Purpose
To compare clinical outcomes after implantation of TECNIS diffractive multifocal intraocular lenses (IOLs) with different add powers.
Design
Prospective, comparative, interventional case series.
Methods
Subjects underwent cataract extraction and diffractive multifocal IOL implant. Subjects had an additional add power of +2.75 diopters (D) (Group 1, 23 eyes), +3.25 D (Group 2, 21 eyes), or +4.00 D (Group 3, 21 eyes). Evaluations were performed 1 and 3 months following cataract surgery, including measurement of monocular uncorrected distance visual acuity (UDVA), refractive error (manifest refraction [MR]), and monocular uncorrected near visual acuity (UNVA) at 33, 40, and 50 cm. At 3 months, internal aberrations and contrast sensitivity were evaluated and subjects completed a questionnaire on outcomes satisfaction, visual symptoms, and spectacle use.
Results
Sixty-five eyes (43 subjects) were included. After surgery, no significant differences between groups were observed in UDVA and MR values. UNVA at 33 cm was best in Group 3 (1 month: P = .032; 3 months: P = .031). Three months after surgery, UNVA was best in Group 1 at 50 cm ( P = .017). No significant differences in internal aberration and contrast sensitivity were observed between groups and surgical satisfaction was worst in Group 3 ( P = .001). A total of 87.0%, 85.7%, and 76.9% had spectacle independence in Groups 1, 2, and 3, respectively ( P = .080).
Conclusions
TECNIS diffractive multifocal IOLs with lower add powers provide good vision over longer working distances and excellent distant vision. Subjects with lower add power had greater satisfaction, more spectacle independence, and fewer visual symptoms than those with +4.00 D add.
Recent design advances in intraocular lenses (IOLs) have resulted in excellent visual outcomes after cataract surgery. As a result, many patients now have higher expectations of vision following surgery, including complete spectacle independence. Implantation of diffractive multifocal IOLs is an effective way to satisfy the desire for both good distance and near visual acuity. The principle by which diffractive IOLs work is to provide a focused image with zero-order diffraction for distance vision and first-order diffraction for near vision. However, with this optical set-up, the focused retinal image is always overlaid by an out-of-focus image from other diffractive portions of the lens and by background light or light scattering due to diffraction inefficiency. This often results in unintended side effects after implantation of diffractive multifocal IOLs, including decreased contrast sensitivity and unwanted visual artifacts (eg, glare and halos).
The TECNIS multifocal IOL is designed with a full diffractive posterior surface. This provides a balanced light distribution between distant and near foci and visual performance is independent of pupil size. In addition, the TECNIS multifocal IOL has the same aspheric anterior surface as the TECNIS monofocal IOL, which improves contrast sensitivity while decreasing spherical aberrations. The first TECNIS multifocal IOL (ZM900) was a 3-piece silicone lens. Later lens models have included 3-piece (ZMA00) and 1-piece (ZMB00) acrylic diffractive lenses with an additional power of +4.00 diopters (D).
The recently introduced ZKB00 and ZLB00 TECNIS multifocal IOLs have the same design as the ZMB00, but have lower additional power options of +2.75 D and +3.25 D, respectively. These IOLs allow physicians and patients to make individualized choices, in terms of target diopters, to customize patients’ lenses to their lifestyle. Lower additional power provides better intermediate vision for using computers and electronic devices. Additionally, fewer and wider concentric diffractive rings are thought to reduce the incidence and severity of unwanted postoperative visual artifacts. Here, we evaluate near and distance visual outcomes of new low-add-power multifocal IOLs. These outcomes are also compared to those obtained with an earlier diffractive IOL model. Postoperative visual quality was also assessed for each add power investigated.
Methods
This study was approved by the Institutional Review Board of Severance Hospital (No. 4-2014-0837) and registered at ClinicalTrials.gov (identification number NCT02424162 ). Study conduct adhered to the principles of the Declaration of Helsinki. All patients provided written informed consent to participate in the study and to have their medical information used in study analyses.
This prospective, comparative, interventional case series included cataract patients who had a desire for spectacle independence for distance and near vision. Exclusion criteria included previous ocular surgery or trauma and the presence of corneal opacities, fundus abnormalities, glaucoma, uveitis, amblyopia, systemic disease, or posterior capsule rupture during cataract surgery. All subjects underwent standard-of-care senile cataract extraction with multifocal IOL implantation with an add power of +2.75 D (TECNIS ZKB00; Abbott Medical Optics, Santa Ana, California, USA; Group 1), +3.25 D (TECNIS ZLB00; Abbott Medical Optics; Group 2), or +4.00 D (TECNIS ZMB00; Abbott Medical Optics; Group 3) at Yonsei University College of Medicine in Seoul, South Korea. Intended near correction distances were 50 cm, 40 cm, and 33 cm for the TECNIS ZKB00, ZLB00, and ZMB00, respectively. The multifocal IOL was selected based on patient visual needs and lifestyle.
Surgical Technique
All surgeries were performed by the same surgeon (T.-I.K.) under topical anesthesia. After creating an initial side port incision with a 15-degree blade, 3-step clear corneal incisions were made using a 2.5 mm disposable metal blade. Sodium hyaluronate 1.0% (Healon; Abbott Medical Optics) was then injected into the anterior chamber. A continuous curvilinear capsulorrhexis, measuring 5.5 mm in diameter, was generated using a 26 gauge bent needle. After hydrodissection, the WhiteStar Signature System (Abbott Medical Optics) was used for phacoemulsification of nuclear fragments and aspiration of residual cortex. In all eyes, a multifocal IOL was implanted in the capsular bag using a disposable implantation system. Irrigation and aspiration were performed to minimize ophthalmic viscoelastic surgical device retention at the end of surgery. The main incision and side port were sealed with stromal hydration using a balanced salt solution.
Patient Examinations
Postoperative evaluations were performed 1 and 3 months after cataract surgery. All subjects underwent measurement of monocular uncorrected distance visual acuity (UDVA) and manifest refraction (MR) values. Monocular uncorrected near visual acuity (UNVA) was also measured using a Rosenbaum near vision card at 33, 40, and 50 cm. Internal aberrations were measured without pharmacologic dilation using a ray-tracing aberrometer (iTrace; Tracey Technologies, Houston, Texas, USA) at a pupil size of 4 mm or more under mesopic conditions. Data were recalculated to reflect a 4.0 mm pupil. The root mean square (RMS) total aberration was calculated, along with RMS total higher-order aberration, using coma, trefoil, and spherical aberration measurements.
Contrast sensitivity was measured using the Functional Acuity Contrast Test (FACT) of the Optec 6500 view-in test system (Stereo Optical Co, Inc, Chicago, Illinois, USA) with stimulus spatial frequencies between 1.5 and 18 cycles/degree (cpd) under photopic (target luminance = 85 candelas/square meter [cd/m 2 ]) and mesopic (target luminance = 3 cd/m 2 ) conditions with and without glare. Twelve white light-emitting diodes (LEDs) were used as the source of glare, which was 10 lux under photopic conditions and 1 lux under mesopic conditions. Finally, 3 months after surgery, subjects were asked to complete a questionnaire regarding overall satisfaction following surgery, presence of visual symptoms, and spectacle dependence for near and far vision. Overall satisfaction was assessed using the 5-point Likert scale (1 = very dissatisfied; 2 = dissatisfied; 3 = neither satisfied nor dissatisfied; 4 = satisfied; 5 = very satisfied).
Statistical Analyses
Results are presented as mean ± standard deviation. All visual acuity measurements were converted to the logarithm of the minimal angle of resolution (logMAR) for data analyses. The Kruskal-Wallis test was used to compare the 3 independent study groups. Categorical data were compared using the Pearson χ 2 test. All statistical analyses were performed using a commercially available statistical software package (SPSS version 20; SPSS, Inc, Chicago, Illinois, USA). Statistical significance was defined as P < .05.
Results
Sixty-five eyes of 43 patients were included in this study, with 23 eyes in Group 1, 21 eyes in Group 2, and 21 eyes in Group 3. Preoperative patient and ocular characteristics of the 3 groups are summarized in Table 1 .
| Parameter | Group 1 TECNIS ZKB00 | Group 2 TECNIS ZLB00 | Group 3 TECNIS ZMB00 | P Value a |
|---|---|---|---|---|
| Eyes (n) | 23 | 21 | 21 | — |
| Female (%) | 73.9 (17 of 23) | 61.9 (13 of 21) | 52.4 (11 of 21) | — |
| Age (y) | 61.70 ± 12.08 | 57.67 ± 14.48 | 61.38 ± 6.21 | .674 |
| Axial length (mm) | 23.73 ± 0.86 | 23.47 ± 1.00 | 23.71 ± 0.91 | .567 |
| BCDVA (logMAR) | 0.18 ± 0.14 | 0.19 ± 0.22 | 0.31 ± 0.34 | .105 |
| IOL power (D) | 20.21 ± 1.74 | 21.38 ± 3.12 | 19.93 ± 2.97 | .280 |
| Target refraction (D) | −0.05 ± 0.17 | −0.11 ± 0.97 | −0.065 ± 0.16 | .785 |
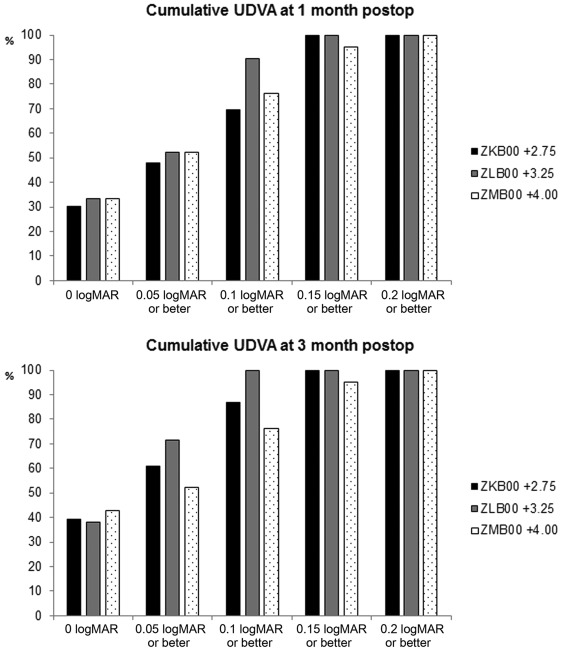
Table 2 shows postoperative UDVA, UNVA, MR values, and the difference between target refraction and MR values. No significant differences between groups were observed in UDVA and MR values. However, UNVA at 33 cm was better in Group 3 than in other groups at 1 ( P = .032) and 3 ( P = .031) months following surgery. The UNVA at 50 cm was significantly better in Group 1 than in Groups 2 and 3 at 3 months after surgery ( P = .017). This was not the case for UNVA at 40 cm, which was not significantly different between groups. Both UDVA and UNVA over the follow-up period are shown in Figures 1 and 2 .
| Parameter | Group 1 TECNIS ZKB00 | Group 2 TECNIS ZLB00 | Group 3 TECNIS ZMB00 | P Value |
|---|---|---|---|---|
| One month after surgery | ||||
| MR spherical equivalent (D) | −0.19 ± 0.38 | −0.11 ± 0.21 | 0.011 ± 0.28 | .098 |
| Target refraction – MR (D) | 0.14 ± 0.39 | 0.011 ± 0.22 | −0.077 ± 0.32 | .139 |
| UDVA (logMAR) | 0.083 ± 0.058 | 0.062 ± 0.052 | 0.071± 0.064 | .479 |
| UNVA 33 cm (logMAR) | 0.18 ± 0.052 | 0.15 ± 0.10 | 0.11 ± 0.079 | .032 a |
| UNVA 40 cm (logMAR) | 0.17 ± 0.049 | 0.16 ± 0.067 | 0.15 ± 0.053 | .601 |
| UNVA 50 cm (logMAR) | 0.13 ± 0.056 | 0.14 ± 0.081 | 0.21 ± 0.11 | .503 |
| Three months after surgery | ||||
| MR spherical equivalent (D) | −0.038 ± 0.28 | −0.097 ± 0.22 | −0.036 ± 0.23 | .818 |
| Target refraction – MR (D) | −0.012 ± 0.23 | −0.033 ± 0.25 | −0.029 ± 0.27 | .914 |
| UDVA (logMAR) | 0.070 ± 0.056 | 0.045 ± 0.042 | 0.067± 0.068 | .368 |
| UNVA 33 cm (logMAR) | 0.17 ± 0.070 | 0.13 ± 0.078 | 0.11 ± 0.085 | .031 a |
| UNVA 40 cm (logMAR) | 0.15 ± 0.067 | 0.14 ± 0.075 | 0.15± 0.075 | .838 |
| UNVA 50 cm (logMAR) | 0.10 ± 0.047 | 0.14 ± 0.074 | 0.18± 0.083 | .017 a |
a Statistical significance, as determined with the Kruskal-Wallis test.
Internal aberrations 3 months after surgery are reported in Table 3 . No significant difference was observed between the 3 study groups in RMS total aberration, RMS total higher-order aberration, coma, trefoil, or spherical aberration ( Table 3 ). Contrast sensitivity was also not statistically different between the 3 groups ( Figure 3 ).
| Parameter | Group 1 TECNIS ZKB00 | Group 2 TECNIS ZLB00 | Group 3 TECNIS ZMB00 | P Value a |
|---|---|---|---|---|
| RMS total (μm) | 0.76 ± 0.52 | 0.99 ± 0.35 | 0.92 ± 0.63 | .051 |
| Total HOA (μm) | 0.49 ± 0.46 | 0.74 ± 0.51 | 0.53 ± 0.54 | .105 |
| Coma (μm) | 0.30 ± 0.28 | 0.32 ± 0.21 | 0.26 ± 0.31 | .474 |
| Spherical (μm) | −0.10 ± 0.21 | −0.19 ± 0.15 | −0.14 ± 0.20 | .059 |
| Trefoil (μm) | 0.19 ± 0.19 | 0.31 ± 0.28 | 0.24 ± 0.23 | .426 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


