Purpose
To assess the agreement of parapapillary retinal nerve fiber layer (RNFL) thickness measurements among 3 spectral-domain optical coherence tomography (SD-OCT) instruments.
Design
Observational, cross-sectional study.
Methods
Three hundred thirty eyes (88 with glaucoma, 206 glaucoma suspects, 36 healthy) from 208 individuals enrolled in the Diagnostic Innovations in Glaucoma Study (DIGS) were imaged using RTVue, Spectralis and Cirrus in a single visit. Agreement among RNFL thickness measurements was assessed using Bland-Altman plots. The influence of age, axial length, disc size, race, spherical equivalent, and disease severity on the pairwise agreements between different instruments was assessed by regression analysis.
Results
Although RNFL thickness measurements between different instruments were highly correlated, Bland-Altman analyses indicated the presence of fixed and proportional biases for most of the pairwise agreements. In general, RTVue measurements tended to be thicker than Spectralis and Cirrus measurements. The agreement in average RNFL thickness measurements between RTVue and Spectralis was affected by age ( P = .001) and spherical equivalent ( P < .001), whereas the agreement between Spectralis and Cirrus was affected by axial length ( P = .004) and spherical equivalent ( P < .001). Disease severity influenced the agreement between Spectralis and both RTVue and Cirrus ( P = .001). Disc area and race did not influence the agreement among the devices.
Conclusions
RNFL thickness measurements obtained by different SD-OCT instruments were not entirely compatible and therefore they should not be used interchangeably. This may be attributable in part to differences in RNFL detection algorithms. Comparisons with histologic measurements could determine which technique is most accurate.
Although the assessment of the parapapillary retinal nerve fiber layer (RNFL) is essential in diagnosis and management of glaucoma, its objective evaluation remains a challenge in clinical practice. Quantitative measurements of RNFL thickness have become possible with the development of imaging technologies, such as optical coherence tomography (OCT). Earlier versions of this technology, known as time-domain OCT (TD-OCT), have demonstrated good reproducibility and accuracy for detection of RNFL loss in glaucoma. Recently, the introduction of spectral-domain optical coherence technology (SD-OCT) has greatly enhanced the resolution and decreased scan acquisition times compared to TD-OCT, potentially improving the ability to diagnose and follow glaucoma.
Three of the current commercially available SD-OCTs are the RTVue (Optovue Inc, Fremont, California, USA), the Cirrus SD-OCT (Carl Zeiss Meditec, Inc, Dublin, California, USA), and the Spectralis OCT (Heidelberg Engineering, Dossenheim, Germany). The principle involved in image acquisition is similar for all these devices and involves a scan with a diode laser that collects information of RNFL thickness in a 3.4-mm-diameter circle centered on the optic disc. Although the working principles are similar among the SD-OCTs, the agreement between them has not yet been reported. With an increasing number of commercially available SD-OCTs, it is likely that patients examined with 1 machine will have subsequent examinations performed with another device. Therefore, it is important to evaluate the agreement in RNFL thickness measurements among those devices.
The purpose of the present study was to assess the agreement of parapapillary RNFL thickness measurements among the RTVue, Cirrus, and Spectralis, and to evaluate the influence of age, race, spherical equivalent, axial length, disease severity, and optic disc size on the agreement among the devices.
Methods
Subjects
This was an observational cross-sectional study. Subjects included in this study were recruited from the longitudinal Diagnostic Innovations in Glaucoma Study (DIGS) conducted at the Hamilton Glaucoma Center (University of California, San Diego).
Each participant underwent a comprehensive ophthalmologic examination including review of medical history, best-corrected visual acuity, slit-lamp biomicroscopy, intraocular pressure measurement, gonioscopy, dilated funduscopic examination with a 78-diopter (D) lens, stereoscopic optic disc photography, and automated perimetry with the 24-2 Swedish Interactive Threshold Algorithm (SITA; Carl Zeiss Meditec, Inc). Axial length was acquired with IOLMaster (Carl Zeiss Meditec). Optic disc area was calculated using the Heidelberg Retina Tomograph (HRT II; software version 3.1.2.0, Heidelberg Engineering). Three scans centered on the optic disc were automatically obtained for each test eye, and a mean topography was created. Trained technicians outlined the optic disc margin while they viewed simultaneous stereoscopic photographs of the optic disc. Only images with a standard deviation of <50 μm were included.
To be included, subjects had to have best-corrected visual acuity of 20/40 or better, spherical refraction within ±5.0 D, cylinder correction within ±3.0 D, and open angles on gonioscopy. Eyes with coexisting retinal disease, uveitis, or nonglaucomatous optic neuropathy were also excluded from the investigation.
To study the agreement among the 3 devices in a broad cohort of patients, we included glaucomatous patients, individuals suspected of having the disease, and normal subjects. To be classified as glaucomatous, patients had to have at least 2 consecutive and reliable standard automated perimetry (SAP) examinations with either a pattern standard deviation (PSD) outside the 95% normal limits or a glaucoma hemifield test (GHT) result outside the 99% normal limits. Patients considered suspect for glaucoma had either an IOP greater than 21 mm Hg or suspicious appearance of the optic nerve head with at least 2 reliable normal visual fields, defined as a PSD within 95% confidence limits and a GHT result within normal limits. Normal control subjects were recruited from the general population and had IOP <22 mm Hg with no history of elevated IOP and with at least 2 reliable normal visual fields, defined as a PSD within 95% confidence limits and a GHT result within normal limits. All visual fields were reviewed by the VisFACT (Visual Field Assessment CenTer) visual field reading center. VisFACT checked for the presence of artifacts such as lid and rim artifacts, fatigue effects, inattention, or inappropriate fixation. To be considered reliable, all tests had to have false-positive responses, fixation loss, and false-negative responses ≤33%.
Instrumentation
Measurements of the parapapillary RNFL thickness were obtained in the same visit using the RTVue (Optovue Inc), the Cirrus HD-OCT (Carl Zeiss Meditec) and the Spectralis OCT (Heidelberg Engineering). Patients were examined without correction. Parapapillary RNFL thickness parameters that were automatically calculated by the machines and investigated in this study included average thickness (360-degree measure), temporal quadrant thickness (316 degrees to 45 degrees), superior quadrant thickness (46 degrees to 135 degrees), nasal quadrant thickness (136 degrees to 225 degrees), and inferior quadrant thickness (226 degrees to 315 degrees). All included images were checked for motion artifacts by inspection of the continuity of the scanned images (alignment of blood vessels). In addition, all images that had errors on RNFL segmentation were excluded from the analysis.
The RTVue (software version 4.0.5.39) uses a scanning laser diode with a wavelength of 840 nm to provide high-resolution images and has an acquisition rate of 26 000 A-scans per second. The imaging protocol used in this study was ONH (optic nerve head scan). This protocol generates a polar RNFL thickness map that is measured along a circle 3.45 mm in diameter centered on the optic disc. The RNFL thickness parameters are measured by assessing a total of 2325 data points between the anterior and posterior RNFL borders. Only good-quality images, as defined by a signal strength index of ≥30, were used for analysis.
The Cirrus SD-OCT (software version 4.5; Carl Zeiss Meditec) uses a superluminescent diode laser with a center wavelength of 840 nm and an acquisition rate of 27 000 A-scans per second. The protocol used for RNFL thickness evaluation was the optic disc cube. This protocol is based on a tridimensional scan of a 6 × 6-mm 2 area centered on the optic disc where information from a 1024 (depth) × 200 × 200-point parallelepiped is collected. Then, a 3.46-mm-diameter circular scan is placed around the optic disc and the information about parapapillary RNFL thickness is obtained. To be included, images were reviewed for noncentered scans and had to have a signal strength ≥7 and the absence of movement artifact.
Spectralis OCT (software version 3.1; Model Spectralis HRA+OCT) uses a dual-beam SD-OCT and a confocal laser scanning ophthalmoscope (CSLO) that uses a scanning laser diode with a wavelength of 870 nm and an infrared reference image simultaneously to provide images of ocular microstructures. The instrument has an acquisition rate of 40 000 A-scans per second. Spectralis OCT incorporates a real-time eye tracking system that couples CSLO and SD-OCT scanners to adjust for eye movements and to ensure that the same location of the retina is scanned over time. The protocol used was the RNFL circle scan, which consists of 1024 A-scan points from a 3.45-mm circle centered on the optic disc. All patients had their corneal curvature inputted into the machine before the examination. To be included, all images were reviewed for noncentered scans and had to have a signal strength >15 dB.
Statistical Analysis
The agreement among RNFL measurements obtained by the different instruments was investigated using Bland-Altman plots. The differences between measurements for each parameter were plotted against their mean. These plots allowed us to determine the existence of any systematic differences between the measurements (ie, a fixed bias). The mean difference between RNFL measurements obtained by the instruments reflects an estimation of the bias. In addition, we calculated the 95% limits of agreement for each comparison; that is, an estimate of how much the measurements differed in most individuals. Because both eyes per individual were used for the analysis, the limits of agreement were corrected for multiple measurements per individual, using a method described by Bland and Altman. Bland-Altman plots were also used to evaluate any possible relationship of the difference between the measurements and their average (ie, a proportional bias). The presence of proportional bias indicates that the limits of agreement will depend on the actual measurement. To formally evaluate this relationship, the difference between methods was regressed on their average. If the slope of the regression line was statistically significant, we considered the existence of proportional bias. To assess correlation between the 3 devices, we calculated the coefficients of determination (R 2 ) for pairwise measurements.
The influence of covariates on the agreement among instruments was assessed by regression analysis. The differences in RNFL thickness measurements between pairs of instruments were included as dependent variables and the covariates (age, axial length, spherical equivalent, race, disc size, mean deviation [MD]) as independent variables.
Generalized estimating equations with robust standard errors were used to adjust for potential correlations between both eyes of the same individual. Our sample size was sufficient to provide a narrow 95% confidence interval of approximately ±1.2 μm for the limits of agreement for the parameter average thickness in all pairwise comparisons.
All statistical analyses were performed with commercially available software (Stata version 10; StataCorp, College Station, Texas, USA, and SPSS ver 16.0; SPSS Inc, Chicago, Illinois, USA). The alpha level (type I error) was set at 0.05.
Results
The study included 330 eyes (88 with glaucoma, 206 glaucoma suspects, 36 normals) from 208 individuals. Table 1 shows clinical and demographic characteristics of included subjects. Mean age was significantly different among groups; glaucomatous patients were older, followed by glaucoma suspects and normals ( P < .001). Mean disease severity as measured by the visual field MD was −0.2 dB for normals, −0.66 dB for suspects, and −5.07 dB for glaucomatous ( P < .001). Mean axial length was similar for all groups; normals had a mean of 24.02 mm, suspects had 23.99 mm, and glaucomatous had 24.07 mm ( P = .986). Disc area, as measured by the HRT, did not significantly differ among groups: normals had a mean disc area of 2.03 mm 2 , glaucoma suspects had a mean of 2.03 mm 2 , and glaucomatous had a mean of 2.09 mm 2 ( P = .802). No difference was found in spherical equivalent among groups ( P = .738).
| Normals n = 36 | Suspects n = 206 | Glaucomatous n = 88 | P Value | |
|---|---|---|---|---|
| Age (years) | 58 ± 12 | 64 ± 12 | 69 ± 10 | <.001 |
| Sex (% male) | 29.4 | 39.8 | 53.4 | .026 |
| Race (% African-American) | 50 | 74.2 | 57.9 | .002 |
| Axial length (mm) | 24.02 ± 1.12 | 23.99 ± 1.1 | 24.07 ± 1.27 | .986 |
| Disc area (mm 2 ) | 2.03 ± 0.57 | 2.03 ± 0.46 | 2.09 ± 0.51 | .802 |
| Mean deviation (dB) | −0.2 ± 0.96 | −0.66 ± 1.45 | −5.07 ± 5.43 | <.001 |
| Spherical equivalent (diopters) | −0.24 ± 2.07 | −0.53 ± 2.21 | −0.445 ± 1.77 | .738 |
| Pseudophakia (%) | 0 | 16.5 | 32.9 | <.001 |
a Mean ± standard deviation values of age, axial length, disc area, spherical equivalent, and mean deviation reported by diagnostic group.
Table 2 shows mean values of average and quadrant RNFL thickness parameters obtained by each instrument. Table 3 shows the agreement between instruments. For average thickness, RTVue measurements were significantly thicker than Cirrus (difference = 9.77 μm; P < .001) and Spectralis (difference = 7.65 μm; P < .001) measurements. Statistically significantly thicker RTVue measurements were also found for all quadrants ( P < .001) compared to Cirrus and Spectralis. Cirrus measurements were significantly thinner than Spectralis for average thickness (difference = 2.12 μm; P < .001), temporal quadrant (difference = 8.01 μm; P < .001), and inferior quadrant (difference = 5.99 μm; P < .001). Measurements with Spectralis were thinner than Cirrus for the nasal quadrant (difference = 3.81 μm; P < .001). No significant difference was found for the superior quadrant between Cirrus and Spectralis ( P = .073).
| RTVue | Cirrus | Spectralis | |
|---|---|---|---|
| Average thickness (μm) | 92 (91.2–94.2) | 83 (81.5–85.3) | 85 (83.4–86.7) |
| Superior thickness (μm) | 110 (108.4–112.3) | 101 (98.7–102.9) | 99 (97–101.7) |
| Temporal thickness (μm) | 69 (67.8–70.6) | 58 (56.4–59) | 66 (64.2–67.2) |
| Inferior thickness (μm) | 119 (116.5–121.2) | 104 (102–106.9) | 111 (107.8–113.2) |
| Nasal thickness (μm) | 72 (70.9–73.8) | 68 (67.2–69.6) | 65 (62.9–66.3) |
| Parameter | Agreement | Mean Difference | P Value | Fixed Bias | R 2 | P Value | Proportional Bias | 95% Limits of Agreement a |
|---|---|---|---|---|---|---|---|---|
| Average thickness (μm) | RTVue-Cirrus | 9.77 | <.001 | YES | 0.029 | .002 | YES | −0.02 to 19.57 |
| RTVue-Spectralis | 7.65 | <.001 | YES | 0.04 | .005 | YES | −3.68 to 18.98 | |
| Cirrus-Spectralis | −2.12 | <.001 | YES | 0.129 | <.001 | YES | −13.13 to 8.89 | |
| Superior thickness (μm) | RTVue-Cirrus | 9.61 | <.001 | YES | 0.014 | .06 | NO | −7.32 to 26.53 |
| RTVue-Spectralis | 11.01 | <.001 | YES | 0.096 | <.001 | YES | −10.71 to 32.73 | |
| Cirrus-Spectralis | 1.4 | .073 | NO | 0.063 | <.001 | YES | −17.23 to 20.03 | |
| Temporal thickness (μm) | RTVue-Cirrus | 11.52 | <.001 | YES | 0.013 | .291 | NO | −4.56 to 27.60 |
| RTVue-Spectralis | 3.51 | <.001 | YES | 0.01 | .296 | NO | −14.77 to 21.79 | |
| Cirrus-Spectralis | −8.01 | <.001 | YES | 0.056 | <.001 | YES | −22.77 to 6.74 | |
| Inferior thickness (μm) | RTVue-Cirrus | 14.33 | <.001 | YES | 0.018 | .048 | YES | −3.47 to 32.12 |
| RTVue-Spectralis | 8.34 | <.001 | YES | 0.107 | <.001 | YES | −12.5 to 29.17 | |
| Cirrus-Spectralis | −5.99 | <.001 | YES | 0.057 | <.001 | YES | −24.37 to 12.39 | |
| Nasal thickness (μm) | RTVue-Cirrus | 3.96 | <.001 | YES | 0.056 | .003 | YES | −17.66 to 25.57 |
| RTVue-Spectralis | 7.77 | <.001 | YES | 0.042 | .001 | YES | −14.73 to 30.27 | |
| Cirrus-Spectralis | 3.81 | <.001 | YES | 0.142 | <.001 | YES | −22.38 to 29.99 |
a Corrected for multiple measurements per individual according to Bland and Altman.
Table 3 also reports the evaluation of the presence of proportional bias. Proportional bias was present if the difference and average of measurements by 2 instruments were significantly correlated. We found proportional biases for all pairwise measurements except for the superior RNFL thickness agreement between RTVue and Cirrus, the temporal RNFL thickness agreement between RTVue and Cirrus, and the temporal RNFL agreement between RTVue and Spectralis. The agreement among the devices and the 95% limits of agreement for the average RNFL thickness parameter are represented graphically in Figures 1 (RTvue vs Cirrus), 2 (RTVue vs Spectralis), and 3 (Cirrus vs Spectralis).
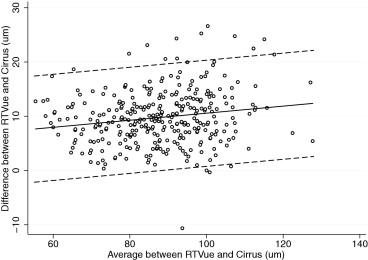
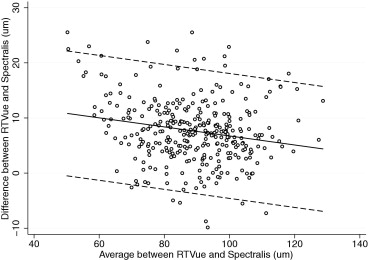
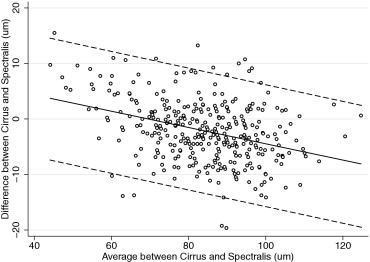
Table 4 shows the coefficients of determination (R 2 ) for all pairwise comparisons. Correlations ranged from R 2 = 0.30 (nasal quadrant, Cirrus-Spectralis) to R 2 = 0.87 (average thickness, RTVue-Cirrus). All correlations were statistically significant ( P < .001).
| Parameter | Agreement | Coefficient of Determination (R 2 ) | P Value |
|---|---|---|---|
| Average thickness (μm) | RTVue-Cirrus | 0.87 | <.001 |
| RTVue-Spectralis | 0.85 | <.001 | |
| Cirrus-Spectralis | 0.86 | <.001 | |
| Superior thickness (μm) | RTVue-Cirrus | 0.80 | <.001 |
| RTVue-Spectralis | 0.73 | <.001 | |
| Cirrus-Spectralis | 0.81 | <.001 | |
| Temporal thickness (μm) | RTVue-Cirrus | 0.62 | <.001 |
| RTVue-Spectralis | 0.58 | <.001 | |
| Cirrus-Spectralis | 0.71 | <.001 | |
| Inferior thickness (μm) | RTVue-Cirrus | 0.84 | <.001 |
| RTVue-Spectralis | 0.82 | <.001 | |
| Cirrus-Spectralis | 0.86 | <.001 | |
| Nasal thickness (μm) | RTVue-Cirrus | 0.37 | <.001 |
| RTVue-Spectralis | 0.49 | <.001 | |
| Cirrus-Spectralis | 0.30 | <.001 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


