overexpression of vascular endothelial growth factor (VEGF) is believed to play an essential role in the pathogenesis of neovascular AMD.
The treatment of neovascular AMD has been revolutionized by the development of anti-VEGF agents. Anti-VEGF agents are widely considered the standard of care for neovascular AMD and are the most promising way of inhibiting the neovascular process and improving a patient’s visual prognosis. Until recently, 2 anti-VEGF agents were approved in the United States for neovascular AMD: pegaptanib (Macugen; Eyetech, Inc., Cedar Knolls, New Jersey, USA) and ranibizumab (Lucentis; Genentech, San Francisco, California, USA). A third agent, aflibercept (Eylea; Regeneron Pharmaceuticals, Inc., Tarrytown, New York, USA), was approved in November 2011. A fourth agent, bevacizumab (Avastin; Genentech), is not approved for intraocular use but is frequently used off label for this indication. Before the approval of aflibercept, bevacizumab and ranibizumab were the most widely used agents because of their perceived superior efficacy compared with pegaptanib. Several randomized controlled trials (RCTs) have demonstrated that with monthly monitoring and frequent injections, bevacizumab and ranibizumab have similar efficacy in improving best-corrected visual acuity (BCVA) in patients with neovascular AMD.
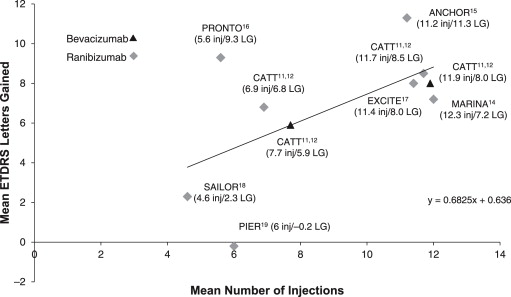
Large prospective clinical trials suggest that monthly monitoring with frequent anti-VEGF injections maximizes the likelihood for visual improvement ( Figure 1 ). Pivotal phase 3 trials (MARINA [Minimally Classic/Occult Trial of the Anti-VEGF Antibody Ranibizumab in the Treatment of Neovascular AMD] and ANCHOR [Anti-VEGF Antibody for the Treatment of Predominantly Classic Choroidal Neovascularization in AMD] ) that used monthly ranibizumab injections and monitoring visits reported 7.2 and 11.3 Early Treatment Diabetic Retinopathy Study (ETDRS) letter gains overall at 12 months, respectively. Studies that used less-frequent anti-VEGF injections (eg, PIER [Phase IIIb, Multicenter, Randomized, Double-Masked, Sham Injection–Controlled Study of the Efficacy and Safety of Ranibizumab] and EXCITE [Efficacy and Safety of Ranibizumab in Patients with Subfoveal CNV Secondary to Age-Related Macular Degeneration] ) have shown less improvement with fewer letters gained. The positive association between anti-VEGF injection frequency and visual outcome was supported by the recently published 2-year CATT (Comparisons of Age-Related Macular Degeneration Treatment Trials) study, in which patients randomly reassigned from monthly to “as-needed” (ie, PRN) administration frequency after the first year lost vision (−1.8 letters in ranibizumab group and −3.6 letters in bevacizumab group; P = .03) compared with those who continued on the monthly regimen despite being monitored monthly for re-treatment. Two other recently published studies, the IVAN (Inhibition of VEGF in Age-related Choroidal Neovascularization) and HARBOR (pHase III, double-masked, multicenter, randomized, Active treatment-controlled study of the efficacy and safety of 0.5 mg and 2.0 mg Ranibizumab administered monthly or on an as-needed Basis in patients with subfoveal neOvasculaR age-related macular degeneration) studies, both of which used different PRN regimens from the CATT study, also found smaller visual improvement in patients treated with PRN regimens compared with those with monthly injections, although the differences were small (<3 ETDRS letters) and not statistically significant. Thus, during the time period of July 2005 to June 2011 (the time of the current analysis), the overall clinical trial evidence indicated that patients required monthly monitoring and frequent anti-VEGF injections for optimal visual outcome.
Relatively little is known about the patterns of anti-VEGF agent utilization in neovascular AMD in clinical practice, and the extent to which they may differ from the monthly injections and monitoring suggested by pivotal RCT studies. One retrospective pooled analysis of 4 European ranibizumab registry studies of 4444 patients with neovascular AMD found a substantially lower range of annual injections (4.3-5.7), and fewer letter gains in BCVA outcomes (0.8-5.6 ETDRS letter gains) in the first 12 months of treatment compared with pivotal RCTs (Bandello F, et al. IOVS 2012;53:ARVO E-Abstract 2031). A smaller comparative, retrospective case series from a single integrated healthcare system in the United States also reported similar and relatively low levels of bevacizumab and ranibizumab utilization, with 4.4 and 6.2 injections, respectively, in the first 12 months.
The paucity of data on clinical utilization patterns of anti-VEGFs in the United States provided the motivation for this analysis. This study examined the intravitreal injection frequency patterns of anti-VEGF agents, specifically focusing on bevacizumab and ranibizumab, in patients with neovascular AMD using one of the largest fully integrated claims databases in the United States. The population of interest in this study included both patients enrolled in managed care systems and those enrolled in fee-for-service health plans. Additionally, this study assessed disease monitoring patterns, such as ophthalmologist visits and diagnostic test utilization.
Methods
Data Source
This retrospective cohort claims analysis used commercial and Medicare supplemental claims from July 1, 2005 to June 30, 2011 from the Truven Health Analytics MarketScan Research Database. MarketScan contains fully integrated, standardized, de-identified, patient-level claims from diverse data sources, including employers, health plans, and government agencies across all 50 states. In 2009, MarketScan included 43.1 million unique patients (39.9 million commercial lives and 3.2 million Medicare lives with employer-sponsored supplemental insurance). No personal identifying information was available in the database. This study did not require human subjects review and did not require institutional review board approval.
Inclusion/Exclusion Criteria
To ensure the inclusion of newly diagnosed and anti-VEGF treatment–naïve patients, the study sample consisted of patients who: (1) had an initial diagnosis of neovascular AMD (International Classification of Diseases, Version 9 [ICD-9] codes of 362.42, 362.43, or 362.52; or 362.50 listed on the same claim with a bevacizumab or ranibizumab intravitreal injection) in at least 2 outpatient visits separated by at least 14 days, or 1 emergency department/inpatient admission in 2006 or later; (2) received the first bevacizumab or ranibizumab injection listed on the same claim with neovascular AMD diagnosis, or an intravitreal injection (Current Procedural Terminology [CPT] code 67028) on the same date between January 1, 2006 and June 30, 2010 and within 12 months of the initial neovascular AMD diagnosis; (3) did not receive bevacizumab or ranibizumab injections for at least 6 months before the initial diagnosis; (4) were 18 years of age or older at the time of the index date (defined as the date of first injection of bevacizumab or ranibizumab); and (5) were continuously enrolled from 6 months before the initial diagnosis date (to confirm no prior neovascular AMD diagnoses) through 12 months after the first anti-VEGF injection. Overall, records from 19 027 patients treated in clinical practice were analyzed.
Patients were assigned to an index cohort year according to their index date (first anti-VEGF injection). Utilization pattern was examined by cohort year (2006-2010 [January-June]). In the 2006 and 2007 cohorts, utilization of bevacizumab and ranibizumab was combined because of shared Healthcare Common Procedure Coding System (HCPCS) J codes prior to 2008. For 2008 and later cohorts, patients were further indexed based on their initial type of anti-VEGF treatment received (bevacizumab or ranibizumab); the utilization pattern was analyzed separately using treatment-specific J codes. To minimize heterogeneity and maximize internal validity, the analysis excluded patients with diagnoses of multiple types of retinal disease; specifically, AMD plus central retinal vein occlusion (ICD-9 code 362.35), branch retinal vein occlusion (ICD-9 code 362.36), or diabetic macular edema (ICD-9 code 362.07 [prior to 2007] or 362.83 or 362.53, and a diagnosis of diabetes 250.xx reported within the preceding 365 days) during the study period. For the 2008-2010 (January-June) cohorts, patients who were treated with both bevacizumab and ranibizumab were also excluded.
Outcome Measures
The primary outcomes measuring anti-VEGF utilization were the mean and median numbers of bevacizumab and ranibizumab injections within 12 months after and including the index date. Claims databases typically do not provide a reliable identifier of which eye receives an intravitreal injection. This analysis therefore estimated the number of injections per patient, which is always equal to or greater than the number of injections per treated eye.
Additional utilization measures included the percentage of patients with 10 or more injections in the 12-month observation period (an approximation of monthly injections), the percentage of patients receiving their first bevacizumab or ranibizumab treatment within 90 days of initial diagnosis, and the percentage receiving 2 or more additional injections within 3 months after the first injection (an approximation of full receipt of 3 “monthly” loading doses). Disease management patterns related to patient monitoring, including mean numbers of ophthalmologist visits, optical coherence tomography (OCT) examinations, and fluorescein angiography (FA) examinations, also were assessed for the 12 months of observation.
Anti–Vascular Endothelial Growth Factor and Healthcare Resource Utilization Identification
Anti-VEGF injections were identified using HCPCS codes J3490 (since 2008), J3590 (since 2008), J9035, Q2024 (fourth quarter of 2009), and C9257 (since 2010) for bevacizumab and J2778 (since 2008) and C9233 (2007) for ranibizumab. Prior to 2008, J3490 and J3590 were used for both bevacizumab and ranibizumab. All bevacizumab and ranibizumab injections were required to have an intravitreal injection CPT code of 67028 on the same date or a neovascular AMD diagnosis code on the same claim. The number of injections per patient was capped at a maximum of 2 per day to control for duplicate claims.
Office visits were defined as the number of days the patient had at least 1 outpatient claim, as claims databases (including MarketScan) do not have an identifier for each unique office visit. Ophthalmologist visits were broadly defined as the number of days the patient had 1 or more claims for the index diagnosis with the “provider specialty” listed as “ophthalmologist” or had an OCT examination or an intravitreal injection. This definition thus likely represents an upper bound estimate of the number of monitoring visits. OCT examinations (capped at a maximum of 2 per patient per day) were identified by CPT codes 92135 (effective through 2010) or 92134 (effective after January 1, 2011). FA examinations were identified in a similar manner using the CPT code 92235.
Results
Sample Characteristics
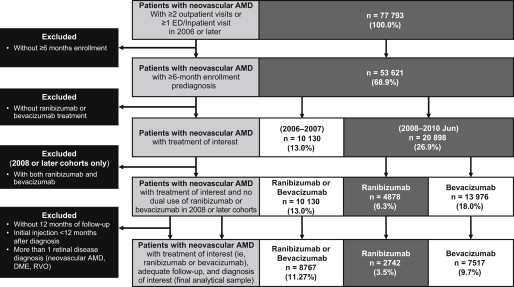
Of the 77 793 patients with a diagnosis of neovascular AMD for at least 2 outpatient visits, or 1 emergency department/inpatient admission in 2006 or later, a total of 53 621 met the 6-month or greater prediagnosis enrollment criteria ( Figure 2 ). Among these patients, 31 028 had at least 1 bevacizumab or ranibizumab injection. Of the 10 130 patients from the 2006 and 2007 cohorts, 8767 met the remaining inclusion and exclusion criteria for these 2 cohorts. Of the 20 898 patients from the 2008 or later cohorts, 7517 bevacizumab patients and 2742 ranibizumab patients met the remaining inclusion/exclusion criteria for these cohorts. Mean ages ranged from 77.0 to 78.7 years across the cohorts, with 40.3%-41.9% being male ( Table 1 ). Between 85.8% and 91.0% of patients in the cohorts were Medicare patients with employer-sponsored supplemental insurance.
| Bevacizumab | Ranibizumab | |
|---|---|---|
| Number of patients by year of index date a | ||
| 2006-2007 (total) b | 8767 | |
| 2006 | 3693 | |
| 2007 | 5074 | |
| Mean age (y) | 78.2 | |
| Male (%) | 40.5 | |
| Medicare (%) | 91.0 | |
| Number of patients by year of index date a | ||
| 2008-2010 (total) | 7517 | 2742 |
| 2008 | 3086 | 1181 |
| 2009 | 3043 | 1065 |
| 2010 (January-June) | 1388 | 496 |
| Mean age (y) | 77.0 | 78.7 |
| Male (%) | 41.9 | 40.3 |
| Medicare (%) | 85.8 | 90.7 |
a Index date defined as the date of first observed intravitreal injection of bevacizumab or ranibizumab.
b Because of shared Healthcare Common Procedure Coding System codes during 2006-2007, analyses combined patients treated with bevacizumab and/or ranibizumab.
Anti–Vascular Endothelial Growth Factor Utilization Patterns
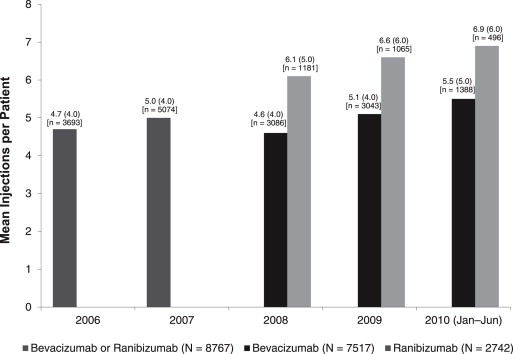
In the 2006 and 2007 cohorts, the mean (median) annual numbers of anti-VEGF (bevacizumab or ranibizumab) injections were 4.7 (4.0) and 5.0 (4.0), respectively. In the 2008-2010 (January-June) cohorts, the mean (median) annual number of bevacizumab injections increased from 4.6 (4.0) to 5.5 (5.0), while mean (median) annual ranibizumab injections grew from 6.1 (5.0) to 6.9 (6.0) ( Figure 3 ).
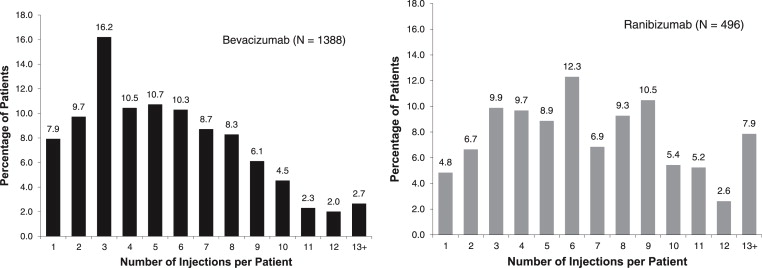
In the 2008-2010 (January-June) cohorts, the proportion of patients who received 10 or more injections (including the first injection) in the 12 months after their first injection ranged from 7.0% to 11.5% and from 18.0% to 21.2% among bevacizumab- and ranibizumab-treated patients, respectively. In the most recent (2010 [January-June]) cohort, less than 12% of the bevacizumab-treated patients received 10 or more injections in the 12 months after their first treatment, and less than 22% of those in the ranibizumab treatment group attained this threshold ( Figure 4 ).