Purpose
To investigate β-amyloid precursor protein (β-APP), ubiquitin, and glial fibrillary acid protein (GFAP) immunostaining as a diagnostic tool to aid in the discrimination between abusive head trauma and nonabusive head trauma in postmortem ocular histopathologic investigation.
Design
Retrospective cross-sectional study.
Methods
Seventy-four eyes of 37 infants were studied for hemorrhage and immunohistochemical expression of β-APP, ubiquitin, and GFAP in the retina and optic nerve. Infants were assigned to abusive head trauma or control groups, according to published criteria.
Results
In the abusive head trauma group, positive β-APP and ubiquitin immunostaining of the retina was significantly more likely to be found than in the control group, odds ratio (OR) 11.4, confidence interval (CI) 2.9–44.3; P < .001 and OR 8.8, CI 2.2–34.5; P = .002, respectively. Positive correlations were found between retinal expression of β-APP and ubiquitin immunostaining and retinal hemorrhage. Vitreal hemorrhages, orbital fat hemorrhages, and macular folds could only be identified in abusive head trauma cases. Retinal hemorrhages were significantly more severe, occupied a larger proportion of the retina, and involved more retinal layers in abusive head trauma compared to controls (OR 2.7, CI 1.7–4.4; P < .001).
Conclusions
This study shows correlations between positive retinal β-APP and ubiquitin immunostaining as a sign of axonal injury in abusive head trauma. Axonal injury is a useful pathologic feature that can be demonstrated in postmortem ocular investigation of deceased children using immunohistochemical staining for β-APP and ubiquitin with a high OR for abusive head trauma when compared to controls.
Abusive head trauma is a form of physical child abuse, characterized by repetitive acceleration-deceleration forces with or without blunt trauma. Characteristic findings for abusive head trauma are different combinations of subdural hematoma, subarachnoid hemorrhage, encephalopathy, retinal hemorrhages, and fractures of the skull, long bones, and ribs. Approximately 30% of the abusive head trauma victims die and approximately 80% of the survivors suffer permanent damage, such as visual and/or neurologic impairment.
Retinal hemorrhage can be found in approximately 85% of the victims, ranging from 50% to 100%, dependent on the study population. However, retinal hemorrhage is not pathognomonic for abusive head trauma, since this can be found in about 20% of neonates. Retinal hemorrhage can be observed in a variety of diseases; the differential diagnosis is described in multiple reviews.
Immunohistochemistry may be used to detect ocular damage in addition to standard histologic evaluation for postmortem study of the eye in cases of suspected abusive head trauma. Axonal injury is the damage of the axons owing to any etiology (ischemia or other pathologic conditions); it has no specific biomechanical relevance as such, and may be perceived focally as a local isolated phenomenon. Axonal bulbs may be observed as a histopathologic hallmark of severe axonal damage. The expression of β-amyloid precursor protein (β-APP) and ubiquitin can reveal axonal injury in the brain after head trauma. Additionally, β-APP immunohistochemistry can be used to reveal axonal injury to the optic nerves and to the retinal nerve fiber layer of abusive head trauma victims. In this retrospective cross-sectional study, we evaluated classical histologic parameters and immunohistochemistry for β-APP, ubiquitin, and glial fibrillary acid protein (GFAP) in the eyes of the subjects, in order to confirm that there is a distinctive pattern of β-APP and ubiquitin immunostaining in the abusive head trauma group compared to the nonabusive head trauma group, implying that β-APP could be applied as an additional tool to distinguish axonal injuries caused by abusive head trauma from nonabusive head trauma.
Methods
The study design was a retrospective cross-sectional study. Cases were randomly selected from 48 infants brought for postmortem investigation to the Netherlands Forensic Institute in The Hague, having a complete evaluation of the eyes and optic nerves, completed with 2 preterm neonates (29+2 and 34 weeks) and 1 infant that died in the Pediatric Department of the Erasmus University Medical Center in Rotterdam to complement the control group. Complete autopsy files were present for each infant, together with a toxicology report, a microbial report, and a metabolic report for all the forensic subjects. The autopsy files, forensic reports, and police investigations were thoroughly studied in order to classify the cause and manner of death. Because of the retrospective postmortem nature of the study, no prior informed consent could be obtained. The Institutional Review Boards of the Netherlands Forensic Institute and the Erasmus University Medical Center waived approval because of the retrospective and noninterventional character of the study. The study was in adherence to the tenets of the Declaration of Helsinki.
The study population was divided into an abusive head trauma group and a nonabusive head trauma control group to assess diagnostic features of the histologic measurements. Maguire and associates showed that for an infant with intracranial injury and either retinal hemorrhage or rib fractures, and any of the other mentioned features (apnea, skull fractures, long bone fractures, seizures, and head and/or neck bruising), the odds ratio (OR) for abusive head trauma is >100. Inclusion criteria for the abusive head trauma group were based on this article; an infant was placed in the abusive head trauma group when the OR of abusive head trauma was >100. Exclusion criteria were identifiable factors that could have influenced bleeding tendency, incomplete medical files, age at the time of death (over 3 years of age), postmortem changes that prevented histopathologic evaluation, and trauma due to impact only. Inclusion criteria for the control group were death not caused by shaken and impact trauma. In total, 37 infants were included in the study. We studied the postmortem eyes of 21 abusive head trauma cases and 16 controls. There was no significant difference in sex or age between the 2 groups. In order to allow correlations to be calculated safely, we chose to match the 4 prematurely born infants among the abusive head trauma cases to 4 prematurely born infants among the control cases. Clinical and forensic information of the abusive head trauma cases is summarized in Table 1 . Table 2 summarizes the clinical data of the control group.
| Case (Sex) | Age (Days) | Type of Trauma | Retinal Hemorrhage | Intracranial Hemorrhage | Fractures | Bruises | Trauma-Death | Cardiopulmonary Resuscitation |
|---|---|---|---|---|---|---|---|---|
| 1 (M) | 102 | ADT | B | SDH | – | – | 72 h | + |
| 2 (F) | 20 | ADT | B | SDH | – | – | 72 h | + |
| 3 (M) | 214 | ADT + impact | B | SDH | – | H | n.a. | + |
| 4 (M) | 239 | ADT | B | SDH | – | – | 24 h | + |
| 5 (M) | 1041 | ADT + impact | B | SDH, SAH | – | HBoE | 48 h | + |
| 6 (M) | 103 | ADT + impact | B | SDH, SAH | 2 old rib # | H | 21 h | + |
| 7 (M) | 54 | ADT | B | SDH | At least 3 old rib # | – | 24 h | + |
| 8 (M) | 122 | ADT + impact | B | SDH | – | HBo | 13 h | + |
| 9 (M) | 65 | ADT + impact | B | SDH, SAH | – | HBo | 3 h | + |
| 10 (M) | 25 | ADT + impact | B | SDH | Skull #, 3 rib # | HE | 18.5 h | + |
| 11 (F) | 10 | ADT + impact | B | SDH, SAH | – | HBo | 17 h | + |
| 12 (M) | 572 | ADT | B | SDH | – | – a | 27 h | + |
| 13 (M) | 33 | ADT + impact | U | SDH, SAH | – | H | 192 h | + |
| 14 (M) | 254 | ADT + impact | B | SDH, SAH | – | HE | 73.5 h | + |
| 15 (F) | 94 | ADT + impact | U | SDH, SAH | Skull #, 4-5 rib # | HBo | 24 h | + |
| 16 (F) | 693 | ADT + impact | B | SDH, SAH | – | HBo | 5 h | + |
| 17 (F) | 152 | ADT + impact | B | SDH, SAH | Several rib #, humerus #, scapula # | HBoE | 96 h | + |
| 18 (F) | 122 | ADT + impact | B | SDH | Clavicle # | HBoE | 2.5 h | – |
| 19 (M) | 201 | ADT + impact | U | SDH, SAH | Skull # | BoE | 17.5 h | + |
| 20 (F) | 137 | ADT + impact | B | SDH, SAH | – | HBo | 168 h | + |
| 21 (M) | 56 | ADT + impact | B | SDH, SAH | – | H | 24 h | + |
a Several scars were present, considered to be probably from cigarette burns, and 2 larger scars, possibly from hot water.
| Case (Sex) | Age (Days) | Cause of Death | Retinal Hemorrhage | Intracranial Hemorrhage | Fractures | Bruises | Presentation-Death | Cardiopulmonary Resustication |
|---|---|---|---|---|---|---|---|---|
| 22 (F) | 122 | Pneumonia | – | – | – | – | 48 h | + |
| 23 (F) | 81 | Prematurity | U | SDH, SAH | – | – | 12 h | + |
| 24 (F) | 183 | Pneumonia | B | – | – | – | 16 h | + |
| 25 (F) | 58 | Pneumonia | – | – | Femur # (4–6 weeks old) | – | <1.5 h | – |
| 26 (M) | 29 | Coagulation disorder | – | SDH, SAH | – | Bo | 7 h | + |
| 27 (F) | 20 | SIDS | – | – | – | – | 70 h | + |
| 28 (M) | 59 | SIDS | – | – | – | – | 9 h | + |
| 29 (F) | 1 | Stab injuries | – | – | – | – a | 24 h | + |
| 30 (F) | 544 | SIDS | – | – | – | – | <11 h | – |
| 31 (M) | 304 | Suffocation | – | – | – | HBo | 1.5 h | + |
| 32 (M) | 44 | Endomyocarditis | B | – | – | – | <6.5 h | – |
| 33 (F) | 0 | Pneumothorax | B | – | – | Bo | n.a. | – |
| 34 (M) | 0 | SIDS | – | – | – | – | n.a. | – |
| 35 (M) | 0 | Congenital anomalies | B | – | – | – | 0.75 h | – |
| 36 (M) | 0 | Congenital anomalies | – | – | – | – | 1 h | – |
| 37 (F) | 108 | Cardiomyopathy | B | – | – | – | n.a. | + |
Specific guidelines for postmortem protocol for ocular investigation were followed. Before sectioning, the eyes and the optic nerves were grossly studied for the presence of hemorrhages. Axial sections were cut following the pupil–optic nerve method, transilluminated, and inspected for retinal hemorrhage, retinoschisis, and macular folds using a dissection microscope. The histologic sections were microscopically studied by 2 different researchers (B.B. and R.M.V.) for the presence of hemorrhages in the conjunctiva, perioptic orbital fat, extraocular muscle tissue, sclera, subdural and subarachnoid layer in the optic nerve, retina, and the vitreous body. When hemorrhages were found in the retina, the affected layer was specified as the nerve fiber layer, the inner plexiform layer, the inner nuclear layer, the outer plexiform layer, the outer nuclear layer, and the photoreceptor layer. It was noted whether retinal hemorrhage was present in the periphery, the equator, or the posterior pole and if they were slight (+), moderate (++), or extensive (+++), in a comparable manner as would be done at clinical evaluation. An estimation was made of the percentage of the affected retina. The sections were evaluated for the presence of hemosiderin, as an indication for earlier hemorrhages.
The immunohistochemical sections (β-APP, ubiquitin, and GFAP) were studied for the presence of positive staining in the different layers and parts of the retina. If the optic nerves were stained for β-APP, the scoring method was adapted according to Reichard and associates, scoring slight and pinpoint (+), moderate (++), or extensive and widespread (+++) staining. Sections stained for β-APP were studied for the presence of axonal bulbs in the optic nerve.
The paraffin-embedded blocks of the eyes and the optic nerves were cut at 4 μm sections and stained with hematoxylin-eosin and Prussian blue for hemosiderin. For immunohistochemistry, following deparaffinization and heat-induced antigen retrieval, the tissue sections were incubated with primary antibody β-APP (polyclonal rabbit antibody; Thermo Fisher Scientific, Fremont, California, USA; dilution 1:100), ubiquitin (monoclonal mouse antibody, clone FPM1; Novocastra Laboratories, Newcastle, UK; dilution 1:400), and GFAP (monoclonal mouse antibody, clone 6F2; DakoCytomation, Glostrup, Denmark; dilution 1:400) for 30 minutes at room temperature. A subsequent amplification step for β-APP and GFAP was followed by application of the diaminobenzidine detection kit (DakoCytomation) according to the manufacturer’s instructions. The slides were counterstained with hematoxylin. Appropriate negative and positive control tissues were used as an external validation of the staining procedures.
The software package IBM SPSS Statistics 20.0 (IBM Corporation, Armonk, New York, USA) was used to perform the statistical analyses. The independent-samples t test was applied for testing the difference between the infant ages in the abusive head trauma group and the controls, for comparing the percentages of retinal hemorrhage area between the 2 groups. The Mann-Whitney U test was used to test the difference in affected retina between the 2 groups. Fisher exact test was used to test the association of several categorical variables with abusive head trauma, by obtaining abusive head trauma/nonabusive head trauma OR from the contingency tables including sex, retinal hemorrhage, optic nerve sheath hemorrhage, intrascleral peripapillary hemorrhage, vitreal hemorrhage, orbital fat and muscle tissue hemorrhage, macular fold, hemosiderin, GFAP immunoreactivity, ubiquitin immunoreactivity in the retina and the optic nerve, β-APP immunoreactivity in the retina and the optic nerve, and axonal bulbs. The ad hoc approach of adding 0.5 to each cell was used to deal with unstructural zeros within the tables. In addition, we tested the association of different aspects of retinal hemorrhage with abusive head trauma, such as the separate retinal layers and the separate retinal divisions. This was also done for ubiquitin and β-APP immunoreactivity. Owing to the sparseness of the data, Spearman rank-order correlation was used to test the correlation between retinal ubiquitin immunoreactivity and retinal hemorrhage, the correlation between retinal β-APP immunoreactivity and different histologic findings (eg, intrascleral peripapillary hemorrhages), the correlation between retinal hemorrhage and β-APP immunoreactivity in different divisions of the retina, the correlation between retinal β-APP and ubiquitin immunoreactivity, the correlation between β-APP optic nerve immunoreactivity and histologic findings (eg, macular fold), and the correlation between axonal bulbs and histologic findings (eg, optic nerve sheath hemorrhages). Significance was defined as P < .005. To decrease the risks for multiple testing, only those correlations were tested that might be pathobiologically explained.
Results
Histopathologic findings of the ocular hemorrhages are given in Table 3 . More detailed data on the severity of the retinal hemorrhage and the affected retinal layers and regions can be found in Supplementary Table 1 (supplemental material available at AJO.com ). In total, 74 eyes were studied, 42 of abusive head trauma cases and 32 controls, among which 50 showed retinal hemorrhage. The average percentage of the affected retina was 23% (standard deviation [SD] 34.53). Retinal hemorrhage was found in 39 of 42 abusive head trauma eyes and in 11 of 32 control eyes (OR 2.7 (confidence interval [CI] 1.7–4.4); P < .001). All statistically significant findings regarding postmortem ocular histopathology in abusive head trauma and comparison with controls are summarized in Table 4 .
| Case | Retinal Hemorrhage | Percentage of Affected Retina | Subdural Optic Nerve Sheath Hemorrhage | Subarachnoidal Optic Nerve Sheath Hemorrhage | Intrascleral Peripapillary Hemorrhage | Vitreal Hemorrhage | Orbital Fat Hemorrhage | Macular Fold | Hemosiderin | |
|---|---|---|---|---|---|---|---|---|---|---|
| Left | Right | |||||||||
| 1 (A) | L/R | 80 | 80 | L/R | L/R | L/R | L/R | L | L/R | – |
| 2 (A) | L/R | 5 | 80 | L/R | L/R | L/R | R | – | – | L/n.a. |
| 3 (A) | L/R | 50 | 90 | L/R | L/R | L/R | L/R | – | L | – |
| 4 (A) | L/R | 85 | 45 | L/R | L/R | L/R | L/R | – | L/R | – |
| 5 (A) | L/R | 5 | 5 | L/R | – | L | L | – | – | – |
| 6 (A) | L/R | 90 | 90 | L/R | L/R | L/R | L/R | L/R | L/R | – |
| 7 (A) | L/R | 80 | 5 | L/R | L/R | L | L/R | L/R | – | L/R |
| 8 (A) | L/R | 100 | 100 | L/R | L/R | L/R | L/R | L/R | L/R | – |
| 9 (A) | L/R | 5 | 95 | L/R | R | L/R | R | – | R | R |
| 10 (A) | L/R | 20 | 20 | L/R | L/R | L/R | L/R | L/R | L/R | – |
| 11 (A) | L/R | 80 | 100 | L/R | L/R | L/R | L/R | R | – | – |
| 12 (A) | L/R | 5 | 50 | L/R | L/R | L/R | L/R | – | R | – |
| 13 (A) | L | 10 | 0 | – | – | – | L | L/R | – | L |
| 14 (A) | L/R | 5 | 5 | L/R | L/R | L/R | L/R | R | – | R |
| 15 (A) | R | 0 | 5 | L/R | R | R | R | R | R | – |
| 16 (A) | L/R | 90 | 90 | L/R | L/R | L/R | L/R | L/R | L | L/R |
| 17 (A) | L/R | 5 | 5 | L/R | L/R | L/R | L/R | R | – | – |
| 18 (A) | L/R | 20 | 5 | L/R | L | L | L | L/R | – | R |
| 19 (A) | L | 5 | 0 | L/R | – | – | – | – | – | – |
| 20 (A) | L/R | 5 | 5 | L/R | L/R | – | L/R | – | – | L/R |
| 21 (A) | L/R | 20 | 20 | L/R | L/R | R | L/R | L/R | L/R | L/R |
| 22 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 23 (C) | R | 0 | 1 | L/R | – | – | – | – | – | – |
| 24 (C) | L/R | 5 | 5 | – | – | – | – | – | – | L |
| 25 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 26 (C) | – | 0 | 0 | L | L | – | – | – | – | – |
| 27 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 28 (C) | – | 0 | 0 | – | – | – | – | – | – | L/R |
| 29 (C) | – | 0 | 0 | – | – | – | – | – | – | n.a./n.a. |
| 30 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 31 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 32 (C) | L/R | 5 | 5 | – | – | – | – | – | – | L/R |
| 33 (C) | L/R | 5 | 5 | L/R | – | L/R | – | – | – | – |
| 34 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 35 (C) | L/R | 5 | 5 | – | – | – | – | – | – | – |
| 36 (C) | – | 0 | 0 | – | – | – | – | – | – | – |
| 37 (C) | L/R | 10 | 5 | – | – | – | – | – | – | – |
| Abusive Head Trauma | Control | Odds Ratio | Confidence Interval a | |
|---|---|---|---|---|
| Retinal hemorrhage present | 39/42 | 11/32 | 2.7 | 1.7–4.4 |
| Moderate and extensive retinal hemorrhage | 29/42 | 0/32 | 57.3 | 3.6–921.2 |
| Percentage of affected retina | 40% (SD 39.09) | 2% (SD 2.71) | P < 0.001 | |
| All retinal regions involved | 35/42 | 4/32 | 4.1 | 2.8–6.0 |
| Peripheral retina | 37/42 | 8/32 | 3.5 | 1.9–6.5 |
| Equatorial retina | 37/42 | 7/32 | 4.0 | 2.0–7.8 |
| Posterior retina | 38/42 | 6/32 | 4.8 | 2.3–10.0 |
| Hemorrhage in all retinal layers | 35/42 | 0/32 | 54.5 | 3.5–855.9 |
| Optic nerve sheath hemorrhage present | 40/42 | 6/32 | 5.1 | 2.5–10.5 |
| Bilateral subdural optic nerve sheath hemorrhage | 20/21 | 2/16 | 7.6 | 2.1–28.0 |
| Bilateral subarachnoidal optic nerve sheath hemorrhage | 15/21 | 0/16 | 24.0 | 1.5–372.5 |
| Intrascleral peripapillary hemorrhage | 31/42 | 2/32 | 11.8 | 3.1–45.7 |
| Bilateral intrascleral peripapillary hemorrhage | 13/21 | 1/16 | 9.9 | 1.4–68.1 |
| Vitreal hemorrhage | 34/42 | 0/32 | 53.0 | 3.4–832.3 |
| Orbital fat hemorrhage | 21/42 | 0/32 | 33.0 | 2.1–525.0 |
| Macular fold | 17/42 | 0/32 | 26.9 | 1.7–430.5 |
A coloboma was found in 1 eye (abusive head trauma Case 2), extramedullary hematopoiesis was found in 3 eyes (abusive head trauma Case 7 and control Cases 26 and 33), and retinopathy of prematurity was found in 4 eyes (control Cases 23 and 35).
Table 5 shows the positive β-APP immunostaining in the retina and optic nerve and the presence of axonal bulbs in the optic nerve. A more detailed overview of β-APP immunostaining can be found in Supplementary Table 2 (supplemental material available at AJO.com ). There was no significant difference in the time interval between presentation to the medical services and death for β-APP-positive vs β-APP-negative infants.
| Case | Retina β-Amyloid Precursor Protein | Retina Ubiquitin | Peripapillary β-Amyloid Precursor Protein | Peripapillary Ubiquitin | Optic Nerve β-Amyloid Precursor Protein | Optic Nerve Axonal Bulbs | Retina Glial Fibrillary Acid Protein | ||
|---|---|---|---|---|---|---|---|---|---|
| Left | Right | Left | Right | ||||||
| 1 (A) | L/R | L/R | L/R | L/R | – | + | – | – | – |
| 2 (A) | R | L/R | R | R | n.a. | n.a. | n.a. | n.a. | – |
| 3 (A) | R | L/R | L/R | – | + | n.a. | – | n.a. | – |
| 4 (A) | L/R | R | L/R | L/R | n.a. | – | n.a. | – | – |
| 5 (A) | – | – | L/R | – | n.a. | n.a. | n.a. | n.a. | – |
| 6 (A) | L/R | – | L/R | L | +++ | +++ | – | – | – |
| 7 (A) | L/R | L | L/R | L | ++ | ++ | – | – | – |
| 8 (A) | L/R | L/R | L/R | L/n.a. | + | + | – | + | – |
| 9 (A) | L/R | – | L/R | L | + | + | + | – | – |
| 10 (A) | L/R | L/R | L/R | L/R | ++ | n.a. | – | n.a. | – |
| 11 (A) | L/R | – | L/R | – | + | +++ | – | + | – |
| 12 (A) | L/R | L | L/R | – | ++ | n.a. | + | n.a. | L |
| 13 (A) | – | L | L | L | + | – | – | – | L |
| 14 (A) | L/R | – | L/R | L/R | – | – | – | – | L/R |
| 15 (A) | – | – | R | – | – | n.a. | – | n.a. | – |
| 16 (A) | L/R | R | L/R | L/R | n.a. | n.a. | n.a. | n.a. | L/R |
| 17 (A) | – | – | – | – | n.a. | n.a. | n.a. | n.a. | L |
| 18 (A) | – | – | L | – | – | – | – | – | L |
| 19 (A) | – | – | – | – | n.a. | – | – | – | R |
| 20 (A) | – | – | – | – | – | n.a. | – | – | L/R |
| 21 (A) | – | – | – | – | n.a. | n.a. | – | – | L/R |
| 22 (C) | – | – | – | – | + | n.a. | – | n.a. | – |
| 23 (C) | – | – | – | – | n.a. | n.a. | – | – | – |
| 24 (C) | L | L | L | – | n.a. | – | n.a. | – | R |
| 25 (C) | – | – | – | – | + | + | + | + | – |
| 26 (C) | – | – | – | – | – | – | – | – | – |
| 27 (C) | – | – | – | – | n.a. | – | n.a. | – | L/R |
| 28 (C) | – | – | – | – | n.a. | – | – | – | L/R |
| 29 (C) | – | – | – | – | – | – | – | – | – |
| 30 (C) | – | – | – | – | – | – | – | – | – |
| 31 (C) | – | – | – | – | n.a. | – | – | – | – |
| 32 (C) | – | – | – | – | – | – | – | – | L/R |
| 33 (C) | – | – | – | – | – | – | – | – | – |
| 34 (C) | – | – | – | – | – | – | – | – | – |
| 35 (C) | L | – | – | – | + | – | – | – | – |
| 36 (C) | – | – | – | R | n.a. | – | – | – | – |
| 37 (C) | – | – | – | – | + | + | – | – | – |
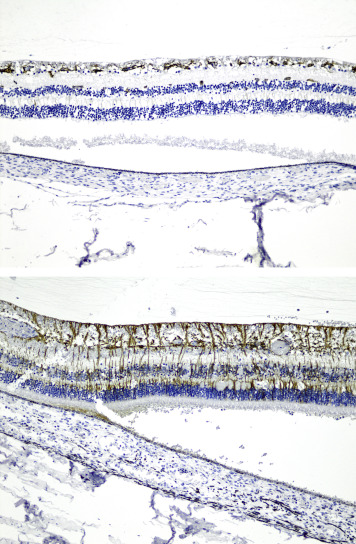
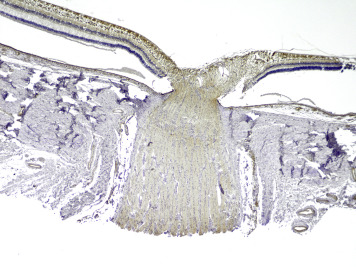
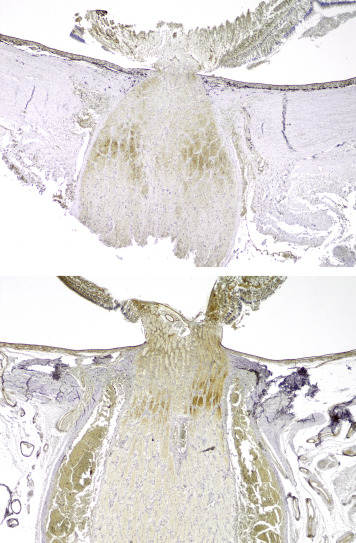
In total, retinal β-APP immunostaining was detected in 32 of 74 eyes studied. In the abusive head trauma group, retinal β-APP immunostaining was found in 30 of 42 eyes ( Figure 1 ), while it was found in only 2 of 32 control eyes (OR 11.4 [CI 2.9–44.3]; P < .001). Positive immunostaining was identified mainly in the nerve fiber layer and the inner plexiform layer, but was also observed in the other retinal layers in 2 cases of abusive head trauma. There was a significant correlation between positive retinal staining and the percentage of the retina covered by hemorrhage (r = 0.797, P < .001) ( Figure 2 ), vitreal hemorrhage (r = 0.735, P < .001), intrascleral peripapillary hemorrhage (r = 0.736, P < .001), and macular fold (r = 0.581, P < .001). When positive, almost all cases showed β-APP positivity in the peripapillary region ( Figure 3 , Top). Of the 32 eyes with positive retinal β-APP immunostaining, only 1 did not show positive staining in the peripapillary region (control case 35).
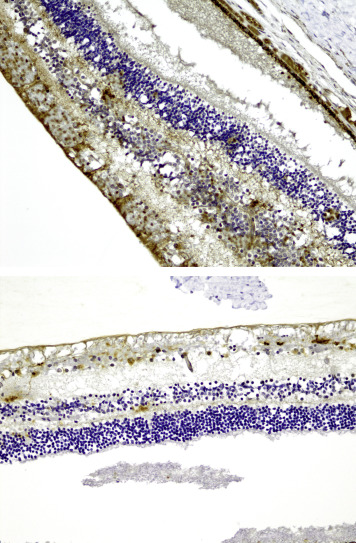
Of the 49 eyes that could be evaluated for β-APP immunostaining in the optic nerve, 21 showed positive β-APP immunostaining in the optic nerve. Fourteen optic nerves showed slight (+) staining (8/25 abusive head trauma eyes vs 6/24 control eyes) ( Figure 4 ). Moderate (++) staining was found in 4 of 25 abusive head trauma optic nerves ( Figure 5 , Top) and in 0 of 24 control optic nerves. Extensive (+++) staining in the optic nerves was found in 3 of 25 abusive head trauma eyes ( Figure 5 , Bottom) and 0 of 24 control eyes. Optic nerve staining did not differ significantly between the 2 groups. Nonetheless, significant correlations were found between optic nerve staining and the severity of retinal hemorrhage (r = 0.473, P = .020), the percentage of the retina affected by retinal hemorrhage (r = 0.682, P = .002), and vitreal hemorrhage (r = 0.543, P = .020). There were no significant correlations with intrascleral peripapillary hemorrhage, macular fold, or subdural and subarachnoid optic nerve sheath hemorrhage.
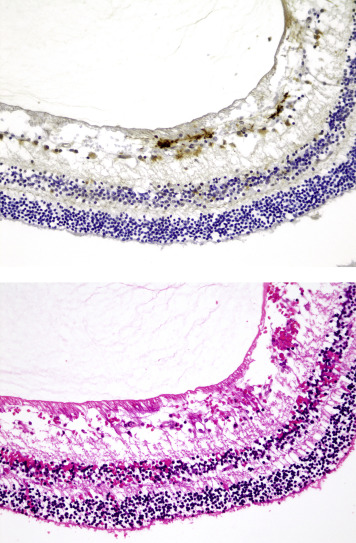
In 8 eyes, axonal bulbs were present in the optic nerves (4/25 abusive head trauma eyes and 4/24 control eyes, Figure 6 ), which was not significantly different between the abusive head trauma and nonabusive head trauma subjects. There was no significant correlation between the presence of axonal bulbs and the presence of subdural or subarachnoid optic nerve sheath hemorrhage.
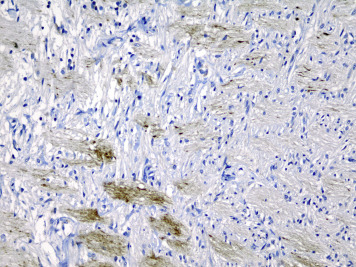
Table 5 shows the positive retinal ubiquitin immunostaining of all cases. More details regarding the ubiquitin immunoreactivity can be found in Supplementary Table 2 (supplemental material available at AJO.com ). There was no significant difference in time interval between presentation to the medical services and death for ubiquitin-positive vs ubiquitin-negative infants.
Of the 74 eyes studied, retinal ubiquitin immunostaining was found in 23 of 42 abusive head trauma eyes and 2 of 32 control eyes (OR 8.8 [CI 2.2–34.5]; P = .002). Positive immunostaining was identified either in the nerve fiber layer or in the inner plexiform layer. A significant correlation was found between retinal hemorrhage and ubiquitin immunostaining in the nerve fiber layer (r = 0.527, P = .001).
Retinal ubiquitin immunostaining in the equator was present in 14 of 42 abusive head trauma eyes and in none of the controls (OR 22.3 [CI 1.4–359.6]; Fisher exact P = .003). Immunoreactivity in the posterior pole could be found in 10 of 42 abusive head trauma eyes vs none of the control eyes (OR 16.1 [CI 1.0–265.2]; P = .036). Immunostaining in the periphery was found in 1 of 42 abusive head trauma eyes and in 1 of 32 control eyes and did not differ significantly. Significant correlation was found between the co-occurrence of retinal hemorrhage and ubiquitin immunostaining in the equator (r = 0.533, P = .001), as well as in the posterior pole (r = 0.577, P < .001), but not between retinal hemorrhage and ubiquitin immunostaining in the periphery.
Seventeen eyes showed retinal ubiquitin immunostaining in the peripapillary region. This was found in 16 of 41 abusive head trauma eyes vs 1 of 32 control eyes (OR 12.5 [CI 1.8–89.3]; P = .016). There was a significant correlation between ubiquitin and β-APP immunostaining in the equator (r = 0.564, P < .001) and the posterior pole (r = 0.570, P < .001) ( Figure 3 , Bottom), but not in the periphery (r = 0.213, P = .205). There was a significant correlation between the retinal ubiquitin and β-APP immunostaining in the nerve fiber layer (r = 0.789, P < .001) and the inner plexiform layer (r = 0.355, P = .031).
Ubiquitin immunostaining was found in 2 optic nerves, both control eyes, which did not differ significantly.
Table 5 displays which cases show increased GFAP immunostaining, identifying gliosis. More specific details on the retinal layers and regions can be found in Supplementary Table 2 (supplemental material available at AJO.com ).
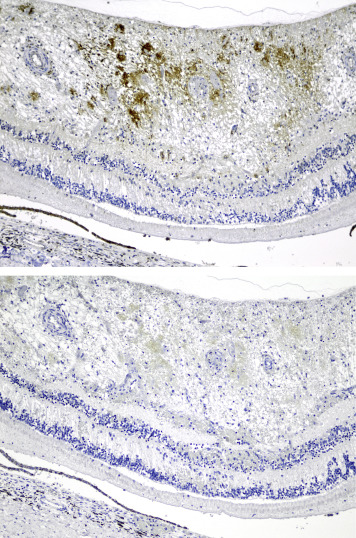
Retinal GFAP immunostaining was found in 20 eyes ( Figure 7 ): 13 of 42 abusive head trauma eyes and 7 of 32 control eyes, which did not differ significantly. There was no significant difference in time interval between presentation to the medical services and death for GFAP-positive vs GFAP-negative cases.