Purpose
To investigate the outcomes of Descemet membrane endothelial keratoplasty (DMEK) using prestripped donor tissue prepared by an eye bank.
Design
Retrospective, noncomparative case series.
Methods
This retrospective, noncomparative, observational study investigated the outcomes of the first 40 consecutive DMEK procedures performed by a single surgeon using prestripped tissues prepared by a single eye bank during the period September 17, 2013 to July 1, 2014. A new technique to unfold the Descemet membrane grafts using a single cannula was described. Medical records were reviewed to obtain the prestripped and poststripped endothelial cell counts (ECC), postoperative ECC, visual acuity measurements, and complications.
Results
Of the 43 prestripped tissues received, 40 were transplanted. The leading indications for DMEK were Fuchs endothelial corneal dystrophy (n = 28) and bullous keratopathy (n = 11). Nine DMEK procedures were performed in combination with phacoemulsification and posterior chamber intraocular lens implantation. Six patients had undergone prior glaucoma surgeries. The mean follow-up duration was 5.3 months (range, 1 week to 11 months). Preoperative spectacle-corrected visual acuity was ≤20/200 in 8 patients (20%) and ≤20/40 in 37 patients (92.5%). Primary graft failure occurred in the first case. Thirty-eight patients had improved vision postoperatively. Among the 39 patients who had successful DMEK, postoperative BCVA was ≥20/20 in 20 patients (51.2%), ≥20/25 in 30 patients (76.9%), and ≥20/40 in 34 patients (87.2%) by the last follow-up. There was no secondary graft failure. Rejection occurred in 2 patients because of self-discontinuation of topical corticosteroid. The most common complication was partial detachment requiring air injection (11 of 40 patients; 27.5%). Mean ECC loss after stripping of Descemet membrane was 3.9% (range, 6.5% gain to 14.5% loss). During the first 6 months after transplantation, the average ECC loss was 30.5% (range, 3.8%–67.4% loss).
Conclusions
DMEK using eye bank–prepared tissue achieved outcomes comparable to those reported for DMEK using surgeon-prepared tissue.
Descemet membrane endothelial keratoplasty (DMEK) offers better and faster visual rehabilitation and a lower risk of immune rejection than Descemet stripping endothelial keratoplasty (DSEK) and penetrating keratoplasty. It is replacing DSEK and becoming the preferred procedure for the treatment of endothelial dysfunction. However, obstacles in performing DMEK remain. DMEK is more technically difficult than DSEK, and it has a steeper learning curve. The success rate has increased drastically with the improvement of the surgical technique since the first report of DMEK in 2006. Many cornea surgeons have not started performing DMEK because of the challenge of donor preparation. If the stripping of the DMEK tissue is unsuccessful, it leads to tissue wastage and cancellation of the surgery.
Currently, an increasing number of eye banks in the United States have started to prepare prestripped DMEK donor tissues, but there is little published information about surgeon preference and factors that may affect the success and clinical outcome of DMEK. Prestripped tissues stored up to 2 days have been shown to achieve a clinical outcome similar to freshly stripped tissues. If tissues that are prestripped by an eye bank are proven to be as reliable and have the same quality as those prepared by the surgeons, eye bank–prepared tissues could help make DMEK more accessible to all cornea surgeons.
In the current study, we retrospectively evaluated the clinical outcomes of DMEK using tissues prepared by an eye bank. A new technique of unfolding the donor tissue is also described.
Methods
This retrospective, noncomparative study evaluated the first 40 consecutive DMEK procedures using tissues prestripped by an eye bank (Sightlife International, Seattle, Washington, USA). All DMEK procedures were performed at the Jules Stein Eye Institute, University of California, Los Angeles, during the period September 17, 2013 to July 1, 2014. The study was approved by the Institutional Review Board, and DMEK was performed in all cases by 1 surgeon (S.X.D.). Information collected from each patient’s medical record included indication for surgery, other ocular diagnoses, preoperative best-corrected Snellen visual acuity (BCVA), prestripped endothelial cell count (ECC), poststripped ECC, postoperative ECC, preoperative manifest refraction, postoperative manifest refraction, and complication type and rate. Prestripped and poststripped ECC were obtained from the eye bank. Postoperative ECC were obtained by specular microscopy (Konan Medical, Irvine, California, USA).
Graft Preparation
On the day before surgery, the eye bank technician manually stripped the Descemet membrane (DM) from the posterior stroma of the donor graft using the SCUBA technique, leaving a small portion of the DM-stromal adhesions intact at the periphery. The donor tissues had been previously stored in Optisol solution at 4°C. The prestripped donor cornea was again stored in Optisol solution and shipped in a container with a viewing chamber (Krolman Corp, Boston, Massachusetts, USA) at 4°C to the Jules Stein Eye Institute for next-day surgery.
Surgery
All surgeries were performed under monitored anesthesia care and retrobulbar block. The prestripped donor DM graft was further prepared as shown in Figure 1 . The corneal button was secured on a vacuum donor punch block. The edge of the donor DM was visualized by staining with 0.06% trypan blue dye (VisionBlue; Duch Ophthalmic, Exeter, New Hampshire, USA). If a small tear was present at the periphery, the graft was slightly decentered to avoid the torn area. An 8.0- to 8.75-mm trephine was used to create a partial-thickness cut. The size of the graft was determined by the size of the recipient cornea to allow a 1.5- to 2.0-mm space between the graft and the limbus. The rim of the DM was removed, and the inner trephined DM tissue was completely detached from the stroma by using 2 pairs of smooth tying forceps to grasp the edge of the DM. The DM was held by 1 pair of tying forceps and submerged in trypan blue dye for 15 seconds; subsequently, the DM was placed in balanced salt solution (BSS; Alcon, Ft Worth, Texas, USA) in a glass dish while the DM of the recipient was stripped.
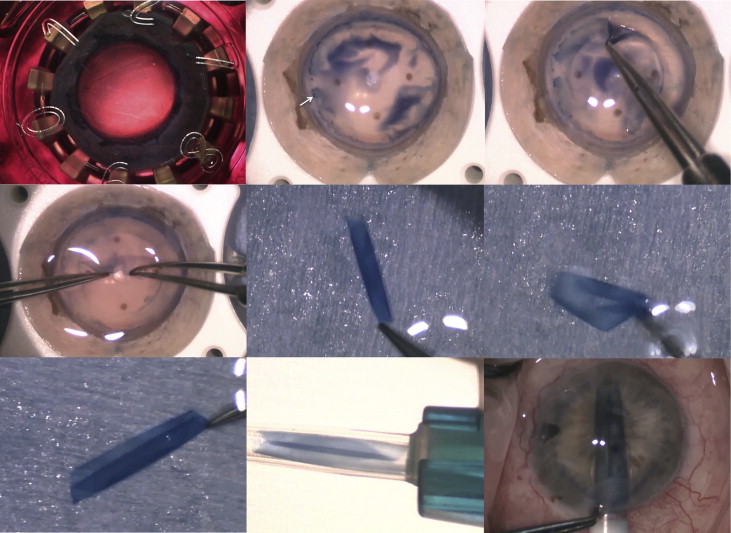
If the surgery was a combined DMEK and cataract surgery, the cataract was removed by phacoemulsification. The intraocular lens was implanted after DM was removed from the host. DM was stripped using a reverse Sinskey hook under viscoelastic (Healon; Abbott, Santa Ana, California, USA). The size of descemetorrhexis was the same as that of the graft. Two small peripheral iridectomies were made at 6 and 12 o’clock by straight intraocular microsurgical scissors (MicroSurgical Technology, Redmond, Washington, USA). A 2.4 mm (for Viscoject) or 3.0 mm (for Staar injector) temporal clear corneal incision was then made and viscoelastic was removed from the anterior chamber. Acetylcholine (Miochol; Bausch and Lomb, Bridgewater, New Jersey, USA) was injected intracamerally to induce miosis.
The edge of the DM graft was grasped with a pair of tying forceps, and the DM graft was placed in trypan blue for another 15 seconds and then back in BSS. A double scroll could be formed by gentle shaking of the DM in BSS as shown in Figure 1 (Middle right); this configuration facilitated unfolding of the DM scroll. The double-scroll DM was loaded into an injector (Staar Surgical, Monrovia, California, USA, or Viscoject; Medicel, Wolfhalden, Switzerland) with special attention to the removal of all air bubbles from the injector and cartridge. The graft was injected into the anterior chamber through the temporal incision and rotated during the insertion to keep the double scroll in an upward-facing position.
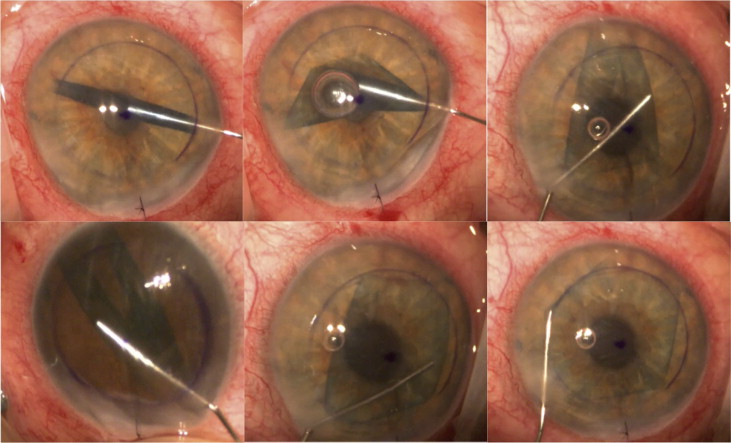
The donor graft was unfolded using a “touch, no touch” technique in which a 30 gauge cannula was used to unfold the DM scroll on the DM side without touching the endothelium ( Figure 2 , and Supplemental Video , available at AJO.com ). The cannula was inserted into the DM scroll. The tip of the cannula gently pushed the wall of the DM scroll in a circular outward motion. A combination of gentle stroking of the cornea, a small air bubble, and small spurs of BSS were used if necessary to completely unfold the DM graft. Once the DM graft was unfolded and centered, air was injected under the graft to attach it to the recipient stroma and to completely fill the anterior chamber for 10 minutes. At the end of this period, air was released to achieve a physiologic IOP <20 mm Hg while leaving the anterior chamber 90% air-filled. All patients received subconjunctival injections of dexamethasone and cefazolin. Vancomycin was used in patients who were allergic to cephalosporins. All patients were instructed to stay in a supine position in the recovery room for 1–2 hours, and then returned to the operating room to confirm proper positioning of the graft. If the IOP was judged to be higher than a physiologic pressure by digital palpation, a small amount of air was released from the anterior chamber. The patient was instructed to stay in the supine position overnight.
Postoperative air injection was performed under aseptic conditions in the minor procedure room if the patient had visually significant graft edema or symptoms of corneal edema within the first 4 weeks after surgery.
Results
A total of 43 prestripped tissues were received, and 40 were transplanted during the review period. The average age of the donors was 61 years (range, 52–73 years). The average time from donor death to surgery was 4 days (range, 2–7 days). All tissues were stripped at the eye bank and shipped in a cornea storage container with a viewing chamber the day before surgery ( Figure 1 , Top left). The viewing chamber permitted stabilization of corneal tissues during shipping. Prior to surgery, all tissues were grossly inspected for stromal opacity and presence of large folds in the DM. Except for 1 donor tissue, all stripped DM remained flat on the stromal bed, undisturbed by the shipping process. In the exception, the edge of the tissue scrolled under the remaining stripped DM. This scrolling was attributable to a 2- to 3-mm tear at the periphery. The scroll was unfolded and flattened successfully to permit trephination, which was slightly off center to avoid the peripheral tear. The edge of the stripped DM was found to be at the junction with the trabecular meshwork ( Figure 1 , Top center) in most tissues.
Three tissues were not transplanted. One was not used because of a tear in the central DM during the final preparation. The central portion of the DM was not peeled and was still tightly adhered to the stroma. A break occurred in the center of the DM during final peeling using a bimanual maneuver. The second tissue was not used owing to a low cell count after stripping. In the third case, the surgery was converted to DSEK. This patient had previous vitrectomy and glaucoma shunt implantation that complicated the DMEK procedure.
The average age of recipients was 72 years (range, 51–93 years). Seventeen patients were male (44%). The median size of the DM grafts was 8.25 mm (range, 7.75–8.75 mm). The indications for DMEK in the 40 patients were Fuchs endothelial corneal dystrophy (n = 28; 70%), bullous keratopathy (n = 11; 27.5%), and failed DMEK graft from outside facility (n = 1; 2.5%). Potential vision-limiting factors included corneal scar (9 patients), glaucoma (7 patients), age-related macular degeneration (3 patients), and cystoid macular edema (2 patients).
The mean follow-up time was 5.3 months (range, 1 week to 11 months). Thirty-one DMEK were performed as a single procedure, 9 as a combined procedure with cataract extraction and posterior intraocular lens implantation, 1 with pars plana vitrectomy, and 1 with amniotic patch graft removal. Four patients had previously undergone trabeculectomy and 2 had both trabeculectomy and glaucoma shunt implantation.
Thirty-nine of the 40 DMEK grafts cleared. One patient (2.5%) had primary graft failure and subsequently underwent successful DSEK. Preoperative spectacle-corrected visual acuity was ≤20/200 in 8 patients (20.0%) and ≤20/40 in 37 patients (92.5%). Thirty-eight of the 39 patients who had successful DMEK had improved vision. The vision of 1 patient remained at hand motion owing to end-stage glaucoma. By postoperative month 1, corrected visual acuity was ≥20/20 in 9 patients (23.1%), ≥20/25 in 18 patients (46.2%), and ≥20/40 in 26 patients (66.7%). By the time of last follow-up, corrected visual acuity ≥20/20 was achieved in 20 patients (51.1%), ≥20/25 in 30 patients (76.9%), and ≥20/40 in 34 patients (87.2%). The median VA was 20/20 (range, 20/20 to hand motion). The number of eyes reaching each BCVA level by the time of last follow-up is shown in Figure 3 .




