After the publication of the recent Perspective entitled “ Counterfeit Avastin in India: Punish the Criminals, Not the Patients ” that referred to several cases of severe intraocular inflammation due to the use of bevacizumab in Gujarat, India, additional information has become available. These patients developed severe vitritis, some with an accompanying hypopyon, within 24 hours after injections from a single Avastin-labeled vial. All of the affected patients subsequently underwent vitrectomy for presumed endophthalmitis and four were found to have retinal necrosis. Microbial cultures from the vitreous specimens were negative in every case as were cultures performed on the contents of the Avastin vial. The final report on the incident stated that “no opinion” could be formulated regarding the contents of the vial.
Recently, a cluster of bevacizumab-related endophthalmitis cases occurred in Chandigarh, India. Twenty-eight of 30 patients that received Avastin from a single vial that had been purchased from an authorized Roche dealer developed inflammation within the first 24 hours after injection. Cultures of the intraocular fluids of 8 patients grew Stenotrophomonas maltophilia as did cultures from the Avastin vial.
The contents of the vial from the earlier Gujarat outbreak had not been analyzed for bevacizumab but the early onset (within 24 hours) of severe inflammation suggests the presence of a potent, pro-inflammatory contaminant. The clinical findings from the Gujarat cluster are more suggestive of a counterfeit product than a compounding-associated outbreak. The outbreak in Chandigarh is still being investigated and analysis of the vial’s contents is likely provide insight on whether this was a counterfeit product.
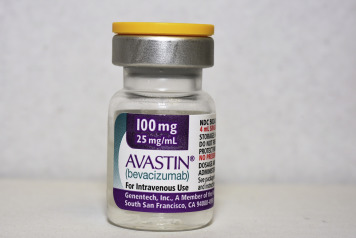
These episodes highlight the fragility of the supply chain in some countries and should remind ophthalmologists and compounding pharmacists to carefully inspect the labeling and integrity of each Avastin vial, including the factory-affixed cap ( Figure ). To avoid such outbreaks compounding pharmacies must aim to follow the guidelines issued by the United States Pharmacopeia Chapter 797 or their equivalent in their own countries.