Purpose
To evaluate the association between amantadine use and corneal toxicity in a nationwide population.
Design
Retrospective cohort study of nationwide population-based administrative database.
Methods
This study analyzed data in the Taiwan Longitudinal Insurance Database for a group of 8195 patients diagnosed with Parkinson disease during a 15-year period (January 1, 1996 to December 31, 2010). A control group of 8195 patients without Parkinson disease was randomly matched with the Parkinson group by age, sex, and comorbidity index. The Kaplan-Meier method was used to calculate the cumulative incidence of corneal edema. Incident rate ratios and Cox proportional hazard regressions were estimated to compare the risk of corneal edema. The same methods were then used to compare the risk between patients with and without amantadine treatment.
Results
The incidence of corneal edema in the Parkinson group (123 patients; 1.50%) was significantly higher than that in the control group (82 patients; 1.0%) ( P = .004). The incidence ratio for corneal edema in the Parkinson group vs the controls was 5.77. When the Parkinson group was further subgrouped by use and non-use of amantadine, the hazard ratio for corneal edema was 1.79 times higher in the amantadine subgroup. Analyses of the amantadine subgroup by cumulative dose revealed that the 30-day hazard ratio for corneal edema was 2.05 higher in patients given moderate doses (2000–4000 mg) of amantadine and 2.84 times higher in the subgroup of patients given high doses (>4000 mg).
Conclusions
Amantadine increases the risk of corneal edema in a dose-dependent manner.
Amantadine, an antagonist of the N-methyl-D-aspartate-type glutamate receptor, was originally used as an antiviral medication for preventing and treating type A influenza. Since 1995, amantadine has been widely used to control tremors, rigidity, and associated drug-induced dyskinesia in Parkinson disease. Its 2 main derivatives are amantadine hydrochloride (Symmetrel) and amantadine sulfate (PK-Merz). Although the effectiveness of amantadine is well established, the mechanisms of its activity are not fully understood.
Amantadine is readily absorbed in the gastrointestinal tract. In adults for whom 200 mg per day is suitable, blood plasma values are in the range of 0.3∼0.6 μg/mL. The treatment duration is very short (from 10 days to 4 weeks) when amantadine is used as an antiviral agent and relatively longer when amantadine is used to treat Parkinson disease.
An association between corneal edema and amantadine use was first reported in 1990. Reported side effects on the cornea include punctate epithelial erosions, subepithelial opacities, and epithelial and stromal edema. Evidence of the corneal toxicity of amantadine use has also accumulated. Specifically, severe corneal edema develops soon after initiation of amantadine therapy and resolves within a few weeks after cessation of amantadine. The damage to the cornea includes irreversibly diminished endothelial density. However, irreversible corneal edema can occur and apparently be relieved several years after amantadine therapy by corneal transplantation performed via penetrating keratoplasty or via Descemet stripping automated endothelial keratoplasty. Therefore, prolonged amantadine use is a presumed cause of irreversible stromal edema.
We previously reported 4 cases of reversible corneal edema and permanent endothelial loss caused by amantadine use. In 2 of the 4 cases, symptoms were delayed for 12 and 18 months. We hypothesized that the presence of amantadine in the aqueous humor may damage the corneal endothelium in a dose-dependent manner. French and Margo had found amantadine-associated corneal edema in a large population. However, the limitation of this study was lack of control group. Therefore, to obtain sufficient statistical power, this study used the Taiwan Longitudinal Health Insurance Database to investigate the association between amantadine use and corneal edema and compared it with the controls. Furthermore, we tried to figure out the dose-related toxic effect of amantadine on the endothelium.
Methods
Database
This is a population-based retrospective cohort study. The single-payer National Health Insurance (NHI) program launched in Taiwan on March 1, 1995 offers comprehensive medical care coverage to all Taiwan residents. As of the year 2010, 23.16 million residents were enrolled in the program. The Longitudinal Health Insurance Database 2010 (LHID 2010) is a sub-dataset created by the Taiwan National Health Research Institute (NHRI). The LHID 2010 contains original claims data for 1 million beneficiaries for year 2010. The NHI Research Database provides encrypted patient data, including identification numbers; sex; birth dates; International Classification of Diseases, Ninth Revision, Clinical Modification (ICD-9-CM) codes for diagnoses and procedures; dates of admission and discharge; and details of prescriptions and other costs covered by the NHI. Researchers have validated the accuracy and completeness of the claims data contained in NHI research database. The NHRI approved the use of the LHID2010 in this study. This study project was reviewed and approved by the Institutional Review Committee of Kaohsiung Medical University Hospital (KMUHIRB-EXE MPT(II)-20160012), Taiwan.
Selection of Patients and Variables
Figure 1 shows the selection criteria applied in this retrospective study. The analysis included all 1 million beneficiaries enrolled in year 2010 (LHID 2010), and the analysis was limited to new episodes of ocular comorbidity by excluding cases previously diagnosed with corneal edema according to the dates on which ICD-9-CM codes were entered since the NHI program began in 1996. The ICD-9-CM codes for corneal edema and the 141 excluded cases were 371.00 (corneal opacity), 371.20 (corneal edema), 371.21 (idiopathic corneal edema), 371.22 (secondary corneal edema), and 371.23 (bullous keratopathy). The analysis also excluded 246 cases in which corneal edema had developed before initiation of systemic amantadine therapy. The number of ophthalmologic outpatient visits was also tracked, and patients were excluded if the number of outpatient claims was lower than 4 (n = 4663). After further exclusion of 195 697 patients who were not aged 18–100 years, 799 253 patients were enrolled. The enrolled subjects included a study cohort and control cohort. The study cohort comprised 8195 patients who had received ICD-9-CM codes 332.0 (paralysis agitans or Parkinsonism, primary) or 332.1 (secondary Parkinsonism) in neurologic care visits between January 1, 1996 and December 31, 2010.
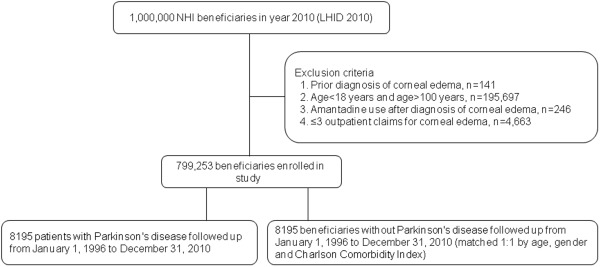
Propensity analysis was performed using logistic regression to obtain a propensity score for covariates in each patient. Because a sample matched by propensity score reduced selection bias in this study, the control cohort was extracted from the same group (n = 799 253) in the LHID 2010, and the same number of patients without Parkinson disease (n = 8195) was randomly selected. The control cohort was matched with the Parkinson cohort in terms of sex, age group (18–50 or >50), urbanization level (2 levels), and Charlson comorbidity index (CCI) (score = 0, 1–2, ≥3).
The effect of amantadine use on the cornea was analyzed by dividing each cohort into subgroups with and without amantadine exposure (amantadine subgroup and non-amantadine subgroup). Therefore, 4 subgroups were analyzed, 2 in the Parkinson cohort and 2 in the control cohort. The daily dose (number of pills × 100 mg), duration (days), and the cumulative dosage of amantadine were also recorded and compared between the amantadine and non-amantadine subgroups. Moreover, the cumulative dosages, durations, and the average daily doses of amantadine use that caused corneal complications in the amantadine subgroups were also investigated.
Statistical Analysis
Pearson χ 2 tests were performed in descriptive statistical analyses to compare the characteristics of the Parkinson and control cohorts in terms of demographic characteristics, urbanization, socioeconomic status, occupation, and CCI score. The dosage and frequency of amantadine use were also recorded.
The Kaplan-Meier method was used to compare the cumulative incidence of corneal edema between individuals with and without Parkinson disease, and the log-rank test was used to compare survival curves. The same method was used to compare the amantadine and non-amantadine subgroups in the Parkinson cohort.
To compare the risk of corneal edema between the Parkinson cohort and the control cohort, the incidence rate ratios (IRRs) were estimated by using a generalized linear model to analyze longitudinal data, and Poisson regression was performed in a log-linear model. Sex comparisons were also made in both the Parkinson cohort and control cohort. Next, the Cox proportional hazards regression model was used to calculate hazard ratios (HRs) with 95% confidence intervals (CIs) and P values between the 2 cohorts after adjusting for possible confounding factors (sex, age, urbanization, socioeconomic status, occupation, and CCI score).
All data analyses were performed using the SAS statistical package (Version 9.4.0; SAS Institute, Cary, North Carolina, USA). A P value less than .05 was considered statistically significant.
Results
Table 1 compares the distribution of demographic characteristics between the Parkinson cohort and control cohort. The mean age was 65.3 (SD, 16.2) in the Parkinson cohort and 65.1 (SD, 16.5) in the control cohort. After classification of each cohort into 2 age categories (18–50 years, older than 50 years), Pearson χ 2 tests revealed no significant differences in age distribution ( P = .4893), sex ( P = .2468), urbanization level ( P = .0884), or CCI score ( P = .0940).
| Parkinson Disease | Controls | P Value | |
|---|---|---|---|
| N | 8195 | 8195 | |
| Corneal edema patients, n (%) | 123 (1.50) | 82 (1.00) | .0040 |
| Mean age (SD), y | 65.3 (16.2) | 65.1 (16.5) | .4893 |
| Age group, n (%) | |||
| 18–50 | 1412 (17.2) | 1444 (17.6) | |
| >50 | 6783 (82.8) | 6751 (82.4) | .5099 |
| Sex, n (%) | |||
| Male | 3889 (47.5) | 3815 (46.6) | |
| Female | 4306 (52.5) | 4380 (53.4) | .2468 |
| Urbanization level, n (%) | |||
| 0 | 4292 (52.4) | 4183 (51.0) | |
| 1 | 3903 (47.6) | 4012 (49.0) | .0884 |
| Socioeconomic status (NTD), n (%) | |||
| <17 280 | 3955 (48.3) | 3470 (42.3) | |
| 17 281–22 880 | 3223 (39.3) | 3275 (40.0) | |
| 22 881–36 300 | 242 (3.0) | 323 (3.9) | |
| 36 301–45 800 | 288 (3.5) | 392 (4.8) | |
| >45 800 | 281 (3.4) | 382 (4.7) | <.0001 |
| Occupation, n (%) | |||
| 0 (Public servant and technical staff) | 3415 (41.7) | 3714 (45.3) | |
| 1 (Self-employed) | 1443 (17.6) | 1810 (22.1) | |
| 2 (Farmer, fisherman, water conservancy worker) | 2015 (24.6) | 1757 (21.4) | |
| 3 (Low income) | 1293 (15.8) | 864 (10.5) | |
| 4 (Other) | 29 (0.4) | 50 (0.6) | <.0001 |
| CCI, a n (%) | |||
| 0 | 363 (4.4) | 527 (6.4) | |
| 1–2 | 1471 (18.0) | 1262 (15.4) | |
| ≥3 | 6361 (77.6) | 6406 (78.2) | .0940 |
a The CCI was categorized into 3 levels (0, 1–2 and ≥3). No outpatients had a CCI of 0.
Of the 16 390 patients recruited, 205 (1.3%) had been diagnosed with corneal edema during the 15-year follow-up period. Of these 205 patients, 123 were in the Parkinson cohort (1.5%) and 82 were in the control cohort (1.0%). Notably, the Parkinson cohort had a significantly higher cumulative incidence proportion of corneal edema (1.5%, vs 1.0% in control cohort, P = .0040). During the follow-up period, the occurrence of corneal edema was significantly earlier in the Parkinson cohort (median, 3.2 years; interquartile range [IQR]: 1.5∼6.1 years, vs 5.1 years; IQR: 2.3∼8.7 in the control cohort; P < .0001).
Table 2 shows the data for amantadine use, including number of users, frequency of use, and cumulative dose. Comparisons of the 8195 subjects in each cohort showed that the percentage of patients who had received amantadine (100 mg) during the 15-year follow-up period was significantly ( P < .0001) higher in the Parkinson cohort (3132 patients; 38.22%) compared with the control cohort (1169 patients; 14.3%). Cumulative orally administered doses of amantadine were also significantly ( P < .0001) higher in the Parkinson cohort (average: 756.8 × 100 mg) compared with the control cohort (average: 27.6 × 100 mg). Finally, the duration of amantadine therapy was significantly ( P < .0001) longer in the Parkinson cohort (average, 402.2 days) compared with the controlled cohort (average, 13.6 days).
| Parkinson Cohort | Control Cohort | P Value | |
|---|---|---|---|
| N | 8195 | 8195 | |
| Amantadine use 100 mg, n (%) | 3132 (38.22) | 1169 (14.3) | <.0001 |
| Amantadine HCl 100 mg, n (%) | 1434 (17.50) | 1005 (12.3) | <.0001 |
| Amantadine sulfate 100 mg, n (%) | 1698 (20.72) | 164 (2.0) | <.0001 |
| Amantadine, median (SD) | |||
| Frequency of total clinic visits | 16.6 (28.7) | 3.1 (7.9) | <.0001 |
| Duration (d) of total drug use | 402.2 (758.9) | 13.6 (66.9) | <.0001 |
| Quantities of total drug tablets | 756.8 (1625.8) | 27.6 (120.7) | <.0001 |
| Quantity of drug tablets/d | 1.9 (0.8) | 2.3 (0.8) | <.0001 |
Incidence Rates of Corneal Edema
Table 3 shows the incidence of corneal edema during the 15-year follow-up period. The incidence of corneal edema per 1000 person-years (PY) was 2.67 in the Parkinson cohort and 0.67 in the control cohort. The Parkinson cohort also had a significantly higher IRR for corneal edema (3.98; CI, 3.01–5.26; P < .0001). After adjusting for age, urbanization, socioeconomic status, occupation, and CCI score, the adjusted HR for corneal edema in the Parkinson cohort was 5.77 (95% CI, 4.14–8.03) in comparison with the control cohort. Figure 2 shows the Kaplan-Meier curves. Table 3 shows that, after categorization by sex, the risk of developing corneal edema was still significantly higher in the Parkinson cohort. In male subjects, the IRR and adjusted HR were 3.50 (95% CI, 2.33–5.25) and 5.29 (95% CI, 3.23–8.65), respectively, for the Parkinson cohort in comparison with the control cohort. In female subjects, the IRR and adjusted HR were 4.46 (95% CI, 3.03–6.57) and 6.49 (95% CI, 4.13–10.21), respectively, for the Parkinson cohort in comparison with the control cohort. Additionally, the comparison of the risks of both sex groups with Parkinsonism also showed no significant difference, which means that corneal edema risk did not significantly differ by sex in the Parkinson cohort ( P = .4304).
| Corneal Edema/Total, Incidence % | Person-Years at Risk | Incidence per 1000 Person-Years (95% CI) | IRR (95% CI) a | Adjusted HR (95% CI) b | P | |
|---|---|---|---|---|---|---|
| Total cohort set | ||||||
| Control | 82/8195, 1.0% | 122 459 | 0.67 (0.67–0.67) | 1.00 | 1.00 | |
| Parkinson | 123/8195, 1.5% | 46 139 | 2.67 (2.64–2.69) | 3.98 (3.01–5.26) | 5.77 (4.14–8.03) | <.0001 |
| Male cohort set | ||||||
| Control | 41/3815, 1.1% | 56 973 | 0.72 (0.71–0.73) | 1.00 | 1.00 | |
| Parkinson | 45/3889, 1.4% | 21 444 | 2.52 (2.48–2.55) | 3.50 (2.33–5.25) | 5.29 (3.23–8.65) | <.0001 |
| Female cohort set | ||||||
| Control | 41/4380, 0.9% | 65 486 | 0.63 (0.62–0.63) | 1.00 | 1.00 | |
| Parkinson | 69/4306, 1.6% | 24 695 | 2.79 (2.76–2.83) | 4.46 (3.03–6.57) | 6.49 (4.13–10.21) | <.0001 |
a IRRs were calculated using PROC GENMOD to perform Poisson regression analysis (log-linear model).
b Hazard ratios with 95% confidence intervals and their P values were calculated using a Cox proportional hazards regression model adjusted for age, urbanization, socioeconomic status, occupation, and Charlson comorbidity index score and adjusted for sex in the combined group.
Relative Risk of Corneal Edema in Amantadine Users and Non-users
The relationship between corneal edema and amantadine uptake was also compared between the Parkinson group and control group. Table 4 shows that each cohort was divided into 2 subgroups, which resulted in 4 subgroups (Subgroup 1: controls without amantadine use [normal control group]; Subgroup 2: controls with amantadine use; Subgroup 3: Parkinson patients without amantadine use; and Subgroup 4: Parkinson patients with amantadine use). The relative risk (RR) of corneal edema during the 15-year follow-up period was calculated in comparison with the control/non-amantadine subgroup (n = 7026) after adjustment for age, sex, urbanization, socioeconomic status, occupation, and CCI score.
| Subgroup a | Amantadine Use | Parkinson Disease | Numbers (n) | Age, Years (SD) | Corneal Edema Events, n (%) | Adjusted RR (95% CI) | P | Adjusted RR (95% CI) | P |
|---|---|---|---|---|---|---|---|---|---|
| 1 (Normal control) | No | No | 7026 | 65.4 (16.4) | 71 (1.01) | 1.00 | |||
| 2 | Yes | No | 1169 | 63.5 (16.8) | 11 (0.94) | 0.94 (0.50–1.78) | .8545 | ||
| 3 | No | Yes | 5063 | 64.2 (17.3) | 57 (1.13) | 1.10 (0.77–1.57) | .5839 | 1.00 | |
| 4 | Yes | Yes | 3132 | 66.8 (14.2) | 66 (2.11) | 1.97 (1.41–2.76) | <.0001 | 1.79 (1.25–2.55) | .0013 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


