6 Radiologic Evaluation of Hemangiomas
6.1 Introduction
Imaging features of vascular lesions generally reflect their histologic composition. The primary histologic features of hemangiomas, namely, endothelial cell proliferation and hypercellularity, are associated with imaging features compatible with a cellular tumor. Vascular malformations, on the other hand, are characterized by relative low cellularity and prominent vascular channels; these features are similarly evident in their appearance on imaging studies. The various cellular subtypes of vascular malformations display adequately diverse imaging appearances to allow their differentiation in most instances.
Although clinical features may allow accurate diagnosis of vascular birthmarks in many cases, imaging may prove helpful in cases where physical findings are less specific and there is concern for alternate diagnosis, when there is potential proximity of the lesion to critical structures, and when there is a need to assess the results of therapeutic intervention. 1 In particular, when an infant has a soft tissue mass, the possibility of an aggressive process, such as a malignant neoplasm, must be at least summarily considered. Imaging may prove comforting to clinicians and family in ruling out entities such as sarcomas in this demographically appropriate cohort.
Available imaging studies are ultrasonography, computed tomography (CT), magnetic resonance imaging (MRI), and arteriography. Ultrasound is a fast, inexpensive, and noninvasive modality that typically can be performed without sedation. Ultrasound has greatest utility during the proliferative phase ( Fig. 6.1 ). 1

High vessel density, rare arteriovenous shunting, and high peak arterial Doppler shift help distinguish hemangiomas from other superficial soft tissue masses, including tumors and vasoformative lesions, 2 , 3 although Paltiel et al demonstrated that the presence of a soft tissue mass is the only multivariate predictor that differentiates hemangiomas from arteriovenous malformations (AVMs). Limitations of ultrasound include the small field of view, restricted depth of penetration, and difficulty detecting small vessels with low flow. 4 Ultrasound is thus felt to be a useful initial screening procedure in patients with soft tissue lesions, when distinction between the various possible diagnoses will influence prognosis and treatment options.
When more precise lesional mapping and characterization are necessary, cross-sectional imaging may be used. Although MRI, because of greater soft tissue discrimination and absent ionizing radiation, is generally considered the study of choice, multidetector CT (MDCT) is currently the cheapest and most expeditious available modality and often can provide sufficient information for both accurate diagnosis and mapping of hemangiomas. 5 Full MDCT studies may take only minutes, obviating the need for sedation in many cases, and the single-acquisition data can be reconstructed in a variety of planes and tissue algorithms. Hemangiomas typically appear as well-defined, multilobular masses that are isodense to muscle on noncontrast studies. During the proliferative phase, when most lesions are imaged, enhancement is intense, and lesion conspicuity is excellent ( Fig. 6.2 ). After intravenous contrast administration, there is vivid contrast enhancement of the mass, often accompanied by enhancement of enlarged feeding and draining vessels. During involutional phases, there is a gradual increase in low-density fatty content of the mass, with a corresponding decrease in the degree of contrast enhancement. 6 When the lesion has fully involuted, the only visible finding may be contour asymmetry of the affected region, which may contain a relative abundance of fatty tissue.

Both calcification and osseous erosion are infrequent in hemangioma, although when present these are better demonstrated on CT than on MRI. Prolonged hyperemia may occasionally result in bony overgrowth, and the resulting skeletal asymmetry is demonstrated to best advantage on CT imaging; three-dimensional reconstructions may be useful in treatment planning.
The morphologic and enhancement characteristics of hemangiomas are similar on MRI, with lesions typically isointense to skeletal muscle on T1-weighted imaging. On T2-weighted imaging, hemangiomas are moderately hyperintense relative to skeletal muscle but hypointense relative to cerebrospinal fluid or vitreous humor. This feature reflects the highly cellular nature of these neoplasms and is seen in other hypercellular masses such as lymphoma and meningioma. During the proliferative phase, voids in internal vascular flow may be seen on both T1- and T2-weighted imaging, accompanied by enlargement of flow voids related to feeding and draining vessels ( Fig. 6.3 ).
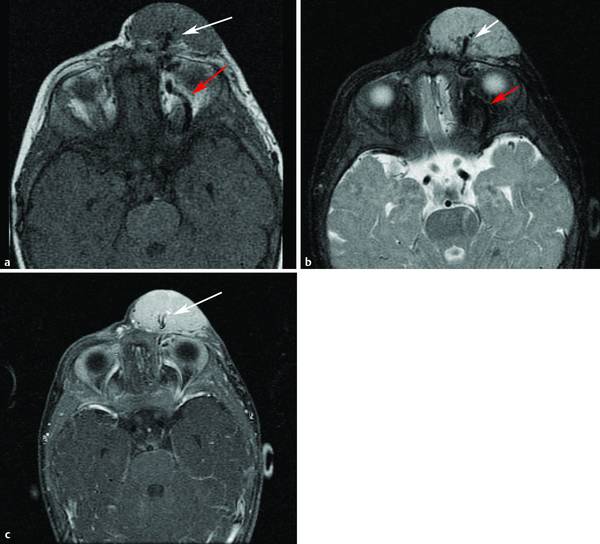
As on CT, involution is characterized by gradual replacement of the parenchyma by fat, with increasing heterogeneity and T1 shortening, and decreasing contrast enhancement ( Fig. 6.4 , Fig. 6.5 , Fig. 6.6 ). The number and size of associated vascular flow voids will similarly gradually diminish.



Perhaps the greatest advantage of MRI over other imaging modalities is its superior tissue contrast and ability to map lesional extent accurately, which is particularly important with regard to critical areas such as the brain, orbit, and airway. Although involvement of the neuraxis by infantile hemangioma occurs in only 1% of cases, 7 , 8 MRI may also detect associated intracranial abnormalities such as those that occur in PHACES (posterior fossae anomalies, hemangioma, arterial anomalies, cardiac and cerebrovascular anomalies, eye anomalies, and midline sternal or supraumbilical defects) syndrome, including cerebellar hypoplasia and arterial stenosis ( Fig. 6.7 ). 9

The periorbital region is a common site for hemangiomas, and treatment is often indicated because of disturbances of vision ( Fig. 6.8 ). 10 , 11 It is particularly important to recognize even small volumes of retrobulbar disease before any planned intervention ( Fig. 6.9 ). Similarly, recognition of involvement of the upper aerodigestive tract is of critical importance because the swelling that may occur during therapy can result in airway compromise.


Magnetic resonance protocols for imaging-suspected hemangiomas should include both precontrast and postcontrast T1-weighted images, the latter performed with fat suppression. Lesions are often most conspicuous on T2-weighted imaging, with fat suppression accomplished either by chemical fat saturation or inversion recovery techniques. Sequences are performed in at least two planes; axial and coronal scans are typically acquired. The field of view and scan range should be broad enough to cover the lesion in toto and to assess adjacent areas of the intracranial and intraorbital compartments. Whole-brain imaging is frequently performed using enhanced T1-weighted imaging in a separate acquisition. Whereas magnetic resonance imaging may demonstrate intralesional vascularity, as well as feeding and draining vessels, it is not routinely used at many institutions as neither diagnosis nor management are significantly impacted. An exception to this occurs in PHACES syndrome because of the characteristic intracranial or extracranial sites of arterial stenosis ( Fig. 6.10 ).

With regard to congenital hemangiomas, some differences in imaging features have been described. Gorincour et al evaluated a group of 26 suspected congenital hemangiomas consisting of eight rapidly involuting congenital hemangiomas (RICHs) and 18 noninvoluting congenital hemangiomas (NICHs) and compared them with a group of 26 common hemangiomas. 12 On ultrasound, 72% of NICHs and 62.5% of RICHs were found to be heterogeneous, compared with 42.3% of common hemangiomas. Vessels were visible in 72% of NICHs and 62.5% of RICHs versus 15.4% of common lesions, and calcifications were identified in 17% of NICHs and 37.5% of RICHs; no calcification was visible in any of the common lesions. On CT or MRI, 100% of the common lesions displayed well-defined limits, compared with 67% of NICHs and 60% of RICHs ( Fig. 6.11 ). Significant differences were also seen in the incidence of fat stranding (29.4% of NICHs and RICHs versus 7.7% of common lesions).

In addition to distinguishing common infantile hemangiomas from their congenital cousins, imaging may be called on to differentiate hemangiomas from other vascular lesions of the head and neck. 13 The distinction between hemangiomas and vascular malformations can often be made quite easily based on MRI characteristics. A lesion that demonstrates intense enhancement may represent a hemangioma or a venous malformation (VM); however, these can usually be distinguished by their T2 signal characteristics, with only moderate hyperintensity in hemangioma and marked hyperintensity in VMs. Lymphatic malformations do not typically enhance and are associated with high signal on T2-weighted sequences in their macrocystic form. Whereas AVMs are characterized by numerous internal flow voids and enlarged feeding and draining vessels, they do not have the well-defined parenchymal masses that are associated with hemangiomas and typically enhance to a much lesser extent.
Distinguishing between hemangiomas and malignant soft tissue masses of the head and neck, typically rhabdomyosarcomas, may be a more difficult endeavor. 14 Teo et al found that although no single MRI feature could reliably distinguish between these entities, a combination of morphologic features and signal characteristics could aid in differentiation. 15 Specifically, lobulation, septation, and central signal voids were all significantly more common in hemangioma, and when all were present, a confident diagnosis was possible. Both dynamic contrast-enhanced and diffusion-weighted imaging have been shown to be of some value in distinguishing benign from malignant soft tissue masses. 16 , 17 In general, the combination of a poorly defined soft tissue mass with an aggressive pattern of bone destruction favors a malignant process such as rhabdomyosarcoma or metastatic neuroblastoma ( Fig. 6.12 ).

A benign but locally aggressive process that can be clinically confused with hemangioma is infantile fibromatosis. 18 These nodular lesions are often associated with nonspecific imaging features but may present a characteristic “target” appearance that reflects internal hemorrhage and necrosis. Other benign soft tissue tumors or pseudotumors may occur in association with a predisposing condition, for example, plexiform neurofibroma in neurofibromatosis type I ( Fig. 6.13 ). 19 In general, correlation with clinical features is essential for accurate interpretation of imaging findings.

Stay updated, free articles. Join our Telegram channel

Full access? Get Clinical Tree


