Purpose
To compare the accommodative performance of the Morcher BioComFold Type 89A bag-in-the-lens intraocular lens (IOL) with a conventional in-the-bag control IOL in presbyopic eyes.
Design
Prospective, randomized clinical trial with intraindividual comparison.
Methods
setting : Department of Ophthalmology, St. Thomas’ Hospital, London, United Kingdom. study population : Fifty-two eyes of 26 patients with bilateral age-related cataracts. intervention : Phacoemulsification cataract extraction with implantation of a bag-in-the-Lens and a control IOL, the Alcon AcrySof SA60AT (Alcon Laboratories, Fort Worth, Texas, USA), randomized to either eye. main outcome measures : Axial IOL shift stimulated by physiologic (near visual effort) and pharmacologic (pilocarpine and cyclopentolate) accommodative stimulation was measured objectively with partial coherence interferometry. Other outcome measures were objective and subjective accommodation, logarithm of the minimal angle of resolution distance-corrected near visual acuity, and defocus curves.
Results
Three months after surgery, axial IOL shift stimulated by near visual effort measured −5.9 ± 10.3 μm in bag-in-the-lens eyes versus −8.4 ± 12.8 μm in control eyes ( P = .37), that stimulated by pilocarpine measured 20.2 ± 165.6 μm versus 50.4 ± 164.4 μm ( P = .36), and that stimulated by cyclopentolate measured −65.8 ± 64.3 μm versus −54.0 ± 37.5 μm ( P = .34), respectively (n = 25). Objective accommodation measured 0.03 ± 0.18 diopters (D) in bag-in-the-lens eyes versus 0.08 ± 0.21 D in control eyes ( P = .40), whereas subjective accommodation measured 2.48 ± 0.72 D versus 2.45 ± 0.80 D ( P = .75), respectively. Distance-corrected near visual acuity and defocus curves showed no difference between IOLs.
Conclusions
The bag-in-the-lens IOL demonstrated negligible axial shift and objective accommodation with physiologic near visual stimulation. The IOL shift demonstrated with pilocarpine also was clinically insignificant. The bag-in-the-lens IOL showed no accommodative or near visual advantage over a conventional in-the-bag IOL, despite its unique capsular fixation method. This provides further evidence that the focus-shift principle fails to produce clinically significant IOL movement.
Posterior capsule opacification (PCO) and postoperative spectacle dependence remain considerable problems after uncomplicated phacoemulsification cataract extraction and in-the-bag intraocular lens (IOL) implantation. Improvements in IOL biomaterials and design over the past 2 decades have reduced the rates of PCO and neodymium:yttrium–aluminum–garnet laser capsulotomy, but despite optimal IOL design and flawless surgical technique, PCO has not yet been eliminated and is a particular problem in pediatric and uveitic eyes. This led to the development of the Morcher BioComFold Type 89A bag-in-the-lens IOL (Morcher GmbH, Stuttgart, Germany). The bag-in-the-lens IOL is designed to prevent PCO completely and uses a unique capsular fixation technique. It is a hydrophilic acrylic, foldable IOL with a 5-mm optic diameter and 2 haptics comprising oval flanges, which are attached circumferentially around the IOL optic, thus forming a groove around the optic edge ( Figures 1 and 2 ). The surgery is challenging, requiring both anterior and posterior capsulorrhexes that must be identical and must measure precisely 5 mm in diameter. Both capsulorrhexes are inserted into the groove around the optic’s circumference, trapping any residual lens epithelial cells (LECs) within the closed space between the anterior and posterior capsule, thereby preventing PCO. The ability of the bag-in-the-lens IOL to prevent PCO has been demonstrated in vitro and in vivo in clinical trials across multiple European sites, and it has been implanted in children as young as 2 months of age. Follow-up in adults has been reported up to 6 years.
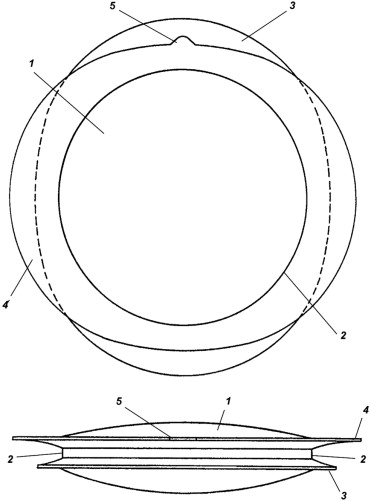
An additional significant clinical finding is that the peripheral lens capsule remains clear. This is supported by in vitro evidence that LEC transformation into fibroblast-like cells does not occur after successful bag-in-the-lens IOL implantation and that residual LEC growth is limited. The finding of preserved postoperative capsular transparency has led to the suggestion that capsular elasticity also may be preserved, and this may permit postoperative pseudophakic accommodation. No subjective or objective accommodative results have been published for presbyopic patients implanted with the bag-in-the-lens IOL, but subjective accommodative amplitudes were evaluated in a heterogeneous group of pediatric eyes and measured 4.30 ± 2.50 diopters (D; range, 2.50 to 12.00 D). Because of the unique capsular fixation technique, it is theoretically possible that ciliary muscle contraction, known to persist beyond the onset of presbyopia, or possibly vitreous pressure may drive forward axial movement of the bag-in-the-lens IOL, thereby delivering good near visual performance. The aim of this study was to evaluate the accommodative and near visual performance of the bag-in-the-lens IOL in presbyopic eyes in comparison with a conventional in-the-bag IOL.
Methods
The study was performed in the Department of Ophthalmology, St. Thomas’ Hospital, London, a tertiary referral center. Twenty-six patients were recruited prospectively to the study from routine outpatient clinics and were randomized to receive a bag-in-the-lens and a control IOL, the AcrySof SA60AT (Alcon Laboratories, Fort Worth, Texas, USA), in either eye. The second eye surgery was performed within 4 weeks of the first eye. Inclusion criteria were bilateral age-related cataracts and otherwise healthy eyes. Exclusion criteria were corneal astigmatism of more than 1.50 D (IOLMaster keratometry; Carl Zeiss Meditec, Jena, Germany), pupillary dilation less than 6.0 mm, and any coexisting ocular pathologic features, including amblyopia, corneal disease, inflammatory eye disease, glaucoma, diabetic retinopathy, age-related macular degeneration, or previous intraocular surgery. Patients requiring a bag-in-the-lens IOL power outside the range of 15.0 to 27.0 D also were excluded because a limited IOL bank was held in stock for practical reasons. Patients were reviewed on day 1 and 1 month and 3 months after surgery.
Intraocular Lenses
The bag-in-the-lens is a hydrophilic acrylic (polyHEMA [hydroxyethyl methacrylate] copolymer), foldable IOL. It has a 5.0-mm biconcave spherical optic that is surrounded circumferentially by 2 oval flanges that form its haptics ( Figure 1 ). The haptics, composed of the same material, are orientated perpendicularly to each other and measure 8.5 mm in length. A groove is formed around the equator of the IOL optic between the 2 haptics; this is where the capsulorrhexes are inserted during implantation. The AcrySof SA60AT, the control IOL, is a hydrophobic acrylic, foldable, single-piece IOL with a 6.0-mm spherical optic.
Preoperative Assessment
All patients underwent a complete ocular examination, including slit-lamp biomicroscopy, applanation tonometry, and dilated fundus examination. Biometry was performed with the IOLMaster (Carl Zeiss Meditec) using optical A-constants of 118.2 and 118.7 for the bag-in-the-lens and the SA60AT IOLs, respectively, aiming for a postoperative refraction of −0.25 D. Pupils were dilated before surgery with G cyclopentolate 1% and G phenylephrine 10% (Bausch & Lomb, Rochester, New York, USA).
Surgical Procedure
All surgery was performed by a single surgeon (D.J.S.) under topical anesthesia with G Tetracaine 1% (Bausch & Lomb). A 2.75-mm temporal clear corneal incision was made and the anterior chamber (AC) was inflated with viscoelastic.
Bag-in-the-lens surgical procedure
A 5.0-mm round polymethyl methacrylate ring caliper (Morcher GmbH) was inserted into the anterior chamber with forceps and placed on the anterior lens surface. The patient was instructed to look at the microscope light, and the caliper was centered around the first and fourth Purkinje images. A 5.0-mm circular continuous curvilinear capsulorrhexis (CCC) was made with capsulorrhexis forceps, guided precisely by the caliper. The caliper was removed, and hydrodissection, phacoemulsification using a stop-and-chop technique, and irrigation–aspiration were performed. The AC was refilled with viscoelastic, taking care not to fill the capsular bag, but instead to press the anterior capsule against the posterior capsule, allowing the anterior CCC to guide an identically sized and centered posterior CCC. The central posterior capsule was punctured with a 27-gauge needle, viscoelastic was injected, and the posterior CCC was made with capsulorrhexis forceps. The AC was refilled with viscoelastic and the bag-in-the-lens IOL was injected into the AC with a Naviject injector and Naviglide cartridge (2.8-1P Naviject injector set; Medicel AG, Luchten, Switzerland) and was maneuvered into position with a hook, inserting both capsulorrhexes into the groove between the bag-in-the-lens IOL’s 2 circumferential haptics.
SA60AT surgical procedure
A 5.0-mm anterior CCC was formed with capsulorrhexis forceps. Hydrodissection, phacoemulsification, and irrigation–aspiration were performed, the capsular bag was refilled with viscoelastic, and the IOL was injected into the bag with the Monarch II delivery system (Alcon Laboratories).
Postoperative Assessment
Axial IOL movement with accommodative stimulation
The main outcome measure, axial IOL movement, was measured 3 months after surgery as a change in anterior chamber depth (ACD) using the ACMaster (Carl Zeiss Meditec), a highly repeatable and precise instrument that uses partial coherence interferometry and measures parallel to the optical axis of the eye. Measurements were performed by a trained operator. Subjective refraction data was entered into the device, and ACD initially was measured with accommodation relaxed, with the patient viewing the instrument’s internal fixation target at their far point, simulating distance viewing. ACD then was remeasured by introducing −2 D of defocus via the instrument’s internal optometer, generating 2 D of accommodative stimulation. During measurements, the patient was encouraged to focus on the fixation target. G pilocarpine 4% then was administered to both eyes, and ACD was remeasured 60 minutes later. Pilocarpine-stimulated axial IOL shift was the outcome measure for which the sample size was calculated. One week later, ACD was remeasured under distance conditions, then again 30 minutes after administration of G cyclopentolate 1%, to assess for backward IOL movement with pharmacologic ciliary muscle relaxation. Central corneal thickness and IOL thickness measurements accompanying each ACD measurement were recorded to control for head and eye movements and to ensure sampling along a consistent optical path.
Objective accommodation
Objective accommodation was measured 3 months after surgery with the Tracey Wavefront Analyzer (Tracey Technologies, Houston, Texas, USA), an aberrometer with an open field of view, allowing patients to view targets at variable distances along the line of sight. An autorefraction was captured while the patient viewed a distance target, an illuminated letter on the logarithm of the minimal angle of resolution (logMAR) acuity chart, at their far point. Another autorefraction was captured while the patient viewed a near target, a black maltese cross on a white background, placed to induce 2 D of accommodative stimulation relative to their far point (e.g., for an eye with a plano refraction, the near target was placed at 50 cm). During near measurements, patients were encouraged to focus on the near target. The mean spherical equivalent refraction under distance and near conditions, respectively, was calculated as the average from 3 measurements. Objective accommodation (in diopters) was calculated as the difference between distance and near autorefractions.
Subjective accommodation
The subjective accommodative amplitude was measured for each eye with the Royal Air Force rule (Clement Clarke, Essex, United Kingdom) using the push-up test. The fellow eye was occluded, and the distance correction and a +2.50-D trial lens was placed in a trial frame. Patients were instructed to view the N8 line on the attached test chart, which initially was positioned at approximately 40 cm. The target then was moved toward the patient’s eye at approximately 2 cm per second, and they were asked to focus on the N8 line, keeping it clear for as long as possible. They were asked to indicate the first sign of blur and this distance was recorded (in centimeters). This procedure was repeated 3 times and averaged, then converted to a near point in diopters. The +2.50-D add was subtracted from this value, giving the final subjective amplitude of accommodation for each eye (in diopters).
Visual acuity
Uncorrected and best-corrected distance VA were measured uniocularly with an Early Treatment Diabetic Retinopathy Study logMAR acuity chart at 4 m (Precision Vision, La Salle, Illinois, USA). Uncorrected, distance-corrected, and near corrected logMAR near VA were measured with the Logarithmic Visual Acuity Chart 2000 at 40 cm (Precision Vision).
Defocus curves
Defocus curves were measured uniocularly with a 4-m logMAR Early Treatment Diabetic Retinopathy Study visual acuity chart with the distance correction in place. Spherical defocus was induced by adding trial lenses, from −3.0 to +3.0 D in 0.5-D steps, in a randomized order. LogMAR acuity was measured under all conditions of defocus.
Subjective refraction
Subjective refraction was performed uniocularly, aiming to achieve the best-corrected visual acuity compatible with maximum plus sphere and minimum minus cylinder. Duochrome testing was performed, aiming for a red-equals-green end point.
Pupil diameter
Pupil diameter (in millimeters) was measured with the Colvard Pupillometer (Oasis Medical, Inc, Glendora, California, USA).
Statistics
Sample size calculation
A sample size calculation was performed for the main outcome measure, axial IOL movement. A previous study conducted by our research group compared axial IOL movement between an accommodating and a nonaccommodating IOL using the same methods (pilocarpine-induced IOL movement measured with partial coherence interferometry with the ACMaster). This study detected a difference in movement between the 2 IOL types of 248 μm ( P < .0001), and the standard deviation of accommodating IOL movement was 169 μm. The power of this study was determined to be 0.85. For the present study, a target difference of 200 μm was used. Using a power of 0.80 and a P value of .05, the sample size was calculated to be approximately 22 patients. Allowing for a 15% drop-out rate, 26 patients were recruited to the study.
Randomization method
A random allocation sequence was generated before commencement of the trial using a computerized randomization program in a single block with a 1:1 allocation ratio. Patients were allocated sequentially to the next available number on this unconcealed randomized sequence after recruitment to the study. The first eye was randomized to receive a bag-in-the-lens or a control IOL (SA60AT), and second-eye surgery was performed with the alternative IOL within 4 weeks. Neither participants or investigators were blinded.
Analysis
The analysis was intention to treat, and continuous variables were described in terms of means and standard deviations. Data were checked for normality with the Kolmogorov-Smirnov test. Paired observations between the 2 IOL groups were compared by calculating differences between means and corresponding 95% confidence intervals, and significance was calculated using paired t tests. The significance level initially was set at P < .05. Multiple comparisons of outcome measures were made (IOL movement under 3 conditions, objective and subjective accommodation, and visual acuity under 5 conditions); therefore, a Bonferroni correction was made, thus correcting the significance level to P < .005. GraphPad Prism software version 5.0b was used (GraphPad Software, La Jolla, California, USA).
Results
Twenty-six patients were recruited to the study between February and November 2008. In 1 patient, the bag-in-the-lens IOL was sticky and failed to unfold completely after injection into the eye. It was bisected and explanted, and an alternative IOL was implanted. This patient was excluded from the study, and all remaining 25 patients completed 3 months of follow-up, concluding in March 2009. No further surgical complications or adverse events relating to either IOL occurred.
The mean age ± standard deviation was 72.2 ± 10.1 years. Thirty-two percent of participants were male, and 48% of bag-in-the-lens IOLs were implanted into right eyes. Because this was an intraindividual study, the 2 IOL groups were identical in terms of age and gender. Mean preoperative keratometry (average of K1 and K2) was 43.12 ± 1.44 D in bag-in-the-lens IOL eyes versus 43.10 ± 1.44 D in SA60AT IOL eyes ( P = .76), and mean preoperative corneal astigmatism (K1−K2) was 0.63 ± 0.36 D versus 0.76 ± 0.38 D, respectively ( P = .19). Mean preoperative axial length was 23.80 ± 0.91 mm in bag-in-the-lens IOL eyes versus 23.86 ± 0.92 mm in SA60AT IOL eyes ( P = .35). Mean pupil diameter 1 month after surgery was 3.25 ± 0.59 mm in bag-in-the-lens IOL eyes versus 3.15 ± 0.63 mm in SA60AT IOL eyes ( P = .17), and 3 months after surgery, it was 3.28 ± 0.50 mm versus 3.20 ± 0.5 mm, respectively ( P = .10). The mean spherical equivalent refractive error 1 month after surgery was −0.17 ± 0.38 D in bag-in-the-lens IOL eyes versus −0.35 ± 0.42 D in SA60AT IOL eyes ( P = .10), and 3 months after surgery it was −0.11 ± 0.41 D versus −0.31 ± 0.46 D, respectively ( P = .09).
Axial Intraocular Lens Movement
IOL movement results are shown in Table 1 and Figure 3 . Negligible axial movement was observed after physiologic accommodative stimulation with both IOLs. Pilocarpine stimulated a small but clinically insignificant amount of forward axial IOL movement in both groups, and greater movement was observed with the control IOL. Cyclopentolate caused a small amount of backward movement of both IOLs. Control measurements for central corneal thickness and central IOL thickness were consistent under all accommodative conditions ( Table 2 ), indicating that ACD was measured along the same optical path.
| Accommodative Stimulus | IOL | Mean Difference (Bag-in-the-Lens–SA60AT) | P Value | |
|---|---|---|---|---|
| Bag-in-the-Lens | SA60AT | |||
| Physiologic (2 D accommodation) | −5.9 ± 10.3 (−10.0 to −1.9) | −8.4 ± 12.8 (−13.4 to −3.4) | 2.5 ± 13.9 (−2.9 to 8.0) | .37 |
| Pilocarpine | 20.2 ± 165.6 (−44.8 to 85.1) | 50.4 ± 164.4 (−14.0 to 114.8) | 30.2 ± 161.9 (−93.7 to 33.2) | .36 |
| Cyclopentolate | −65.8 ± 64.3 (−91.0 to −40.6) | −54.0 ± 37.5 (−68.7 to −39.3) | −11.8 ± 60.8 (−35.7 to 12.0) | .34 |
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


