Purpose
To investigate the association between trabecular meshwork anteroposterior length and anterior chamber angle width in nonglaucomatous subjects.
Design
Prospective, cross-sectional study.
Methods
Time-domain and spectral-domain anterior segment optical coherence tomography images for 561 eyes from 366 nonglaucomatous subjects were analyzed to determine trabecular meshwork anteroposterior length and 3 anterior chamber angle width parameters measured at different distances from the scleral spur: angle opening distance at 250 μm (AOD250), 500 μm (AOD500), and 750 μm (AOD750) from the scleral spur; trabecular–iris space area at 500 μm (TISA500) and 750 μm (TISA750) from the scleral spur; angle recess area at 750 μm (ARA750) from the scleral spur. Univariable and multivariable linear mixed-effect regression models, the latter adjusting for age, sex, ethnicity, axial length, and the use of both eyes in the same subject, were used to evaluate the association between trabecular meshwork anteroposterior length and anterior chamber angle width parameters.
Results
Mean trabecular meshwork anteroposterior length was 824.86 ± 181.77 μm. Univariate regression analyses showed a significant positive association between trabecular meshwork anteroposterior length and all anterior chamber angle width parameters: AOD250 ( P < .001; β = 335.13), AOD500 ( P < .001; β = 271.84), AOD750 ( P < .001; β = 202.56), TISA500 ( P < .001; β = 780.78), TISA750 ( P < .001; β = 449.17), ARA750 ( P < .001; β = 381.39). Multivariate regression analyses showed a significant positive association between trabecular meshwork anteroposterior length and all anterior chamber angle width parameters: AOD250 ( P = .005; β = 294.02), AOD500 ( P = .036; β = 172.94), AOD750 ( P = .049; β = 125.58), TISA500 ( P = .004; β = 611.51), TISA750 ( P = .011; β = 333.96), ARA750 ( P < .0001; β = 309.12).
Conclusions
Wider anterior chamber angle is associated with greater trabecular meshwork anteroposterior length.
Glaucoma is the leading cause of global irreversible vision loss. The number of people affected with glaucoma was approximately 60.5 million in 2010 and future projections estimate an increase to 76.0 million in 2020 and 111.8 million in 2040. Elevation of intraocular pressure (IOP) is the most consistent risk factor for glaucoma development. IOP arises from the balance between inflow and outflow of aqueous humor. Aqueous humor is secreted by the ciliary processes and is drained through the trabecular meshwork and uveoscleral outflow facilities. Flow through the uveoscleral outflow pathway is theoretically independent of IOP, whereas the trabecular meshwork outflow pathway is directly related to IOP. IOP builds up in response to resistance in the trabecular meshwork until the pressure gradient is high enough to drive aqueous humor across the trabecular meshwork into the Schlemm canal. Obstruction of aqueous humor outflow through the trabecular meshwork pathway results in the rise of IOP that is central to the pathogenesis of glaucoma.
Foster and associates demonstrated in a population-based study that an inverse association exists between IOP and anterior chamber angle width. They postulated that eyes with wider angles have lower IOP owing to greater trabecular meshwork outflow facility. Since IOP is regulated by the trabecular meshwork, it is plausible that one of the mechanisms in which the angle configuration influences IOP may be through modulation of the trabecular meshwork outflow facility. Hypothetically, greater trabecular meshwork anteroposterior length could provide larger surface area for aqueous humor outflow. The purpose of this study is to evaluate the association between trabecular meshwork anteroposterior length and anterior chamber angle width in nonglaucomatous subjects.
Methods
Study Design
Approval for this prospective, cross-sectional study was obtained from the University of California, San Francisco (UCSF) Committee on Human Research. The study was carried out in accordance with the tenets of the Declaration of Helsinki. All enrolled subjects provided written informed consent after explanation of the study and possible consequences. This study enrolled consecutive subjects who met the inclusion criteria and were willing to participate from the UCSF general ophthalmology and glaucoma clinics between January 31, 2012 and August 1, 2015. Inclusion criteria for subject enrollment included: (1) adult subjects (age >18 years); (2) subjects who consented to undergo standardized ophthalmic examination, time-domain anterior segment optical coherence tomography (TD-ASOCT) imaging, and spectral-domain anterior segment optical coherence tomography (SD-ASOCT) imaging; (3) confirmation of normal perimetry and normal-appearing optic discs; (4) absence of corneal abnormalities that may obscure the view of anterior segment structures; (5) absence of prior laser or incisional eye surgery; (6) absence of previous ocular trauma; and (7) absence of peripheral anterior synechiae on gonioscopy. Exclusion criteria for enrollment included the following: (1) eyes with glaucomatous optic neuropathy; (2) eyes with corneal abnormalities such as edema, abrasion or dystrophy, pterygium, and other degenerative changes; (3) eyes with iridociliary cysts; (4) aphakic or pseudophakic eyes; (5) inability to complete the standardized ophthalmic examination, TD-ASOCT imaging, or SD-ASOCT imaging; and (6) poor TD-ASOCT imaging or SD-ASOCT imaging quality (evaluated on the basis of corneal reflection, continuity of anterior segment structures, motion artifacts, and indeterminate scleral spurs). The International Society of Geographic and Epidemiologic Ophthalmology (ISGEO) created a classification scheme for diagnosing glaucoma in cross-sectional population-based research. In this study, we used the diagnostic criteria developed by the ISGEO as the basis for diagnosis of glaucomatous optic neuropathy. Glaucomatous optic nerve damage was confirmed by a glaucomatous optic nerve appearance (vertical cup-to-disc ratio >0.6) and a corresponding reliable visual field defect. A glaucomatous visual field defect was considered to be present when the hemifield test was graded outside normal limits (with a probability of less than 5% based upon comparison with age-matched controls in the pattern deviation plot) and showed a cluster of 3 or more non-edge contiguous points that did not cross the horizontal meridian. Visual fields were defined as reliable if they fulfilled the following criteria: fixation losses <33%, false positives <20%, and false negatives <20%.
Data Collection
All enrolled subjects received a standardized ophthalmic examination that included visual field testing using the 24-2 Swedish Interactive Threshold Algorithm (SITA) standard strategy with the Humphrey Visual Field Analyzer (model 750i; Carl Zeiss Meditec, Inc, Dublin, California, USA), IOP measurement by Goldmann applanation tonometry (model AT900; Haag-Streit AG, Koeniz, Switzerland), slit-lamp examination (model BM900; Haag-Streit AG), axial length measurement by optical biometry (Model Lenstar LS 900; Haag-Streit AG), and gonioscopy with a Zeiss-style 4-mirror lens (model OPDSG; Ocular Instruments, Inc, Bellevue, Washington, USA). A single trained ophthalmologist (S.L.) performed gonioscopy at ×16 magnification with slit-lamp biomicroscopy in a darkroom setting.
Both TD-ASOCT imaging and SD-ASOCT imaging took place under standardized dark conditions with illuminations below 1 lux as measured by EasyView Digital Light Meter (model EA30; Extech Instruments, Inc, Waltham, Massachusetts, USA). Subjects were allowed 5 minutes for dark adaptation before image acquisition.
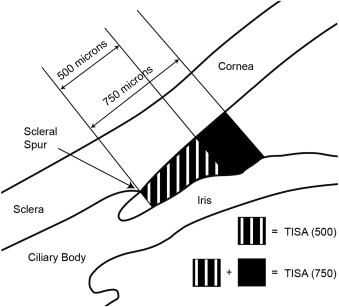
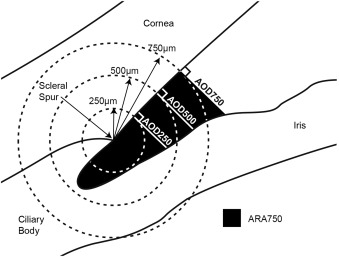
TD-ASOCT imaging was performed with Visante OCT (Carl Zeiss Meditec Inc). Each TD-ASOCT scan captured both the temporal and nasal quadrants (nasal-temporal 0–180 degrees) in a single image while the patient looked straight ahead. A total of 3–5 images were acquired for each eye and the image with the best quality was selected for analysis using the Zhongshan Angle Assessment Program (Zhongshan Ophthalmic Centre, Guangzhou, China). Image quality was evaluated on the basis of a steady central fixation as judged by a clear corneal reflection, good visibility of the scleral spurs, the presence of continuity in anterior segment structures, and the absence of motion artifacts. The Zhongshan Angle Assessment Program contained algorithms that automatically defined the borders and curvatures of anterior segment structures after the scleral spurs were localized manually on the TD-ASOCT images. Measurements for both the nasal and temporal angles were simultaneously produced, but only the nasal angles were analyzed in this study. This procedure has previously been used in our other studies. The following anterior chamber angle width parameters were derived from the Zhongshan Angle Assessment Program: angle opening distance at 250 μm from the scleral spur (AOD250), angle opening distance at 500 μm from the scleral spur (AOD500), angle opening distance at 750 μm from the scleral spur (AOD750), trabecular–iris space area at 500 μm from the scleral spur (TISA500), trabecular–iris space area at 750 μm from the scleral spur (TISA750), and angle recess area at 750 μm from the scleral spur (ARA750). AOD250, AOD500, and AOD750 were defined as the length of the line extending from the anterior iris to the corneal endothelium, perpendicular to the line drawn along the trabecular meshwork at 250 μm, 500 μm, and 750 μm anterior to the scleral spur. TISA500 and TISA750 were defined as the trapezoidal area with the following boundaries: anteriorly, a perpendicular line between the inner corneoscleral wall and the iris surface at 500 μm and 750 μm anterior to the scleral spur; posteriorly, a line perpendicular to the inner corneoscleral wall extending from the scleral spur to the iris surface; superiorly, the inner corneoscleral wall; and inferiorly, the iris surface. ARA750 was defined as the area bordered by the anterior iris surface, corneal endothelium, and a line perpendicular to the corneal endothelium that is drawn to the iris surface from a point 750 μm anterior to the scleral spur. An illustration of TISA500 and TISA750 is provided in Figure 1 and an illustration of AOD250, AOD500, AOD750, and ARA750 is provided in Figure 2 .
SD-ASOCT imaging was performed with Cirrus OCT (Carl Zeiss Meditec Inc) using the anterior segment 5-line raster protocol. Measurements were derived from the 3rd scan (middle line). Study participants were instructed to fixate at an external point to center the nasal portion of the eye for the scan. Scans were conducted over the limbal area, perpendicular to the limbus and radial from the center of the cornea. SD-ASOCT images were analyzed with ImageJ software (US National Institutes of Health, Bethesda, Maryland, USA) to measure trabecular meshwork anteroposterior length. Trabecular meshwork anteroposterior length was defined as the distance from the scleral spur to the Schwalbe line. Two experienced operators (R.C. and D.B.), masked to the standardized ophthalmic examination findings, performed all the TD-ASOCT and SD-ASOCT scans.
Statistical Analysis
All statistical analyses were conducted with SPSS statistical software (version 21.0; SPSS Inc, Chicago, Illinois, USA). P values < .05 were considered to indicate statistical significance. Mean and standard deviation were calculated for all variables using data from both eyes. Univariable and multivariable linear mixed-effect regression models were used to evaluate the association between trabecular meshwork anteroposterior length and the 6 anterior chamber angle width parameters. Two separate multivariable linear mixed-effect regression models were used. The first multivariable linear mixed-effect regression model adjusted for age, sex, and the use of both eyes in the same subject. The second multivariable linear mixed-effect regression model adjusted for age, sex, ethnicity, axial length, and the use of both eyes in the same subject. Ethnicities were self-designated by the patients. Standardized regression coefficients (β) were calculated from both univariate and multivariate regression models to determine the predictive strength of AOD250, AOD500, AOD750, TISA500, TISA750, and ARA750 for trabecular meshwork anteroposterior length. To evaluate intraobserver variance, 20 eyes were randomly selected and the anterior segment biometric parameters were remeasured by the same observer at a separate session.
Results
This study enrolled a total of 410 consecutive nonglaucomatous subjects who met the inclusion criteria and were willing to participate. Among them, 44 subjects were excluded from the study because of 1 or more of the following reasons: (1) the subject could not or did not complete the TD-ASOCT or SD-ASOCT examination and (2) incomplete data in at least 1 of the analysis variables owing to poor image quality or inability to assess scleral spur. Following exclusion, 366 subjects were available, among whom 561 eyes were included in the analysis. Table 1 provides the demographics and clinical characteristics of the included study subjects. The study population had a trabecular meshwork anteroposterior length average of 824.86 ± 181.77 μm, AOD250 average of 0.17 ± 0.11 mm, AOD500 average of 0.24 ± 0.15 mm, AOD750 average of 0.34 ± 0.20 mm, TISA500 average of 0.10 ± 0.05 mm 2 , TISA750 average of 0.18 ± 0.09 mm 2 , and ARA750 average of 0.22 ± 0.13 mm 2 .



