Purpose
To investigate the efficacy and safety of 2% rebamipide ophthalmic suspension administered 4 times daily for 52 weeks in patients with dry eye.
Design
Multicenter (17 sites), open-label, single-arm study.
Methods
A total of 154 patients with dry eye were enrolled in this study. After a 2-week screening period, patients received 2% rebamipide, instilled as 1 drop in each eye, 4 times daily for 52 weeks. The signs and symptoms measures were assessed at baseline, at weeks 2 and 4, and at every 4 weeks thereafter. The objective signs were fluorescein corneal staining score, lissamine green conjunctival staining score, and tear film break-up time, while subjective symptoms were dry eye–related ocular symptoms (foreign body sensation, dryness, photophobia, eye pain, and blurred vision). The safety variable was the occurrence of adverse events.
Results
For all objective signs and subjective symptoms, the scores significantly improved at week 2 compared with baseline ( P < .001, paired t test). Interestingly, further improvements of those scores were observed at every visit up to week 52. No deaths were reported, yet serious adverse events that were not thought to be drug related were observed in 6 patients. The incidence of any of the adverse events did not markedly increase throughout the 52-week treatment period.
Conclusion
The results of this study show that 2% rebamipide is effective in improving both the objective signs and subjective symptoms of dry eye patients for at least 52 weeks. In addition, 2% rebamipide treatment was generally well tolerated.
Dry eye is defined by the International Dry Eye Workshop as a multifactorial disease of the tears and ocular surface that results in symptoms of discomfort, visual disturbance, and tear film instability, with potential damage to the ocular surface. It is estimated that dry eye affects up to 35% of the human population worldwide, and it has been reported that the disease is associated with deleterious effects on quality of life and represents a potential substantial economic burden. As the disease is often chronic and thus requires long-term treatment, effective treatments must be tolerable and maintain their efficacy over a long-term period of use.
In healthy eyes, the ocular surface system functions to maintain and protect the refractive surface of the cornea. In patients with dry eye, however, the disease reportedly disrupts components of the ocular surface, thus resulting in tear film instability. The tear film can be destabilized by decreased tear production, delayed clearance, or altered tear composition, ultimately leading to inflammation and exacerbating tear film instability.
Given the importance of the tear film in maintaining a healthy ocular surface, the role of mucins has been a recent focus in dry eye research. Mucins are either expressed on the membranes of ocular surface epithelia or secreted by conjunctival goblet cells, and play a role in lubrication and ocular defense. Reduced amounts of mucins and changes in their distribution and glycosylation have been reported in patients with dry eye, indicating that agents with the ability to increase mucin levels present a therapeutic option for treating the disease.
Rebamipide (Otsuka Pharmaceutical Co, Ltd, Tokyo, Japan) is a mucosal protective agent with mucin secretagogue activity and is marketed in Japan as an oral drug for the treatment of gastric mucosal disorders and gastritis. It has been reported that rebamipide increases the production of mucin-like substances in the cornea and conjunctiva of a rabbit model in which ocular mucin was decreased by N-acetylcysteine, and it has also been shown to increase the density of periodic acid–Schiff reagent–positive cells in the conjunctiva of normal rabbit eyes. In addition, rebamipide reportedly increased MUC1 and MUC4 gene expression in human corneal epithelial cells, promoting glycoconjugate production, an indicator of mucin-like glycoprotein. In a randomized, double-masked, placebo-controlled, 4-week phase II trial, rebamipide ophthalmic suspension (1% and 2%) was found to be significantly more effective than the placebo in improving the fluorescein corneal staining score, the lissamine green conjunctival staining score, and tear film break-up time (TBUT), as well as being well tolerated by the patients and improving their subjective symptoms (foreign body sensation, dryness, photophobia, eye pain, and blurred vision), thus providing them with a positive overall treatment impression. In addition, a phase III study found that 2% rebamipide was effective in improving the objective signs and subjective symptoms of dry eye. Currently, rebamipide ophthalmic suspension has been marketed for the treatment of dry eye since 2012 in Japan.
The objective of this present study was to verify the efficacy and safety of 2% rebamipide ophthalmic suspension for the long-term treatment of dry eye, using objective and subjective outcome measures evaluated up to 52 weeks.
Subjects and Methods
Study Design
This was a multicenter (17 sites), open-label, single-arm study of the efficacy and safety of 2% rebamipide treatment over a 52-week period. The study was performed in compliance with the tenets set forth in the Declaration of Helsinki. The study was conducted according to Good Clinical Practice Guidelines, and the study protocol and informed consent were reviewed and approved by the Institutional Review Boards at 17 Japanese clinical sites before initiation. In addition, informed consent was obtained from all potential patients prior to undergoing prescreening. The trial was registered at www.clinicaltrials.gov prior to patient enrollment (clinical trial identifier: NCT00818324 ).
The study design included a 2-week screening period for each patient to minimize the effects of any eye drops that had been used prior to the screening. During this screening period, preservative-free artificial tears (Soft Santear; Santen Pharmaceutical Co Ltd, Osaka, Japan) were administered 4 times daily. After a baseline examination, eligible patients underwent the administration of 1 drop of 2% rebamipide in each eye 4 times daily for 52 weeks; the use of the Soft Santear was not allowed during this period. The efficacy examinations were conducted at baseline, at weeks 2 and 4, and at every 4 weeks thereafter up to 52 weeks.
Patients
Subjects considered eligible for enrollment in the study were dry eye patients ≥20 years of age. Other inclusion criteria were as follows: (1) a score of ≥2 out of a maximum score of 4 for 1 or more dry eye–related ocular symptoms; (2) a fluorescein corneal staining score ≥3 out of a maximum score of 15; (3) a lissamine green conjunctival staining score ≥5 out of a maximum score of 18; (4) an unanesthetized Schirmer test value at 5 minutes of ≤5 mm or TBUT ≤5 seconds; and (5) a best-corrected visual acuity ≥20/100. Criteria 2-4 needed to be met in the same eye, but not necessarily in both eyes. Criteria 2 and 5 needed to be met at both the screening and baseline examinations, and criteria 1, 3, and 4 needed to be met at the screening examination.
Patients were excluded from the study if they had anterior ocular disease (such as blepharitis or blepharospasm), or if they were unable to discontinue the use of eye drops, currently had a punctal plug or had a punctal plug removed within 3 months prior to the screening examination, or had a history of surgical punctal occlusion. Other exclusion criteria included operation to the ocular surface within 12 months or intraocular surgery within 3 months prior to the screening examination.
The use of the following was prohibited during the study: oral rebamipide, ophthalmic drugs (with the exception of Soft Santear during the screening period), or contact lenses. In addition, ocular surgery or any other treatment affecting the dynamics of tear fluid, including its nasolacrimal drainage process, was prohibited during the study period. Eye drops, including all prescriptions and over-the-counter drugs, and other treatments for adverse events were permitted.
Outcome Measures
Efficacy
Efficacy was evaluated with objective and subjective measures. The objective signs were fluorescein corneal staining score, lissamine green conjunctival staining score, and TBUT. The subjective symptoms were dry eye–related ocular symptoms (foreign body sensation, dryness, photophobia, eye pain, and blurred vision). All of these parameters were assessed at baseline and at weeks 2 and 4, then at every 4 weeks up to week 52, or at discontinuation.
For the fluorescein corneal staining, 5 μL of 2% fluorescein solution (provided by the sponsor) was instilled in the conjunctival sac as the patient was instructed to blink normally. Corneal staining was examined under standard illumination using slit-lamp microscopy. As per the NEI/Industry Workshop report, the cornea was divided into 5 sections, each section was given a staining score from 0-3, and the total score was then calculated. The sponsor provided each investigator with a set of photographs of the fluorescein corneal staining to ensure standardization when scoring.
For the lissamine green conjunctival staining, 20 μL of 1% lissamine green solution (provided by the sponsor) was instilled in the conjunctival sac and the conjunctiva were divided into 6 sections. Conjunctival staining was evaluated under low illumination by slit-lamp microscopy and scored from 0-3 for each section using photographs of lissamine green conjunctival staining provided by the sponsor. The total score was then calculated.
For the TBUT, 5 μL of 2% fluorescein solution was instilled in the conjunctival sac and slit-lamp microscopy was used for evaluation. The time from normal blinking to first appearance of a dry spot in the tear film was measured 3 times.
Dry eye–related ocular symptoms, such as foreign body sensation, dryness, photophobia, eye pain, and blurred vision, were examined by questioning each patient. Answers to the questions regarding the severity of those symptoms were scored according to the following criteria: 0, no symptoms; 1, mild symptoms; 2, moderate symptoms; 3, severe symptoms; 4, very severe symptoms.
Safety
The safety variable was the occurrence of adverse events, determined at each visit by assessment of physical signs and symptoms, external eye examination and slit-lamp microscopy, visual acuity, intraocular pressure, funduscopy, and clinical laboratory tests. Clinical laboratory tests included hematology, biochemistry, and urinalysis.
Statistical Analysis
The efficacy and safety analysis sets comprised patients who received 2% rebamipide at least once. Missing efficacy and safety data were treated as missing data. The eye in which objective efficacy endpoints were analyzed was determined as follows: (1) if only 1 eye met the inclusion criteria, this eye was used; (2) if both eyes met the inclusion criteria, the eye with the higher fluorescein corneal staining score was used; (3) if both eyes had the same fluorescein corneal staining score, the eye with the higher lissamine green conjunctival staining score was used; (4) if both eyes had the same lissamine green conjunctival staining score, the right eye was used. In the analysis of dry eye–related ocular symptoms, patients with a dry eye–related ocular symptom score of 0 at baseline were excluded.
Analysis of change from baseline to each time point was calculated for the fluorescein corneal staining score, lissamine green conjunctival staining score, TBUT, and dry eye–related ocular symptoms. Scores at all visits were compared with baseline using the paired t test. With a target of 100 patients completing the 52-week study, the sample size was set at 153 patients.
Results
Characteristics of the Participants
A total of 154 patients were treated, and 127 of those patients (82.5%) completed the study ( Table 1 ). All 154 patients who received treatment were included in the efficacy and safety analyses. Demographic and other baseline characteristics of the patients are shown in Table 2 . Of the 154 total patients, 15 patients (9.7%) were male and 139 patients (90.3%) were female. The mean age was 59.3 years (range: 24-86 years). Of the 154 patients, 26 patients (16.9%) had primary or secondary Sjögren syndrome and 5 patients (3.2%) had Stevens-Johnson syndrome as the underlying cause of dry eye. At baseline, the most frequent patient-reported symptom was dryness, followed by foreign body sensation.
| 2% Rebamipide n (%) | |
|---|---|
| Number of patients treated | 154 |
| Number of patients completing treatment | 127 (82.5) |
| Number of patients discontinuing treatment | 27 (17.5) |
| -Discontinuation owing to occurrence of adverse events | 22 (14.3) |
| -Discontinuation owing to the patient’s wish | 1 (0.6) |
| -Discontinuation owing to protocol deviation | 2 (1.3) |
| -Discontinuation for other reason | 2 (1.3) |
| 2% Rebamipide (N = 154) n (%) | |
|---|---|
| Sex | |
| Male | 15 (9.7) |
| Female | 139 (90.3) |
| Age | |
| 20-49 years | 37 (24.0) |
| 50-64 years | 53 (34.4) |
| ≥65 years | 64 (41.6) |
| Main cause or primary disease of dry eye | |
| Primary Sjögren syndrome | 21 (13.6) |
| Secondary Sjögren syndrome | 5 (3.2) |
| Stevens-Johnson syndrome | 5 (3.2) |
| Unknown | 123 (79.9) |
| Fluorescein corneal staining score a | |
| 3-6 | 91 (59.1) |
| 7-9 | 36 (23.4) |
| 10-15 | 27 (17.5) |
| Dry eye–related ocular symptoms a | |
| Foreign body sensation | 120 (77.9) |
| Dryness | 145 (94.2) |
| Photophobia | 98 (63.6) |
| Eye pain | 90 (58.4) |
| Blurred vision | 88 (57.1) |
Efficacy Evaluation
Objective signs
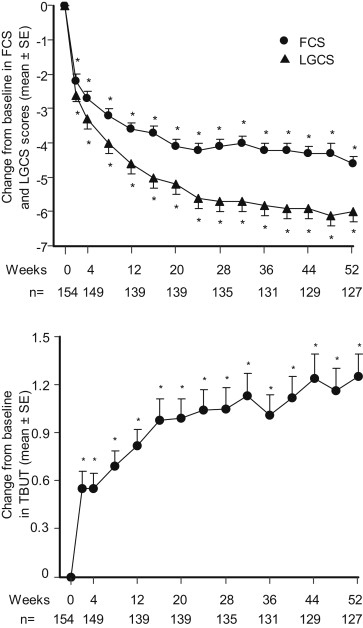
At baseline, the mean fluorescein corneal staining score was 6.6. The mean fluorescein corneal staining score at week 2 (4.3) was significantly decreased compared with that at baseline ( P < .001, paired t test), and further improvements were observed at almost every visit up to 52 weeks (1.9 at week 52) ( Figure 1 ). The mean lissamine green conjunctival staining score at baseline was 9.4. At week 2, the mean lissamine green conjunctival staining score (6.8) was significantly decreased compared with that at baseline ( P < .001, paired t test), and further improvements were observed at every visit up to 52 weeks (3.2 at week 52) ( Figure 1 ). Figure 2 shows the fluorescein corneal staining and lissamine green conjunctival staining photographs at baseline and at week 52, in which corneal and conjunctival disorders were improved.
Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


